Abstract
The malignant peripheral nerve sheath tumor is a relatively uncommon type of soft tissue sarcoma arising from a peripheral nerve or extraneural soft tissues and showing nerve sheath differentiation. The diagnosis of malignant peripheral nerve sheath tumor is one of the most challenging tasks in surgical pathology because of its uncommon type (5–10% soft tissue sarcomas), morphologic resemblance to other spindle cell neoplasms and lack of sensitive and specific immunohistochemical markers. The pathologic diagnosis is more straightforward in the clinical setting of neurofibromatosis-1, but problems are mainly centered on the non-neurofibromatosis-1 malignant peripheral nerve sheath tumors. To date, S100 protein is the most widely applied marker in the case of a suspected malignant peripheral nerve sheath tumor, yet its suboptimal sensitivity and its expression in other spindle cell neoplasms, including spindle cell melanoma, clear-cell sarcoma, leiomyosarcoma and monophasic synovial sarcoma, add to the diagnostic conundrum. Growth-associated protein 43 (GAP43), a membrane-associated phosphoprotein expressed in neuronal growth cones and Schwann cell precursors during neural development and axonal regeneration, was applied to a set of nerve sheath and non-nerve sheath spindle cell neoplasms. The findings in this study indicate that GAP43 is expressed in malignant peripheral nerve sheath tumors (n=18/21; 86%) and demonstrates a sensitivity superior to S100 protein (n=13/21; 62%). GAP43 is also positive in neurofibromas (n=17/18; 94%), schwannomas (n=11/12; 92%) and desmoplastic melanomas (n=7/10; 70%). In contrast, it is negative in the non-desmoplastic spindle cell melanomas (n=20/22; 91%). Of the other non-neural soft tissue sarcomas, GAP43 is non-reactive in most leiomyosarcomas (n=14/16; 88%) and clear-cell sarcomas (n=8/8), and only focally positive in monophasic synovial sarcomas (n=3/7; 43%). GAP43 is seemingly a highly sensitive marker for peripheral nerve sheath tumors and may serve as a useful diagnostic adjunct in the diagnosis of malignant peripheral nerve sheath tumor from other spindle cell neoplasms, including spindle cell melanoma.
Similar content being viewed by others
Main
Malignant spindle cell neoplasms, as determined by the presence of pleomorphic spindle cells, mitotic figures, often numerous and atypical, and necrosis, are with few exceptions a diagnostic challenge to the pathologist. The primary site may provide the most useful clue to the nature of the neoplasm, as in the case of a sarcomatoid carcinoma of the kidney or lung, but a metastasis from either one of these tumors can be diagnostically problematic. On the other hand, the amelanotic spindle cell melanoma is the quintessential mimic of a spindle cell sarcoma with the well-documented morphologic and immunophenotypic overlap with malignant peripheral nerve sheath tumor.1
Malignant peripheral nerve sheath tumor is one among several spindle cell sarcomas from which it should be differentiated for the sake of diagnostic accuracy, prognosis and therapeutic management in some cases. When a malignant peripheral nerve sheath tumor arises in the setting of neurofibromatosis-1 (overall lifetime risk of 10%), the sarcoma often arises within a peripheral nerve involved by a neurofibroma.2 However, other non-neural sarcomas occur infrequently in individuals with neurofibromatosis-1. The long-standing diagnostic problem is the sporadic malignant peripheral nerve sheath tumor whose anatomic relation to a peripheral nerve is either uncertain or not present by imaging studies or intraoperative observation. The subsequent pathologic examination may reveal the presence of a nerve in direct proximity to the sarcoma, which has microscopic and immunophenotypic features of a malignant peripheral nerve sheath tumor.
The archetypical histologic pattern of a malignant peripheral nerve sheath tumor is a uniform high-grade spindle cell sarcoma with the so-called herringbone and/or fascicular architecture. A light cell–dark cell pattern of interlacing fascicles is composed of spindle cells, which are more or less compactly apposed allowing light to pass through the less compactly cellular foci. Monophasic synovial sarcoma is the most proximate mimic of malignant peripheral nerve sheath tumor as we have learned, but immunohistochemistry and molecular studies can resolve the question of a synovial sarcoma.3 The rare glandular malignant peripheral nerve sheath tumor may also have a resemblance to the biphasic synovial sarcoma.4
Leiomyosarcoma, adult-type fibrosarcoma, dermatofibrosarcoma protuberans, low-grade fibromyxoid sarcoma (in the case of a low-grade malignant peripheral nerve sheath tumor), spindle cell rhabdomyosarcoma, clear-cell sarcoma (also known as melanoma of soft parts) and spindle cell melanoma are the other considerations in the differential diagnosis of a malignant peripheral nerve sheath tumor. Obviously, each of these other spindle cell neoplasms has clinical and/or pathologic features to influence the selection of ancillary immunohistochemical and/or molecular genetic studies.
The foundation of the usual diagnostic evaluation beyond light microscopy in the case of a malignant spindle cell neoplasm is immunohistochemistry. Rodriguez and co-workers5 summarized the difficulties in the pathologic diagnosis of the malignant peripheral nerve sheath tumor owing to the ‘lack of entirely specific morphological criteria and/or ancillary immunohistochemical or molecular tests.’ There is general acknowledgement that S100 protein is neither lineage-specific nor consistently expressed by malignant peripheral nerve sheath tumors.6 Nonetheless, before a superior antibody becomes available, S100 protein, with all its limitations, remains the first-line marker in the case of a suspected peripheral nerve sheath tumor. Other markers such as CD57 (Leu 7), neurofilament and collagen type IV are often utilized in the immunohistochemical panels to corroborate or exclude the diagnosis of a suspected malignant peripheral nerve sheath tumor, albeit with limited sensitivity and specificity.7, 8
This study evaluates the diagnostic applicability of the neural developmental membrane-associated phosphoprotein, growth-associated protein 43 (GAP43), to a group of peripheral nerve sheath tumors, including 21 malignant peripheral nerve sheath tumors and a set of benign nerve sheath tumors, selected non-neural soft tissue sarcomas and the problematic spindle cell melanomas.
Materials and methods
Case Selection
Archival formalin-fixed, paraffin-embedded tissue blocks and slides were retrieved from the archival files of the Lauren V Ackerman Laboratory of Surgical Pathology, Washington University Medical Center (St Louis, MO, USA). All cases were de-identified, and histologic slides were independently reviewed by the study pathologists.
A total of 129 specimens were included in this study: (1) tumors of presumed peripheral nerve sheath type: 21 malignant peripheral nerve sheath tumors, 12 neurofibromas, 6 plexiform neurofibromas and 12 schwannomas; (2) tumors of melanocytic lineage: 22 spindle cell melanomas (non-desmoplastic type: 16 primary, 6 metastatic) and 10 desmoplastic melanomas; (3) tumors of uncertain differentiation: 7 monophasic synovial sarcomas, 8 clear-cell sarcomas; (4) 16 leiomyosarcomas; and (5) 15 normal tissue types from various organs, including the peripheral nerves, skin, skeletal muscle, adipose tissue, salivary gland, pharyngeal tonsil, thyroid gland, adrenal gland, lung, small and large intestines, pancreas, liver, uterine cervix and testis. Patient demographics (age, gender, tumor diagnosis and location) are detailed in Table 1.
All selected cases of malignant peripheral nerve sheath tumor occurred either in the clinical setting of confirmed neurofibromatosis-1 (n=20) or arose in a long-standing neurofibroma (n=1); these cases were selected as indisputable examples of malignant peripheral nerve sheath tumor to avoid any questions about the diagnosis of the latter. The examples of desmoplastic melanomas, including both mixed and pure types, were based on established morphologic criteria.9 All cases of synovial sarcomas harbored the SYT-SSX t(X;18) chromosomal translocation, and all cases of clear-cell sarcomas harbored the EWS break apart by fluorescent in situ hybridization (FISH).
Approval for this study was obtained from the Institutional Review Board of Washington University in St Louis.
Immunohistochemistry
Immunoperoxidase studies were performed on whole tissue sections using the Ventana Benchmark XT automated immunostainer (Ventana Medical Systems, Tucson, AZ, USA) according to standard protocols after antigen retrieval. The antibodies used in this study were anti-GAP43 (1:1000; Sigma-Aldrich) and anti-S100 protein (prediluted from Ventana) antibodies. The pattern and localization of staining were evaluated in the context of the histologic features. GAP43 and S100 protein expression was scored as negative (−), or semiquantitatively scored as 1+ (5–25% of the tumor cells positive), 2+ (25–50%) and 3+ (>50%) by visual confirmation of membranous expression. Each immunostain was independently reviewed by the study pathologists.
Statistics
The statistical association between each antibody and the tumors tested was analyzed using Fisher’s exact test. A P-value <0.05 was considered statistically significant.
Results
GAP43 Staining in Neoplasms of Peripheral Nerve Sheath Type
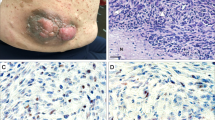
GAP43 immunoreactivity in the various tumors of peripheral nerve sheath type is summarized in Tables 2 and 3. Of the 21 cases of malignant peripheral nerve sheath tumors examined, two showed GAP43 positivity in 5–25% of tumor cells (n=2/21; 10%), four with positivity in 25–50% of tumor cells (n=4/21; 19%) and 12 with positivity in >50% of tumor cells (n=12/21; 57%) (Figures 1a, b, d, e, g and h). Overall, GAP43 immunoreactivity was observed in the majority of malignant peripheral nerve sheath tumors (n=18/21; 86%).
Malignant peripheral nerve sheath tumor. (a) Malignant peripheral nerve sheath tumor case no. 4, hematoxylin and eosin. (b) Growth-associated protein 43 (GAP43) staining strongly highlights 25–50% of tumor cells. (c) S100 protein shows a similar staining pattern to GAP43. (d) Malignant peripheral nerve sheath tumor case no. 12, hematoxylin and eosin. (e) GAP43 staining strongly highlights >50% of tumor cells. (f) S100 protein shows a similar staining pattern to GAP43. (g) Malignant peripheral nerve sheath tumor case no. 7, hematoxylin and eosin. (h) GAP43 staining strongly highlights >50% of tumor cells. (i) S100 protein is negative in malignant peripheral nerve sheath tumor case no. 7. (a–i) Magnification, × 200.
Anti-S100 protein antibody was also applied to all cases of malignant peripheral nerve sheath tumor using consecutive whole tissue sections for direct comparison (Tables 2 and 3). S100 protein was positive in 62% of malignant peripheral nerve sheath tumors (n=13/21) (Figures 1c, f and i). Of the eight cases that were negative for S100 protein staining, GAP43 was moderately to strongly positive in the majority of these cases (n=6/8; 75%), highlighting at least 5% or greater of tumor cells (Figures 1g–i and Table 3). Overall, GAP43 was highly sensitive for malignant peripheral nerve sheath tumor and showed a superior sensitivity to that of S100 protein.
In addition to the malignant peripheral nerve sheath tumors, GAP43 expression was also examined in neurofibromas, plexiform neurofibromas and schwannomas; it was moderately to strongly positive in nearly all neurofibromas/plexiform neurofibromas (n=17/18; 94%; 3+) (Figures 2a and b) and schwannomas (n=11/12; 92%; 3+) (Figures 2c and d) (Table 2).
GAP43 Staining in Tumors of Melanocytic Lineage
GAP43 immunoreactivity in tumors of melanocytic lineage is summarized in Table 2. A total of 22 non-desmoplastic, spindle cell melanomas were examined; GAP43 was negative in 20/22 cases (91%) (Figures 3a and b). In contrast, GAP43 was moderately to strongly positive in the desmoplastic component of pure and mixed variant desmoplastic melanomas (n=7/10; 70%; 3+) (Figures 3c and d).
Spindle cell and desmoplastic melanoma. (a) Spindle cell melanoma, hematoxylin and eosin. (b) Growth-associated protein 43 (GAP43) is negative in spindle cell melanoma. (c) Desmoplastic melanoma, hematoxylin and eosin. (d) GAP43 is strongly positive in desmoplastic melanoma. (a–d) Magnification, × 200.
GAP43 Staining in Spindle Cell Sarcomas of Diverse Types
GAP43 immunoreactivity was also examined in leiomyosarcomas, monophasic synovial sarcomas and clear-cell sarcomas, and the results are summarized in Table 2. Of the seven cases of monophasic synovial sarcoma, four were negative for GAP43, whereas three showed faint, very focal (∼5% of tumor cells), inconclusive immunoreactivity (n=3/7; 43%; 1+) (Figures 4a and b). GAP43 was negative in 14/16 cases of leiomyosarcoma (88%) (Figures 4c and d) and positive in 2/16 cases. Of the eight cases of clear-cell sarcoma (also known as melanoma of soft parts) examined, seven showed no GAP43 staining (n=7/8; 88%) (Figures 4e and f), whereas one case showed only limited focal positivity in tumor cells (<5% of tumor cells) and was scored as negative.
Growth-associated protein 43 (GAP43) staining in spindle cell sarcomas of diverse types. (a) Monophasic synovial sarcoma, hematoxylin and eosin. (b) This case showed focal, inconclusive immunoreactivity for GAP43. (c) Leiomyosarcoma, hematoxylin and eosin. (d) GAP43 is negative in leiomyosarcoma. (e) Clear-cell sarcoma. (f) GAP43 is negative in clear-cell sarcoma. (a–f) Magnification, × 200.
GAP43 Staining in Non-Neoplastic Tissues
A total of 15 normal tissue types or organs were tested for GAP43 expression, including the peripheral nerves, skin, skeletal muscle, adipose tissue, salivary gland, pharyngeal tonsil, thyroid gland, adrenal gland, lung, small and large intestines, pancreas, liver, uterine cervix and testis. In the skin, GAP43 staining was positive in the nerve twigs that innervate the skin, but negative in the melanocytes or other epithelial structures. In the pharyngeal tonsil, moderate GAP43 expression was observed in scattered dendritic cells. Other than these, GAP43 was only expressed in the peripheral nerves and nerve twigs that innervated the various tissue types above, but not in the epithelial or mesenchymal structures.
Discussion
This study reports our experience with the immunohistochemical expression of GAP43 or Neuromodulin, an intracellular growth-associated protein, which has a critical role in guiding axonal growth through the transduction of intra- and extracellular signals to regulate the cytoskeletal organization at the ends of neurites.10, 11 GAP43 is a protein key to the normal central nervous system development through its function of assisting neuronal path finding and branching. Initially, it was believed that the function of GAP43 protein was restricted to neurons; however, its expression has been demonstrated in Schwann cell precursors during development and in immature non-myelin-forming Schwann cells, which have lost axonal contact after denervation. Other than neurons and Schwann cells, GAP43 was reportedly expressed in some Merkel cells and Meissner corpuscles of the skin,12 but is not known to be expressed in other tissue and cellular types, including the melanocytes.
The utility of GAP43 has been explored to a very limited degree in the context of diagnostic surgical pathology. An earlier study by Huttenbach et al13 examined 12 cases of desmoplastic/spindle cell melanomas for GAP43 expression, and all 12 cases were non-reactive. As GAP43 is expressed in both axons and Schwannian precursors but not in melanocytes, we hypothesized that GAP43 may be a useful phenotypic marker in the context of peripheral nerve sheath tumors, including the malignant peripheral nerve sheath tumors. Is there a role for GAP43 in the diagnostic evaluation of a malignant spindle cell neoplasm whose differential diagnosis includes malignant peripheral nerve sheath tumor as well as other non-neural spindle cell sarcomas and desmoplastic and spindle cell melanomas?
As GAP43 is strongly expressed in peripheral nerves, we first turned our attention to its utility in malignant peripheral nerve sheath tumor, neurofibroma and schwannoma using whole tissue sections. Our results showed that GAP43 is a consistently expressed marker in peripheral nerve sheath neoplasms, with a sensitivity of 86%, 94% and 92% for the malignant peripheral nerve sheath tumors, neurofibromas/plexiform neurofibromas and schwannomas, respectively. Not only was GAP43 expressed in 86% of malignant peripheral nerve sheath tumors examined, it would appear to be a superior marker to S100 protein, which demonstrated a sensitivity of 62%, similar to the published experience.7, 14 Among the eight malignant peripheral nerve sheath tumors that were non-reactive for S100 protein, six (75%) showed strong GAP43 expression. Of the 21 malignant peripheral nerve sheath tumors examined, 20 represented resection specimens, and 1 case was a biopsy specimen. The biopsy specimen, despite its limited sample size, showed strong, diffuse positivity for GAP43. We, therefore, propose that this marker may have a role, together with the other more established immunohistochemical markers, such as S100 protein and CD57, in the diagnostic evaluation in the case of a suspected malignant peripheral nerve sheath tumor. Neurofibromas/plexiform neurofibromas and schwannomas were consistently immunoreactive for GAP43.
Another aspect of our study was to evaluate the expression of GAP43 in three non-neural soft tissue sarcomas, leiomyosarcoma, monophasic synovial sarcoma and clear-cell sarcoma, which in one circumstance or another may be considered in the initial differential diagnosis after the microscopic examination. Three monophasic synovial sarcomas showed limited immunoreactivity, and one leiomyosarcoma displayed intense, diffuse positivity. The latter result should come as no surprise to the experienced pathologist as we have progressed from one ‘specific’ immunohistochemical marker after another over the past 20–30 years. In the case of monophasic synovial sarcoma, other markers in conjunction with GAP43 and FISH would settle any lingering issues about the diagnosis of synovial sarcoma.
The diagnostic pitfall of the spindle cell melanoma and malignant peripheral nerve sheath tumor is well documented in the literature as either case reports or a case series as in the study of 16 cases by King and co-workers.1 In fact, Cruz et al15 reported a case of ‘malignant peripheral nerve sheath tumor-like’ primary cutaneous melanoma. The histopathologic features alone are clearly insufficient to differentiate between some spindle cell melanomas and malignant peripheral nerve sheath tumors. There is one report of a putative malignant peripheral nerve sheath tumor of the cervix, which was immunoreactive for S100 protein and HMB-45, but the tumor cells had schwannian differentiation and no melanosomes by electron microscopy.16 On the other hand, a number of the so-called melanocyte markers are not consistently expressed in spindle cell melanomas, including HMB-45, microphthalmia transcription factor and melan-A.17 Only approximately 20% of spindle cell/desmoplastic melanomas are immunoreactive for the latter marker.17
In this study, we have shown that GAP43 is clearly non-reactive in the majority of spindle cell melanomas (20/22; 91%). GAP43 was also applied to 10 cases of desmoplastic melanomas. Contrary to the result reported by Huttenbach et al,13 we observed strong expression of GAP43 in seven cases (70%) of desmoplastic melanomas; this may be attributed to the different clones of GAP43 antibody used between the two studies. Unfortunately, the antibody used in the previous report is no longer commercially available for comparison. The GAP43 antibody was clearly expressed in desmoplastic melanomas in our study, in contrast to the non-desmoplastic spindle cell melanomas. We hypothesized that the expression of GAP43 in desmoplastic melanoma, a distinct subset of spindle melanomas known for their neurotropism, may be attributed to its adoption of a more ‘neurotized/Schwannian’ cell fate.
The pathologic diagnosis of malignant peripheral nerve sheath tumor relied in the past on electron microscopy and immunohistochemistry. Molecular genetic and cytogenetic studies have had their limitations with the identification of complex, often random karyotypes. Thirty years ago, Weiss et al14 reported the value of S100 protein expression in benign nerve sheath tumors in almost 100% of cases, but in only 50% of malignant peripheral nerve sheath tumors or ‘malignant schwannomas.’ However, even in positive cases, the staining pattern may be only weak and focal. Given the acknowledged limitations of S100 protein sensitivity in suspected malignant peripheral nerve sheath tumors, other immunohistochemical markers, including CD57 (Leu7), collagen type IV (basement membrane staining), SOX10, nestin and glial fibrillary acidic protein, have been reported as more sensitive and/or specific than S100 protein.6, 7, 8, 18, 19, 20
We have shown in this study that GAP43 is expressed to a greater or lesser degree in the entire set of malignant peripheral nerve sheath tumors. To avoid any initial concerns about the diagnosis of malignant peripheral nerve sheath tumor, we chose tumors that occurred in a pre-existing neurofibroma or from individuals with neurofibromatosis-1. The only non-neural sarcoma with strong, diffuse GAP43 positivity was a cutaneous leiomyosarcoma. Swanson et al21 previously reported S100 protein and Leu7 positivity in cutaneous leiomyosarcoma to suggest a different origin than deep leiomyosarcomas. Lastly, in this study, we demonstrated that GAP43 can potentially differentiate malignant peripheral nerve sheath tumor from spindle cell melanoma, the quintessential mimicker of malignant peripheral nerve sheath tumor.
Before recommending that GAP43 should be added to the immunohistochemical panel in the diagnostic work-up of a suspected malignant peripheral nerve sheath tumor, our plan is to evaluate a group of soft tissue sarcomas in the non-neurofibromatosis-1 or sporadic setting to determine the efficacy and value of GAP43 over the entire range of malignant peripheral nerve sheath tumors.
References
King R, Busam K, Rosai J . Metastatic malignant melanoma resembling malignant peripheral nerve sheath tumor: report of 16 cases. Am J Surg Pathol 1999;23:1499–1505.
Ferner RE, O’Doherty MJ . Neurofibroma and schwannoma. Curr Opin Neurol 2002;15:679–684.
Coindre JM, Hostein I, Benhattar J et al. Malignant peripheral nerve sheath tumors are t(X;18)-negative sarcomas. Molecular analysis of 25 cases occurring in neurofibromatosis type 1 patients, using two different RT-PCR-based methods of detection. Mod Pathol 2002;15:589–592.
Christensen WN, Strong EW, Bains MS et al. Neuroendocrine differentiation in the glandular peripheral nerve sheath tumor. Pathologic distinction from the biphasic synovial sarcoma with glands. Am J Surg Pathol 1988;12:417–426.
Rodriguez FJ, Folpe AL, Giannini C et al. Pathology of peripheral nerve sheath tumors: diagnostic overview and update on selected diagnostic problems. Acta Neuropathol 2012;123:295–319.
Nonaka D, Chiriboga L, Rubin BP . Sox10: a pan-schwannian and melanocytic marker. Am J Surg Pathol 2008;32:1291–1298.
Wick MR, Swanson PE, Scheithauer BW et al. Malignant peripheral nerve sheath tumor. An immunohistochemical study of 62 cases. Am J Clin Pathol 1987;87:425–433.
Scheithauer BW, Erdogan S, Rodriguez FJ et al. Malignant peripheral nerve sheath tumors of cranial nerves and intracranial contents: a clinicopathologic study of 17 cases. Am J Surg Pathol 2009;33:325–338.
Miller DD, Emley A, Yang S et al. Mixed versus pure variants of desmoplastic melanoma: a genetic and immunohistochemical appraisal. Mod Pathol 2012;25:505–515.
Benowitz LI, Routtenberg A . GAP43: an intrinsic determinant of neuronal development and plasticity. Trends Neurosci 1997;20:84–91.
Denny JB . Molecular mechanisms, biological actions, and neuropharmacology of the growth-associated protein GAP43. Curr Neuropharmacol 2006;4:293–304.
Verze L, Viglietti-Panzica C, Maurizo S et al. Distribution of GAP43 nerve fibers in the skin of the adult human hand. Anat Rec A Discov Mol Cell Evol Biol 2003;272:467–473.
Huttenbach Y, Prieto VG, Reed JA . Desmoplastic and spindle cell melanomas express protein markers of the neural crest but not of later committed stages of Schwann cell differentiation. J Cutan Pathol 2002;29:562–568.
Weiss SW, Langloss JM, Enzinger FM . Value of S100 protein in the diagnosis of soft tissue tumors with particular reference to benign and malignant Schwann cell tumors. Lab Invest 1983;49:299–308.
Cruz J, Reis-Filho JS, Lopes JM . Malignant peripheral nerve sheath tumour-like primary cutaneous malignant melanoma. J Clin Pathol 2004;57:218–220.
Kim NR, Chung DH, Park CY et al. Malignant peripheral nerve sheath tumor of the uterine cervix expressing both S100 protein and HMB-45. J Obstet Gynaecol Res 2009;35:1136–1141.
Ohsie SJ, Sarantopoulos GP, Cochran AJ et al. Immunohistochemical characteristics of melanoma. J Cutan Pathol 2008;35:433–444.
Hirose T, Scheithauer BW, Sano T . Perineurial malignant peripheral nerve sheath tumor (MPNST): a clinicopathologic, immunohistochemical, and ultrastructural study of seven cases. Am J Surg Pathol 1998;22:1368–1378.
Shimada S, Tsuzuki T, Kuroda M et al. Nestin expression as a new marker in malignant peripheral nerve sheath tumors. Pathol Int 2007;57:60–67.
Giangaspero F, Fratamico FC, Ceccarelli C et al. Malignant peripheral nerve sheath tumors and spindle cell sarcomas: an immunohistochemical analysis of multiple markers. Appl Pathol 1989;7:134–144.
Swanson PE, Stanley MW, Scheithauer BW et al. Primary cutaneous leiomyosarcoma. A histological and immunohistochemical study of 9 cases, with ultrastructural correlation. J Cutan Pathol 1988;15:129–141.
Acknowledgements
We thank the Department of Pathology and Immunology, Washington University School of Medicine, for the funding of this project, Anatomic and Molecular Pathology Core Lab for expert immunohistochemical staining and Stacey Yates for imaging and technical support.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Chen, WS., Chen, PL., Lu, D. et al. Growth-associated protein 43 in differentiating peripheral nerve sheath tumors from other non-neural spindle cell neoplasms. Mod Pathol 27, 184–193 (2014). https://doi.org/10.1038/modpathol.2013.128
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.2013.128
Keywords
This article is cited by
-
What’s new in nerve sheath tumors
Virchows Archiv (2020)
-
GAP43, a novel metastasis promoter in non-small cell lung cancer
Journal of Translational Medicine (2018)
-
Identification, by systematic RNA sequencing, of novel candidate biomarkers and therapeutic targets in human soft tissue tumors
Laboratory Investigation (2015)
-
Tinospora cordifolia Induces Differentiation and Senescence Pathways in Neuroblastoma Cells
Molecular Neurobiology (2015)