Abstract
Intestinal neuronal dysplasia type B is a controversial entity expressed by complex changes in the enteric nervous system. Diagnosis depends on rectal biopsy histopathology and diagnostic criteria, both qualitative and quantitative, have changed over time, hindering the diagnostic practice. We analyzed the morphological criteria for the histological diagnosis of intestinal neuronal dysplasia type B in a series of patients with intestinal neuronal dysplasia type B according to the 1990 Frankfurt Consensus criteria and verified the applicability of the numerical criteria proposed by Meier-Ruge et al in 2004 and 2006. Qualitative criteria adopted for the histological diagnosis of intestinal neuronal dysplasia type B included hyperplasia of the submucous plexus with hyperganglionosis and hypertrophy of the nerve trunks. Quantitative criteria considered more than 20% giant ganglia in the submucosa, with more than eight neurons each on 25 ganglia, and children aged over 1 year. Distal colon surgical specimens from 29 patients, aged 0–16 years, diagnosed with intestinal neuronal dysplasia type B were retrospectively analyzed using sections processed for conventional histology (H&E) and calretinin immunohistochemistry. Hyperplasia of the submucosal nerve plexi with hyperganglionosis and hypertrophy of the nerve trunks was observed in all cases. Ganglia with small, immature neurons were detected in the majority of cases. Quantitative analysis confirmed hyperganglionosis (mean number=10.7 neurons per ganglion) and hypertrophy of the nerve trunks (median=44.6 μm thickness). Neurons showed immunostaining for calretinin, but neuron counts in calretinin-stained sections were lower compared with H&E (P<0.01). No significant differences were verified between children aged under and over 1 year regarding hyperganglionosis (P=0.79), neuron counts (P=0.36), and immature ganglia (P=0.66). Only one patient met the numerical criteria proposed by Meier-Ruge et al in 2004 and 2006. In conclusion, the numerical criteria showed limited applicability when transposed to conventional histopathology. Children aged over 1 year presented very similar histological features of neuronal immaturity to younger children, questioning the need for an age criterion when diagnosing intestinal neuronal dysplasia type B.
Similar content being viewed by others
Main
Intestinal neuronal dysplasia type B affects the submucosal nerve plexi of the intestine, in isolation or in combination with other neuropathies, such as Hirschsprung’s disease.1, 2 More than 40 years after its original description,3 this pathological condition is yet to be fully elucidated.2, 4
A suspected diagnosis for intestinal neuronal dysplasia type B arises with signs of severe constipation, as observed in newborns, with acute intestinal obstruction, and in older children, with minimal response to conventional treatment.5, 6 These patients should be investigated for possible organic causes of constipation in childhood, particularly Hirschsprung’s disease, the most prevalent of the intestinal dysganglionoses.7
Diagnosing intestinal neuronal dysplasia type B fundamentally depends on histopathological analysis of rectal biopsies.8, 9 However, the morphological criteria for its diagnosis have been extensively altered over the years, which hinders diagnostic practice and comparisons between studies.3, 6, 8, 10, 11, 12, 13 Hyperplasia of the submucosal nerve plexi is the morphological finding that defines intestinal neuronal dysplasia type B, but it is characterized differentially in the proposed criteria.12
The consensus meeting held in Frankfurt in 1990 established diagnostic criteria that have been used in clinical practice, follow-up studies, and genetic investigations.12, 14, 15, 16, 17, 18, 19 These criteria, classified as mandatory and optional, should be determined by qualitative analysis of the nerve plexi by standard histology with hematoxylin and eosin (H&E) and histochemically by acetylcholinesterase.14 On the other hand, new criteria were proposed in the 1990s, emphasizing the need to identify giant ganglia in the submucosa for a diagnosis of intestinal neuronal dysplasia type B.8, 20 These giant ganglia were defined by the presence of a minimum number of ganglion cells, which varied from 6 to more than 10 per ganglion, among the different criteria described.6, 9, 13, 21, 22
Given the lack of diagnostic standardization, Meier-Ruge et al11 proposed the use of quantitative analysis for the histopathological diagnosis of intestinal neuronal dysplasia type B, which underwent certain changes in 2006.12 Although these quantitative criteria have been accepted in the scientific community,1, 2, 4 there are few reports of their use in large series of patients.23 The requirement of fresh material for frozen sections and specific histochemical stains, the availability of which may be restricted in some centers, are limitations that should be considered. Moreover, it has not been established whether these same numerical criteria can be applied to 5 μm histological sections, embedded in paraffin, for analysis by standard histology (H&E) or to immunohistochemical studies.2, 12
Recently, several immunohistochemical markers have been used in the diagnosis of intestinal dysganglionoses, particularly for Hirschsprung’s disease.24, 25, 26, 27, 28 Calretinin is a vitamin D-dependent 29-kDa protein that binds to calcium and has an important role as a sensor and modulator of calcium ions.29 The absence of this protein leads to the accumulation of these ions in the cytoplasm of nerve cells, promoting hyperexcitability and neurodegeneration.30, 31 Cell bodies of neurons in the extrinsic enteric nervous system, present in the submucosal and myenteric plexi, and nerve fibers of the extrinsic enteric nervous system present in the lamina propria and muscularis mucosa show immunohistochemical expression for calretinin.29, 32 Thus, immunohistochemistry for calretinin was established as a valuable method in the diagnostic investigation of Hirschsprung’s disease, demonstrating loss of protein immunostaining in the ganglion cells of the nerve plexi and mucosal nerve fibers.1, 27, 28, 30, 31, 33, 34 However, no descriptions of the immunohistochemical expression profile for calretinin have been reported for cases of intestinal neuronal dysplasia type B.
For these reasons, we aimed to analyze the morphological characteristics of the enteric nervous system in a series of cases previously diagnosed as intestinal neuronal dysplasia type B using the criteria proposed by the 1990 Frankfurt Consensus,14 by standard histology (H&E) and calretinin immunohistochemistry, and verify the applicability of the numerical criteria proposed by Meier-Ruge et al11, 12 for this end.
Materials and methods
Patients
A retrospective study was conducted on surgical specimens of patients submitted to transanal endorectal pull-through between 1992 and 2012, at the Clinics Hospital of the Botucatu Medical School of São Paulo State University (FMB-UNESP). During this period, 113 patients under 18 years of age were submitted to transanal endorectal pull-through due to dysganglionoses. All patients with the histopathological diagnosis of intestinal neuronal dysplasia type B (n=29), based on the criteria proposed by the 1990 Frankfurt Consensus,14 were included in the study. Surgical specimens of patients diagnosed with additional associated dysganglionoses, like Hirschsprung’s disease, were excluded from the study. This study was approved by the Research Ethics Committee of FMB-UNESP, under protocol no. CAAE 46521315.2.0000.5411.
Processing of the Surgical Specimens
The surgical specimens of patients submitted to transanal endorectal pull-through were collected while fresh and transported immediately postsurgery to the Department of Pathology, where they were opened along the antimesenteric border, washed with water, fixed to a rigid surface to maintain their shape, and immersed in 10% formalin solution. After 24 h, longitudinal cuts were made along the specimens. These cuts were sectioned into smaller pieces of 2 cm each and placed in numbered cassettes. For this study, the rectal distal fragments, 2 cm fragments of the specimens, were recovered and new histological sections were made and processed for standard histology (H&E) and calretinin immunohistochemistry.
Standard Histology (H&E)
Serial histological sections of 5 μm were made and stained with H&E. Histopathological analysis of these histological sections sought to characterize the structures of the submucosal nerve plexi.
Calretinin Immunohistochemistry
The calretinin immunohistochemical study was performed on 4-μm-thick histological sections, using the FLEX monoclonal mouse anti-human calretinin antibody, DAK-Calret 1 clone, prediluted (0.015 mol/l) ready for use in the DakoAutostainer system. The avidin–biotin–peroxidase protocol model was used, following the manufacturer’s recommendations.35 The cells labeled by the antibody exhibit brownish cytoplasmic and nuclear staining.
Histopathological Analysis
The histopathological analysis was performed first by H&E, followed by calretinin immunohistochemistry, to characterize the submucosal nerve plexi through qualitative and quantitative analysis. The 1990 Frankfurt criteria used included hyperplasia of the submucous plexus, giant ganglia (hyperganglionosis), bud-like nerve cell groups along nerve trunks, increased acetylcholinesterase activity on initial rectal biopsies, and heterotopic neurons. Table 1 summarizes the qualitative and quantitative criteria adopted for the histopathological analysis by H&E, according to the Frankfurt criteria,14 Meier-Ruge et al,11, 12 and Koletzko et al.36 Nerve ganglia with small neurons, with nuclear hyperchromasia, were interpreted as signs of immaturity. A submucosal nerve plexus with more than 8 neurons was considered as a giant, hyperplastic ganglion.
Calretinin immunostaining was assessed in neurons in the submucosal nerve plexi and at heterotopic sites in the lamina propria and near to the muscularis mucosa. Immunostaining for calretinin allowed to identify nerve fibers in the lamina propria of the mucosa and was graded as focal expression (+) visualized only at x400 magnification, focal expression (++) visualized from x200 magnification and diffuse expression (+++) visualized from x200 magnification.
Morphometric analysis was performed using images obtained by the Software Pannoramic Viewer (version: 1.15.2 SP 2; developed by 3DHistech). The numerical criteria proposed by Meier-Ruge et al11, 12 were applied in all cases, both in conventional histological analysis by H&E and in the immunohistochemical analysis by calretinin. According to these numerical criteria, a diagnosis of intestinal neuronal dysplasia type B depends on the presence of at least 20% giant ganglia, with more than eight neurons among 25 submucosal ganglia analyzed.11, 12 The histopathological analysis was performed jointly by two medical pathologists with expertise in the diagnosis of intestinal dysganglionoses.
Statistical Analysis
The results of the histopathological analysis were compared with the method used (standard H&E histology and calretinin immunohistochemistry) and between age groups (children aged < or >1 year), based on the age cutoff proposed by the criteria of Meier-Ruge et al.11, 12 The Stats Direct 3 software was used for statistical comparison of the different methods and age groups. Categorical variables were analyzed by the Fisher's exact test to compare the variables of qualitative analysis between the two age groups. After application of the Shapiro–Wilk normality test, the continuous quantitative variables used to compare the different methods of histopathology were analyzed by the Student's t-test for paired samples or the Wilcoxon's test, while for the variables used to compare the different age groups, the Student's t-test for independent samples or the Mann–Whitney test were used. Agreement between the methods of histopathology was determined by the intraclass correlation coefficient.37, 38
The relationship between number of giant ganglia, age group (< or >1 year), and the presence of ganglia with signs of immaturity was investigated by a generalized linear model with gamma probability distribution and identity link function (robust estimator), using the SPSS 22.0 software. The significance level adopted for the tests used was 5%.
Results
The diagnosis of intestinal neuronal dysplasia type B was achieved after examining the surgical specimens in 29 cases. The median age of patients at surgery was 26 months (1–199 months). Ten patients (34.5%) were <1 year and 19 (65.5%) were >1 year. Nineteen patients (65.5%) were male and 10 (34.5%) were female.
Histopathological Analysis of the Surgical Specimens by H&E
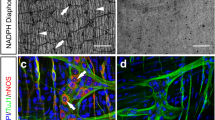
In all cases, the submucosal nerve plexi showed morphological alterations, characterized by hyperplasia of the nerve plexi due to increased number of neurons and hypertrophy of the parasympathetic nerve trunks, with bud-like arrangement of neurons along nerve fibers (Figure 1). The majority of cases showed ganglia with signs of immaturity characterized by small neurons with hyperchromatic nuclei (Table 2). It was not possible to identify conclusively the presence of heterotopic neurons in the lamina propria and muscularis mucosa using H&E staining. Table 2 summarizes the qualitative results of the histopathological analysis by H&E.
Quantitative analysis by standard histology (H&E) confirmed qualitative observation of hyperganglionosis with a mean number of 10.7 (±4.1) neurons per ganglion and hypertrophy of the nerve trunks with median thickness of 44.6 μm (Table 3).
Analysis of Nerve Plexi by Calretinin Immunohistochemistry
Immunostaining for calretinin was observed in neurons of the submucosal nerve plexi in all cases analyzed (Figure 2a). However, not all the neurons previously identified by standard histology (H&E) were stained by calretinin. Immunohistochemical analysis by calretinin enhanced the identification of heterotopic neurons in 26/29 cases (89.7%), 14 of which presented neurons in the lamina propria and the remaining 12 located in the muscularis mucosa (Table 2 and Figure 2b). Immunohistochemical expression of calretinin was observed in nerve fibers in the lamina propria and muscularis mucosa (Figure 2b). This expression showed considerable variation between patients (Table 2), with focal expressions being the most common.
Patterns of calretinin immunostaining in intestinal neuronal dysplasia type B. (a) Positive nuclear and cytoplasmic calretinin staining in neurons from a submucosal nerve plexus (calretinin, x400). (b) Positive staining in dark brown in nerve fibers in the lamina propria. Heterotopic neuron in the muscularis mucosa (arrow) (calretinin, x400).
Application of the Numerical Criteria Proposed by Meier-Ruge et al11,12
The numerical criteria for the diagnosis of intestinal neuronal dysplasia type B proposed by Meier-Ruge et al11, 12 were applied to 5 μm sections stained with H&E and 4 μm sections processed for calretinin immunohistochemistry. Two cases achieved the required minimum of 20% giant ganglia by H&E staining and only one case by calretinin immunohistochemistry. However, considering the cutoff age of 1 year recommended by these quantitative criteria, only one case fulfilled the required criteria proposed by Meier-Ruge et al.11, 12
Correlation Between the Results of Quantitative Analysis by H&E and Calretinin
Comparison between H&E staining and calretinin immunostaining showed that, for all quantitative assessment, analysis by calretinin identified a smaller number of neurons, with significant differences (P<0.01) in the number of neurons identified in 25 ganglia (H&E 92.8±22.4 vs calretinin 75.8±18.1). Concordance analysis determined by the intraclass correlation coefficient showed that the results obtained by these two methods of analysis had poor levels of agreement, 0.09 (−0.17–0.38). However, the number of fields required to identify 25 ganglia was significantly lower (P<0.01) when using calretinin immunohistochemistry (H&E 18.0±2.4 vs calretinin 13.7±3.0).
Comparison Between the Different Age Groups
The results of the histopathological analysis were divided into two groups according to age at surgery: children aged <1 year and children aged >1 year. Qualitative analysis of the plexi by H&E staining identified the presence of nerve ganglia showing signs of neuronal immaturity and anisomorphic neurons in both age groups (Table 2). No statistically significant differences were verified between the two age groups for any of the morphological characteristics analyzed by H&E staining and calretinin immunohistochemistry (Table 2).
No significant differences were observed between the two age groups for the neuronal quantitative variables analyzed by H&E staining (Table 3). The number of giant ganglia was significantly higher in infants presenting ganglia with signs of immaturity (P<0.01). Although the number of giant ganglia was higher in children aged >1 year, no significant difference in their distribution by age group was determined (P=0.56), which indicates that the main factor associated with the presence of giant ganglia was ganglia with small, immature neurons, rather than the age group (Table 4).
Discussion
Despite intensive scientific research conducted since its original description in the 1970s3 and more than 250 scientific articles published, lacunas remain concerning the definition, pathogenesis, diagnostic criteria, and therapeutic possibilities of intestinal neuronal dysplasia type B.2, 6, 12 On the other hand, in medical practice, we continue to identify children with severe constipation or intestinal obstruction, who are subject to diagnostic investigation for intestinal dysganglionoses by rectal biopsy and who present hyperplasia of submucosal nerve plexi consistent with a diagnosis of intestinal neuronal dysplasia type B.12
One of the biggest challenges is the lack of consensus regarding the criteria for a histopathological diagnosis of intestinal neuronal dysplasia type B. The most recent proposal is the quantitative criteria developed by Meier-Ruge et al.11, 12 This proposal has certain limitations, like the need for fresh samples to produce 15 μm frozen sections, stained by specific histochemical reactions that are available to few diagnostic centers. Moreover, this proposal considers hyperganglionosis as the main diagnostic criterion, defined by the minimum numerical value of 20% giant ganglia, each presenting with more than eight nerve cells for 25 ganglia examined among the submucosal nerve plexi. They also prose a minimum age of 1 year, which limits further the diagnosis of this pathological entity. Given their specificity, there are few reports of the applicability of these criteria in series of patients diagnosed with intestinal neuronal dysplasia type B.12, 23
In this study, we examined surgical specimens from patients with a previous diagnosis of intestinal neuronal dysplasia type B, established by the criteria proposed in the 1990 Consensus of Frankfurt.14 Only 2 of the 29 cases analyzed achieved the numerical values required by the criteria proposed by Meier-Ruge et al.11, 12 Of these cases, only one child was >1 year at the time of surgery and thus fulfilled age cutoff requirement for these diagnostic criteria.11, 12 Similar results were reported by Taguchi et al,23 in a retrospective multicenter study of cases of intestinal neuronal dysplasia type B in 167 centers in Japan, from 2000 to 2009. Thirteen cases of intestinal neuronal dysplasia type B were diagnosed using morphological criteria common to all the centers. However, when the authors used the numerical criteria proposed by Meier-Ruge et al,11, 12 only 4 of these 13 cases maintained a diagnosis of intestinal neuronal dysplasia type B.23 Thus, these quantitative criteria seem to be considerably restrictive when applied to conventional histopathology performed using 5 μm sections stained with H&E. Histological sections of greater thickness, stained with techniques that facilitate the identification of nerve cells, clearly influence the quantitative determination of ganglion neurons of the submucosa, which could explain the low number of cases that achieved the numerical criteria in our series. Since conventional histopathological analysis by H&E is the most common method used in the diagnostic investigation of rectal biopsies for intestinal dysganglionoses, it is of fundamental importance that numerical values for quantitative criteria are established, or that the histopathological diagnosis of intestinal neuronal dysplasia type B is qualitative in these situations, based on morphological changes that characterize this pathological entity.
In this case series, histopathological analysis enabled the morphological characterization of intestinal neuronal dysplasia type B, highlighting changes like anisomorphic neurons, neuronal ectopia in the lamina propria and muscularis mucosa and ganglia with signs of immaturity such as small neurons (Table 2). These findings have been verified in other case series and form part of previously proposed criteria for the diagnosis of intestinal neuronal dysplasia type B.14, 16, 36, 39 Quantitative analysis of the submucosal nerve plexi confirmed hyperganglionosis, with a maximum mean number of 10.7 neurons per ganglion and revealed that the majority of the cases presented hypertrophic nerve trunks, with a median thickness of 44.6μm. This last finding, characteristic of Hirschsprung’s disease, is rarely discussed in intestinal neuronal dysplasia type B and can be considered as one of the criteria for its diagnosis.8, 14, 36, 40, 41, 42, 43
When comparing different age groups, based on the cutoff age of 1 year, no statistically significant differences were observed in the qualitative analysis of the morphological changes verified in the submucosal nerve plexi (Table 2). Despite evidence that the histopathological changes suggestive of immaturity could change with age, through maturation and apoptosis,21, 36, 44, 45, 46 our results showed that these changes were present in both age groups and call into question the need to establish an age cutoff for the diagnosis of intestinal neuronal dysplasia type B. Moreover, quantitative analysis of the plexi showed no significant differences between the groups regarding the characterization of hyperganglionosis, defined as the total number of neurons in 25 ganglia and by the maximum number of neurons per ganglion (Table 3). The number of giant ganglia identified was influenced more by the presence of signs of neuronal immaturity than by patient age (Table 4).
Calretinin immunohistochemical analysis gained prominence in recent years, establishing itself as a key method in the diagnostic investigation of Hirschsprung’s disease, which is characterized by total loss of expression of this protein.27, 28, 30, 31, 33, 34 Since one of the main differential diagnoses of Hirschsprung’s disease is intestinal neuronal dysplasia type B, knowledge concerning the immunohistochemical expression profile for calretinin in intestinal neuronal dysplasia type B is essential. Herein, we observed calretinin expression in all the cases analyzed, both in the neurons of the submucosal nerve plexi and the nerve fibers of mucosa (Table 2), in contrast to that observed in Hirschsprung’s disease.27, 28, 30, 31, 33, 34 This expression also occurred in ectopic neurons present in the lamina propria and muscularis mucosa, substantially facilitating their identification, which is part of the morphological findings present in intestinal neuronal dysplasia type B16, 36, 39 (Figure 2). The identification of these heterotopic neurons is limited in analysis by standard histology, as difficulties exist in differentiating them from other structures, such as inflammatory and stromal cells.
Agreement between the quantitative analyzes obtained by H&E and calretinin was considered poor. Furthermore, calretinin immunohistochemistry identified fewer neurons than H&E staining. This is justified by the difficulties involved in determining the limits of the nerve cells when using calretinin immunohistochemistry (Figure 2) and the fact that not all neurons in the enteric nervous system present immunostaining for calretinin.47 In their study of surgical specimens from 28 patients, Beuscher et al48 showed that 79% of the neurons of the submucosal nerve plexi located in the colon were immunostained for calretinin and vasoactive intestinal peptide and this percentage varied according to intestinal segment analyzed. Therefore, calretinin immunostaining may be of help in the diagnosis of intestinal neuronal dysplasia type B, but is not as useful as it is for the diagnosis of Hirschsprung’s disease.
In conclusion, our study showed that the numerical criteria proposed by Meier-Ruge et al11, 12 for the analysis of 15 μm sections stained by specific histochemical reactions is of limited applicability when transposed to conventional histopathology performed on 5 μm sections stained with H&E. Our study also showed that children aged >1 year presented similar histological features to younger children, both in the qualitative and quantitative analysis of nerve plexi, questioning the need for the applicability of the minimum age cutoff of 1 year when diagnosing intestinal neuronal dysplasia type B.
References
Knowles CH, De Giorgio R, Kapur RP et al, The London Classification of gastrointestinal neuromuscular pathology: report on behalf of the Gastro 2009 International Working Group. Gut 2010; 59:882–887.
Schäppi MG, Staiano A, Milla PJ et al, A practical guide for the diagnosis of primary enteric nervous system disorders. J Pediatr Gastroenterol Nutr 2013;57:677–686.
Meier-Ruge WA . Casuistic of colon disorder with symptoms of Hirschsprung's disease (author’s transl). Verh Dtsch Ges Pathol 1971;55:506–510.
de Arruda Lourenção PL, Terra SA, Ortolan EVP et al, Intestinal neuronal dysplasia type B: a still little known diagnosis for organic causes of intestinal chronic constipation. World J Gastrointest Pharmacol Ther 2016;7:397–405.
Csury L, Peña A . Intestinal neuronal dysplasia. Pediatr Surg Int 1995;10:441–446.
Holschneider AM, Puri P, Homrighausen LH et al. Intestinal neuronal malformation (IND): clinical experience and treatment. In: Holschneider AM, Puri P (eds). Hirschsprung’s Disease and Allied Disorders, 3rd edn. Springer: Berlin, Heidelberg, Germany and New York, NY, USA, 2008, pp 229–251.
Tabbers MM, Di Lorenzo C, Berger MY et al, European Society for Pediatric Gastroenterology, Hepatology, and Nutrition; North American Society for Pediatric Gastroenterology. Evaluation and treatment of functional constipation in infants and children: evidence-based recommendations from ESPGHAN and NASPGHAN. J Pediatr Gastroenterol Nutr 2014;58:258–274.
Meier-Ruge WA, Brönnimann PB, Gambazzi F et al, Histopathological criteria for intestinal neuronal dysplasia of the submucosal plexus (type B). Virchows Arch 1995;426:549–556.
Kobayashi H, Hirakawa H, Puri P . What are the diagnostic criteria for intestinal neuronal dysplasia? Pediatr Surg Int 1995;10:459–464.
Schärli AF, Meier-Ruge W . Localised and disseminated forms of neuronal intestinal dysplasia mimicking Hirschsprung’s disease. J Pediatr Surg Int 1981;16:164–170.
Meier-Ruge WA, Ammann K, Bruder E et al, Updated results on intestinal neuronal dysplasia (IND B). Eur J Pediatr Surg 2004;14:384–391.
Meier-Ruge WA, Bruder E, Kapur RP . Intestinal neuronal dysplasia type B: one giant ganglion is not good enough. Pediatr Dev Pathol 2006;9:444–452.
Mahesha V, Saikia UN, Shubha AV et al, Intestinal neuronal dysplasia of the myenteric plexus – new entity in humans? Eur J Pediatr Surg 2008;18:59–60.
Borchard F, Meier-Ruge W, Wiebecke B et al, Disorders of the innervation of the large intestine – classification and diagnosis. Results of a consensus conference of the Society of Gastroenteropathology, 1 December 1990 in Frankfurt. Pathologe 1991;12:171–174.
Schärli AF . Neuronal intestinal dysplasia. Pediatr Surg Int 1992;7:2–7.
Koletzko S, Ballauff A, Hadziselimovic F et al, Is histological diagnosis of neuronal intestinal dysplasia related to clinical and manometric findings in constipated children? Results of a pilot study. J Pediatr Gastroenterol Nutr 1993;17:59–65.
Sacher P, Briner J, Hanimann B . Is neuronal intestinal dysplasia (NID) a primary disease or a secondary phenomenon? Eur J Pediatr Surg 1993;3:228–230.
Costa M, Fava M, Seri M et al, Evaluation of the HOX11L1 gene as a candidate for congenital disorders of intestinal innervation. J Med Genet 2000;37:E9.
Martucciello G, Torre M, Pini Prato A et al, Associated anomalies in intestinal neuronal dysplasia. J Pediatr Surg 2002;37:219–223.
Meier-Ruge WA, Schmidt PC, Stoss FS . Intestinal neuronal dysplasia and its morphometric evidences. Pediatr Surg Int 1995;10:447–453.
Schofield DE, Yunis EJ . Intestinal neuronal dysplasia. J Pediatr Gastroenterol Nutr 1991;12:182–189.
Moore SW, Laing L, Kaschula ROC et al, A histological grading system for the evaluation of co-existing NID with Hirschsprung's disease. Eur J Pediatr Surg 1994;4:293–297.
Taguchi T, Kobayashi H, Kanamori Y et al, Isolated intestinal neuronal dysplasia Type B (IND-B) in Japan: results from a nationwide survey. Pediatr Surg Int 2014;30:815–822.
Nogueira A, Campos M, Soares-Oliveira M et al, Histochemical and immunohistochemical study of the intrinsic innervation in colonic dysganglionosis. Pediatr Surg Int 2001;17:144–151.
Kapur RP . Can we stop looking? Immunohistochemistry and the diagnosis of Hirschsprung’s disease. Am J Clin Pathol 2006;126:9–12.
Geramizadeh B, Akbarzadeh E, Izadi B et al, Immunohistochemical study of enteric nervous system in Hirschsprung’s disease and intestinal neuronal dysplasia. Histol Histopathol 2013;28:345–351.
de Arruda Lourenção PL, Takegawa BK, Ortolan EV et al, A useful panel for the diagnosis of Hirschsprung disease in rectal biopsies: calretinin immunostaining and acetylcholinesterase histochesmistry. Ann Diagn Pathol 2013;17:352–356.
de Arruda Lourenção PL, Takegawa BK, Ortolan EV et al, Does calretinin immunohistochemistry reduce inconclusive diagnosis in rectal biopsies for Hirschsprung disease? J Pediatr Gastroenterol Nutr 2014;58:603–607.
Walters JR, Bishop AE, Facer P et al, Calretinin and calbidin – D28k immunoreactivity in the human gastrointestinal tract. Gastroenterology 1993;104:1381–1389.
Barshack I, Fridman E, Goldberg I et al, The loss of calretinin expression indicates aganglionosis in Hirschsprung’s disease. J Clin Pathol 2004;57:712–716.
Kapur RP, Reed RC, Finn LS et al, Calretinin immunohistochemistry versus acetylcholinesterase histochemistry in the evaluation of suction rectal biopsies for Hirshsprung disease. Pediatr Dev Pathol 2009;12:6–15.
Wattchow DA, Porter AJ, Brookes SJ et al, The polarity of neurochemically defined myenteric neurons in the human colon. Gastroenterology 1997;113:497–506.
Guinard-Samuel V, Bonnard A, De Lagausie P et al, Calretinin immunohistochemistry: a simple and efficient tool to diagnose Hirschsprung’s disease. Mod Pathol 2009;22:1379–1384.
Holland SK, Ramalingam P, Podolsky RH et al, Calretinin immunostaining as an adjunct in the diagnosis of Hirshsprung disease. Ann Diagn Pathol 2011;15:323–328.
Dako Autostainer PlusUserGuide, 2007. Available at: http://www.dako.jp/0003107_rev_d_man_user_guide_autostainer_plus_english.pdf.
Koletzko S, Jesch I, Faus-Kebetaler T et al, Rectal biopsy for diagnosis of intestinal neuronal dysplasia in children: a prospective multicentre study on interobserver variation and clinical outcome. Gut 1999;44:853–861.
Muller R, Buttner P . A critical discussion of intraclass correlation coefficients. Stat Med 1994;13:2465–2476.
Lee MK, Lee J, Chung CY et al, Pitfalls and important issues in testing reliability using intraclass correlation coefficients in orthopaedic research. Clin Orthop Surg 2012;4:149–155.
Montedonico S, Acevedo S, Fadda B . Clinical aspects of intestinal neuronal dysplasia. J Pediatr Surg 2002;37:1772–1774.
Kapur RP . Calretinin-immunoreactive mucosal innervation in very short-segment hirschsprung disease: a potentially misleading observation. Pediatr Dev Pathol 2014;17:28–35.
Meier-Ruge W . Epidemiology of congenital innervation defects of the distal colon. Virchows Arch A 1992;420:171–177.
Munakata K, Fukuzawa M, Nemoto N . Histologic criteria for the diagnosis of allied diseases of Hirschsprung’s disease in adults. Eur J Pediatr Surg 2002;12:186–191.
Fadda B, Maier WA, Meier-Ruge W et al, Neuronal intestinal dysplasia – critical 10-years’ analysis of clinical and biopsy diagnosis. Z Kinderchir 1983;38:305–311.
Wester T, O’Briain S, Puri P . Morphometric aspects of the submucous plexus in whole-mount preparations of normal human distal colon. J Pediatr Surg 1998;33:619–622.
Cord-Udy CL, Smith VV, Ahmed S et al, An evaluation of the role of suction rectal biopsy in the diagnosis of intestinal neuronal dysplasia. J Pediatr Gastroenterol Nutr 1997;24:1–6.
Coerdt W, Michel JS, Rippin G et al, Quantitative morphometric analysis of the submucous plexus in age-related control groups. Virchows Arch 2004;444:239–246.
Burcher E, Kandiah CJ . Neurochemical classification of myenteric neurons in the guinea pig ileum. Neuroscience 1996;75:949–967.
Beuscher N, Jabari S, Strehl J et al, What neurons hide behind calretinin immunoreactivity in the human gut? Histochem Cell Biol 2014;141:393–405.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Terra, S., de Arruda Lourenção, P., G Silva, M. et al. A critical appraisal of the morphological criteria for diagnosing intestinal neuronal dysplasia type B. Mod Pathol 30, 978–985 (2017). https://doi.org/10.1038/modpathol.2017.4
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2017.4
This article is cited by
-
Dedicated macrophages organize and maintain the enteric nervous system
Nature (2023)
-
Identification of potential molecular pathogenesis mechanisms modulated by microRNAs in patients with Intestinal Neuronal Dysplasia type B
Scientific Reports (2019)
-
Abnormal serum vitamin A levels and retinoic acid receptor α expression patterns in children with anorectal malformation
Pediatric Surgery International (2019)