Abstract
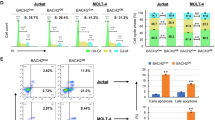
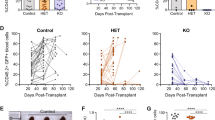
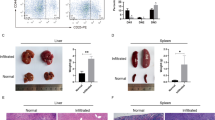
The initial stages of T-cell differentiation are characterized by a progressive commitment to the T-cell lineage, a process that involves the loss of alternative (myelo-erythroid, NK, B) lineage potentials. Aberrant differentiation during these stages can result in T-cell acute lymphoblastic leukemia (T-ALL). However, the mechanisms regulating the initial stages of human T-cell differentiation are obscure. Through loss of function studies, we showed BCL11B, a transcription factor recurrently mutated T-ALL, is essential for T-lineage commitment, particularly the repression of NK and myeloid potentials, and the induction of T-lineage genes, during the initial stages of human T-cell differentiation. In gain of function studies, BCL11B inhibited growth of and induced a T-lineage transcriptional program in T-ALL cells. We found previously unknown differentiation stage-specific DNA binding of BCL11B at multiple T-lineage genes; target genes showed BCL11B-dependent expression, suggesting a transcriptional activator role for BCL11B at these genes. Transcriptional analyses revealed differences in the regulatory actions of BCL11B between human and murine thymopoiesis. Our studies show BCL11B is a key regulator of the initial stages of human T-cell differentiation and delineate the BCL11B transcriptional program, enabling the dissection of the underpinnings of normal T-cell differentiation and providing a resource for understanding dysregulations in T-ALL.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Plum J, De Smedt M, Leclercq G, Taghon T, Kerre T, Vandekerckhove B . Human intrathymic development: a selective approach. Semin Immunopathol 2008; 30: 411–423.
Hao QL, George AA, Zhu J, Barsky L, Zielinska E, Wang X et al. Human intrathymic lineage commitment is marked by differential CD7 expression: identification of CD7− lympho-myeloid thymic progenitors. Blood 2008; 111: 1318–1326.
Weerkamp F, Baert MR, Brugman MH, Dik WA, de Haas EF, Visser TP et al. Human thymus contains multipotent progenitors with T/B lymphoid, myeloid, and erythroid lineage potential. Blood 2006; 107: 3131–3137.
Ferrando AA, Neuberg DS, Staunton J, Loh ML, Huard C, Raimondi SC et al. Gene expression signatures define novel oncogenic pathways in T cell acute lymphoblastic leukemia. Cancer Cell 2002; 1: 75–87.
Van Vlierberghe P, Ferrando A . The molecular basis of T cell acute lymphoblastic leukemia. J Clin Invest 2012; 122: 3398–3406.
Durinck K, Van Loocke W, Van der Meulen J, Van de Walle I, Ongenaert M, Rondou P et al. Characterization of the genome-wide TLX1 binding profile in T-cell acute lymphoblastic leukemia. Leukemia 2015; 29: 2317–2327.
Treanor LM, Zhou S, Janke L, Churchman ML, Ma Z, Lu T et al. Interleukin-7 receptor mutants initiate early T cell precursor leukemia in murine thymocyte progenitors with multipotent potential. J Exp Med 2014; 211: 701–713.
Wallaert A, Durinck K, Van Loocke W, Van de Walle I, Matthijssens F, Volders PJ et al. Long noncoding RNA signatures define oncogenic subtypes in T-cell acute lymphoblastic leukemia. Leukemia 2016; 30: 1927–1930.
Yui MA, Rothenberg EV . Developmental gene networks: a triathlon on the course to T cell identity. Nat Rev Immunol 2014; 14: 529–545.
Parekh C, Crooks GM . Critical differences in hematopoiesis and lymphoid development between humans and mice. J Clin Immunol 2013; 33: 711–715.
Van de Walle I, De Smet G, De Smedt M, Vandekerckhove B, Leclercq G, Plum J et al. An early decrease in Notch activation is required for human TCR-alphabeta lineage differentiation at the expense of TCR-gammadelta T cells. Blood 2009; 113: 2988–2998.
Van de Walle I, Dolens AC, Durinck K, De Mulder K, Van Loocke W, Damle S et al. GATA3 induces human T-cell commitment by restraining Notch activity and repressing NK-cell fate. Nat Commun 2016; 7: 11171.
Li L, Leid M, Rothenberg EV . An early T cell lineage commitment checkpoint dependent on the transcription factor Bcl11b. Science 2010; 329: 89–93.
Califano D, Cho JJ, Uddin MN, Lorentsen KJ, Yang Q, Bhandoola A et al. Transcription factor Bcl11b controls identity and function of mature type 2 innate lymphoid cells. Immunity 2015; 43: 354–368.
Albu DI, Feng D, Bhattacharya D, Jenkins NA, Copeland NG, Liu P et al. BCL11B is required for positive selection and survival of double-positive thymocytes. J Exp Med 2007; 204: 3003–3015.
Punwani D, Zhang Y, Yu J, Cowan MJ, Rana S, Kwan A et al. Multisystem anomalies in severe combined immunodeficiency with mutant BCL11B. N Engl J Med 2016; 375: 2165–2176.
De Keersmaecker K, Real PJ, Gatta GD, Palomero T, Sulis ML, Tosello V et al. The TLX1 oncogene drives aneuploidy in T cell transformation. Nat Med 2010; 16: 1321–1327.
Gutierrez A, Kentsis A, Sanda T, Holmfeldt L, Chen SC, Zhang J et al. The BCL11B tumor suppressor is mutated across the major molecular subtypes of T-cell acute lymphoblastic leukemia. Blood 2011; 118: 4169–4173.
Nagamachi A, Yamasaki N, Miyazaki K, Oda H, Miyazaki M, Honda Z et al. Haploinsufficiency and acquired loss of Bcl11b and H2AX induces blast crisis of chronic myelogenous leukemia in a transgenic mouse model. Cancer Sci 2009; 100: 1219–1226.
Coustan-Smith E, Mullighan CG, Onciu M, Behm FG, Raimondi SC, Pei D et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia. Lancet Oncol 2009; 10: 147–156.
Park E, Gang EJ, Hsieh YT, Schaefer P, Chae S, Klemm L et al. Targeting survivin overcomes drug resistance in acute lymphoblastic leukemia. Blood 2011; 118: 2191–2199.
La Motte-Mohs RN, Herer E, Zuniga-Pflucker JC . Induction of T-cell development from human cord blood hematopoietic stem cells by Delta-like 1 in vitro. Blood 2005; 105: 1431–1439.
Trapnell C, Pachter L, Salzberg SL . TopHat: discovering splice junctions with RNA-Seq. Bioinformatics 2009; 25: 1105–1111.
Anders S, Pyl PT, Huber W . HTSeq—a Python framework to work with high-throughput sequencing data. Bioinformatics 2015; 31: 166–169.
Anders S, Huber W . Differential expression analysis for sequence count data. Genome Biol 2010; 11: R106.
Subramanian ATP, Mootha VK, Mukherjee S, Ebert BL, Gillette MA, Paulovich A et al. Gene set enrichment analysis: a knowledge-based approach for interpreting genome-wide expression profiles. Proc Natl Acad Sci USA 2005; 102: 15545–15550.
Casero D, Sandoval S, Seet CS, Scholes J, Zhu Y, Ha VL et al. Long non-coding RNA profiling of human lymphoid progenitor cells reveals transcriptional divergence of B cell and T cell lineages. Nat Immunol 2015; 16: 1282–1291.
Langmead B, Trapnell C, Pop M, Salzberg SL . Ultrafast and memory-efficient alignment of short DNA sequences to the human genome. Genome Biol 2009; 10: R25.
Zhang Y, Liu T, Meyer CA, Eeckhoute J, Johnson DS, Bernstein BE et al. Model-based analysis of ChIP-Seq (MACS). Genome Biol 2008; 9: R137.
Machanick P, Bailey TL . MEME-ChIP: motif analysis of large DNA datasets. Bioinformatics 2011; 27: 1696–1697.
Ma W, Noble WS, Bailey TL . Motif-based analysis of large nucleotide data sets using MEME-ChIP. Nat Protoc 2014; 9: 1428–1450.
Ross-Innes CS, Stark R, Teschendorff AE, Holmes KA, Ali HR, Dunning MJ et al. Differential oestrogen receptor binding is associated with clinical outcome in breast cancer. Nature 2012; 481: 389–393.
McLean CY, Bristor D, Hiller M, Clarke SL, Schaar BT, Lowe CB et al. GREAT improves functional interpretation of cis-regulatory regions. Nat Biotechnol 2010; 28: 495–501.
Zhang JA, Mortazavi A, Williams BA, Wold BJ, Rothenberg EV . Dynamic transformations of genome-wide epigenetic marking and transcriptional control establish T cell identity. Cell 2012; 149: 467–482.
Lanier LL, Allison JP, Phillips JH . Correlation of cell surface antigen expression on human thymocytes by multi-color flow cytometric analysis: implications for differentiation. J Immunol 1986; 137: 2501–2507.
Awong G, Herer E, Surh CD, Dick JE, La Motte-Mohs RN, Zuniga-Pflucker JC . Characterization in vitro and engraftment potential in vivo of human progenitor T cells generated from hematopoietic stem cells. Blood 2009; 114: 972–982.
Li W, Okuda A, Yamamoto H, Yamanishi K, Terada N, Yamanishi H et al. Regulation of development of CD56 bright CD11c+NK-like cells with helper function by IL-18. PLoS One 2013; 8: e82586.
Burger R, Hansen-Hagge TE, Drexler HG, Gramatzki M . Heterogeneity of T-acute lymphoblastic leukemia (T-ALL) cell lines: suggestion for classification by immunophenotype and T-cell receptor studies. Leuk Res 1999; 23: 19–27.
Petitjean A, Mathe E, Kato S, Ishioka C, Tavtigian SV, Hainaut P et al. Impact of mutant p53 functional properties on TP53 mutation patterns and tumor phenotype: lessons from recent developments in the IARC TP53 database. Hum Mutat 2007; 28: 622–629.
Eidson M, Wahlstrom J, Beaulieu AM, Zaidi B, Carsons SE, Crow PK et al. Altered development of NKT cells, gammadelta T cells, CD8 T cells and NK cells in a PLZF deficient patient. PLoS One 2011; 6: e24441.
Avram D, Fields A, Senawong T, Topark-Ngarm A, Leid M . COUP-TF (chicken ovalbumin upstream promoter transcription factor)-interacting protein 1 (CTIP1) is a sequence-specific DNA binding protein. Biochem J 2002; 368: 555–563.
Zhang LJ, Vogel WK, Liu X, Topark-Ngarm A, Arbogast BL, Maier CS et al. Coordinated regulation of transcription factor Bcl11b activity in thymocytes by the mitogen-activated protein kinase (MAPK) pathways and protein sumoylation. J Biol Chem 2012; 287: 26971–26988.
Topark-Ngarm A, Golonzhka O, Peterson VJ, Barrett Jr B, Martinez B, Crofoot K et al. CTIP2 associates with the NuRD complex on the promoter of p57KIP2, a newly identified CTIP2 target gene. J Biol Chem 2006; 281: 32272–32283.
Ikawa T, Hirose S, Masuda K, Kakugawa K, Satoh R, Shibano-Satoh A et al. An essential developmental checkpoint for production of the T cell lineage. Science 2010; 329: 93–96.
Rothenberg EV, Zhang J, Li L . Multilayered specification of the T-cell lineage fate. Immunol Rev 2010; 238: 150–168.
Cismasiu VB, Ghanta S, Duque J, Albu DI, Chen HM, Kasturi R et al. BCL11B participates in the activation of IL2 gene expression in CD4+ T lymphocytes. Blood 2006; 108: 2695–2702.
Cismasiu VB, Adamo K, Gecewicz J, Duque J, Lin Q, Avram D . BCL11B functionally associates with the NuRD complex in T lymphocytes to repress targeted promoter. Oncogene 2005; 24: 6753–6764.
Acknowledgements
We thank A George and Jonathan Kirzner for technical assistance; Dr Sara Twogood and Dr Wittenberg (Hollywood Presbyterian Hospital) for collecting CB; the Cardiothoracic Surgery Department at the Children’s Hospital Los Angeles (CHLA) for collecting thymuses; the CHLA Next generation sequencing core; CHLA and the Broad Stem Cell Research Center flow cytometry facilities; the Center for AIDS Research Virology Core Lab (supported by the US National Institutes of Health (AI-28697), the UCLA AIDS Institute and the UCLA Council of Bioscience Resources) for CB and reagents; Dr D Kohn for the MND-PGK-GFP plasmid; Dr D Trono for psPAX2, pMD2.G and pLVTHM plasmids; Dr D Sabatini for the scramble shRNA plasmid; and Dr Zuniga-Pflucker for the OP9-DLL1 cell line. This work was supported by the St Baldrick’s Foundation (CP), the US National Institutes of Health (K12 HD052954, K12 HD034610 and P30CA014089 to CP; P01 HL073104 to GMC; R01 CA172896 to YMK), the Nautica Foundation (CP), the Tower Cancer Research Foundation (CP), the Couples Against Leukemia Foundation (CP), the Joseph Drown Foundation (CP) and the UCLA Broad Stem Cell Research Center (GMC and DC).
Author contributions
VLH: collection and assembly of data (performance of experiments), data analysis and interpretation; AL: collection and assembly of data (performance of experiments), data analysis and interpretation; FL: bioinformatic analysis of RNA-Seq and ChIP-Seq data; DC: bioinformatic analysis of RNA-Seq data from normal bone marrow and thymic progenitors and assembly of annotated lncRNA database; JM: statistical analysis; YMK: generation of T-ALL xenograft; RB: conception and design, data analysis and interpretation; GMC: conception and design, data analysis and interpretation, manuscript writing; CP: conception and design, collection and assembly of data (performance of experiments), data analysis and interpretation, manuscript writing and final approval of manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Ha, V., Luong, A., Li, F. et al. The T-ALL related gene BCL11B regulates the initial stages of human T-cell differentiation. Leukemia 31, 2503–2514 (2017). https://doi.org/10.1038/leu.2017.70
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2017.70
This article is cited by
-
γδ T cells: origin and fate, subsets, diseases and immunotherapy
Signal Transduction and Targeted Therapy (2023)
-
Multi-objective optimization reveals time- and dose-dependent inflammatory cytokine-mediated regulation of human stem cell derived T-cell development
npj Regenerative Medicine (2022)
-
Epigenome-wide analysis of T-cell large granular lymphocytic leukemia identifies BCL11B as a potential biomarker
Clinical Epigenetics (2022)
-
Human induced-T-to-natural killer cells have potent anti-tumour activities
Biomarker Research (2022)
-
Inhibition of BCL11B induces downregulation of PTK7 and results in growth retardation and apoptosis in T-cell acute lymphoblastic leukemia
Biomarker Research (2021)