Abstract
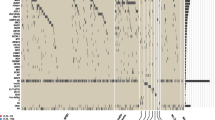
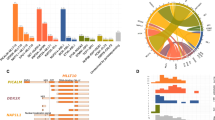
Constitutively activated FLT3 signaling is common in acute myeloid leukemia, and is currently under evaluation for targeted therapy, whereas little data is available in T-cell acute lymphoblastic leukemia (T-ALL). We analyzed 357 T-ALL cases for FLT3 mutations and transcript expression. FLT3 mutations (3% overall) and overexpression (FLT3 high expresser (FLT3High)) were restricted to immature/TCRγδ T-ALLs. In vitro FLT3 inhibition induced apoptosis in only 30% of FLT3High T-ALLs and did not correlate with mutational status. In order to investigate the mechanisms of primary resistance to FLT3 inhibition, a broad quantitative screen for receptor kinome transcript deregulation was performed by Taqman Low Density Array. FLT3 deregulation was associated with overexpression of a network of receptor kinases (RKs), potentially responsible for redundancies and sporadic response to specific FLT3 inhibition. In keeping with this resistance to FLT3 inhibition could be reversed by dual inhibition of FLT3 and KIT with a synergistic effect. We conclude that immature T-ALL may benefit from multitargeted RK inhibition and that exploration of the receptor kinome defines a rational strategy for testing multitarget kinase inhibition in malignant diseases.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Look AT . Molecular pathways in T-cell acute lymphoblastic leukemia: ramifications for therapy. Clin Adv Hematol Oncol 2004; 2: 779–780.
Asnafi V, Beldjord K, Boulanger E, Comba B, Le Tutour P, Estienne MH et al. Analysis of TCR, pT alpha, and RAG-1 in T-acute lymphoblastic leukemias improves understanding of early human T-lymphoid lineage commitment. Blood 2003; 101: 2693–2703.
Asnafi V, Radford-Weiss I, Dastugue N, Bayle C, Leboeuf D, Charrin C et al. CALM-AF10 is a common fusion transcript in T-ALL and is specific to the TCRgammadelta lineage. Blood 2003; 102: 1000–1006.
Ferrando AA, Neuberg DS, Staunton J, Loh ML, Huard C, Raimondi SC et al. Gene expression signatures define novel oncogenic pathways in T cell acute lymphoblastic leukemia. Cancer Cell 2002; 1: 75–87.
Soulier J, Clappier E, Cayuela JM, Regnault A, Garcia-Peydro M, Dombret H et al. HOXA genes are included in genetic and biologic networks defining human acute T-cell leukemia (T-ALL). Blood 2005; 106: 274–286.
Rubnitz JE, Onciu M, Pounds S, Shurtleff S, Cao X, Raimondi SC et al. Acute mixed lineage leukemia in children: the experience of St Jude Children’s Research Hospital. Blood 2009; 113: 5083–5089.
Rubio MT, Dhedin N, Boucheix C, Bourhis JH, Reman O, Boiron JM et al. Adult T-biphenotypic acute leukaemia: clinical and biological features and outcome. Br J Haematol 2003; 123: 842–849.
Coustan-Smith E, Mullighan CG, Onciu M, Behm FG, Raimondi SC, Pei D et al. Early T-cell precursor leukaemia: a subtype of very high-risk acute lymphoblastic leukaemia. Lancet Oncol 2009; 10: 147–14 56.
Asnafi V, Buzyn A, Thomas X, Huguet F, Vey N, Boiron JM et al. Impact of TCR status and genotype on outcome in adult T acute lymphoblastic leukemia: a LALA-94 study. Blood 2005; 105: 3072–3078.
Brown P, Meshinchi S, Levis M, Alonzo TA, Gerbing R, Lange B et al. Pediatric AML primary samples with FLT3/ITD mutations are preferentially killed by FLT3 inhibition. Blood 2004; 104: 1841–1849.
Taketani T, Taki T, Sugita K, Furuichi Y, Ishii E, Hanada R et al. FLT3 mutations in the activation loop of tyrosine kinase domain are frequently found in infant ALL with MLL rearrangements and pediatric ALL with hyperdiploidy. Blood 2004; 103: 1085–1088.
Paietta E, Ferrando AA, Neuberg D, Bennett JM, Racevskis J, Lazarus H et al. Activating FLT3 mutations in CD117/KIT(+) T-cell acute lymphoblastic leukemias. Blood 2004; 104: 558–560.
Van Vlierberghe P, Meijerink JP, Stam RW, van der Smissen W, van Wering ER, Beverloo HB et al. Activating FLT3 mutations in CD4+/CD8− pediatric T-cell acute lymphoblastic leukemias. Blood 2005; 106: 4414–4415.
Scharnhorst V, Wals J, Beverloo HB, Langerak AW, van der Velden VH . Mutation of FLT3 is not a general phenomenon in CD117-positive T-ALL. Leuk Res 2006; 30: 245–246.
Birg F, Courcoul M, Rosnet O, Bardin F, Pebusque MJ, Marchetto S et al. Expression of the FMS/KIT-like gene FLT3 in human acute leukemias of the myeloid and lymphoid lineages. Blood 1992; 80: 2584–2593.
Carow CE, Levenstein M, Kaufmann SH, Chen J, Amin S, Rockwell P et al. Expression of the hematopoietic growth factor receptor FLT3 (STK-1/Flk2) in human leukemias. Blood 1996; 87: 1089–1096.
Rosnet O, Buhring HJ, Marchetto S, Rappold I, Lavagna C, Sainty D et al. Human FLT3/FLK2 receptor tyrosine kinase is expressed at the surface of normal and malignant hematopoietic cells. Leukemia 1996; 10: 238–248.
Ozeki K, Kiyoi H, Hirose Y, Iwai M, Ninomiya M, Kodera Y et al. Biologic and clinical significance of the FLT3 transcript level in acute myeloid leukemia. Blood 2004; 103: 1901–1908.
Swerdlow S, Campo E, Harris N, Jaffe E, Pileri S, Stein H et al. WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. 4th edn. IARC Press: Lyon, 2008.
Libura M, Asnafi V, Tu A, Delabesse E, Tigaud I, Cymbalista F et al. FLT3 and MLL intragenic abnormalities in AML reflect a common category of genotoxic stress. Blood 2003; 102: 2198–2204.
Eisen MB, Spellman PT, Brown PO, Botstein D . Cluster analysis and display of genome-wide expression patterns. Proc Natl Acad Sci USA 1998; 95: 14863–14868.
Chou TC . Theoretical basis, experimental design, and computerized simulation of synergism and antagonism in drug combination studies. Pharmacol Rev 2006; 58: 621–681.
Chou TC, Talalay P . Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul 1984; 22: 27–55.
Tse KF, Novelli E, Civin CI, Bohmer FD, Small D . Inhibition of FLT3-mediated transformation by use of a tyrosine kinase inhibitor. Leukemia 2001; 15: 1001–1010.
Georgin-Lavialle S, Lhermitte L, Suarez F, Yang Y, Letard S, Hanssens K et al. Mast cell leukemia: identification of a new c-Kit mutation, dup(501–502), and response to masitinib, a c-Kit tyrosine kinase inhibitor. Eur J Haematol 2012; 89: 47–52.
Kotecha N, Krutzik PO, Irish JM . Web-based analysis and publication of flow cytometry experiments. Curr Protoc Cytom, Chapter 10: Unit10 17.
Ferrando AA, Armstrong SA, Neuberg DS, Sallan SE, Silverman LB, Korsmeyer SJ et al. Gene expression signatures in MLL-rearranged T-lineage and B-precursor acute leukemias: dominance of HOX dysregulation. Blood 2003; 102: 262–268.
Asnafi V, Beldjord K, Libura M, Villarese P, Millien C, Ballerini P et al. Age-related phenotypic and oncogenic differences in T-cell acute lymphoblastic leukemias may reflect thymic atrophy. Blood 2004; 104: 4173–4180.
Macintyre EA, Smit L, Ritz J, Kirsch IR, Strominger JL . Disruption of the SCL locus in T-lymphoid malignancies correlates with commitment to the T-cell receptor alpha beta lineage. Blood 1992; 80: 1511–1520.
Hayakawa F, Towatari M, Kiyoi H, Tanimoto M, Kitamura T, Saito H et al. Tandem-duplicated Flt3 constitutively activates STAT5 and MAP kinase and introduces autonomous cell growth in IL-3-dependent cell lines. Oncogene 2000; 19: 624–631.
Masson K, Ronnstrand L . Oncogenic signaling from the hematopoietic growth factor receptors c-Kit and Flt3. Cell Signal 2009; 21: 1717–1726.
Mizuki M, Fenski R, Halfter H, Matsumura I, Schmidt R, Muller C et al. Flt3 mutations from patients with acute myeloid leukemia induce transformation of 32D cells mediated by the Ras and STAT5 pathways. Blood 2000; 96: 3907–3914.
Kim KT, Baird K, Ahn JY, Meltzer P, Lilly M, Levis M et al. Pim-1 is upregulated by constitutively activated FLT3 and plays a role in FLT3-mediated cell survival. Blood 2005; 105: 1759–1767.
Kim KT, Levis M, Small D . Constitutively activated FLT3 phosphorylates BAD partially through pim-1. Br J Haematol 2006; 134: 500–509.
Vu HA, Xinh PT, Kano Y, Tokunaga K, Sato Y . The juxtamembrane domain in ETV6/FLT3 is critical for PIM-1 up-regulation and cell proliferation. Biochem Biophys Res Commun 2009; 383: 308–313.
Pratz KW, Sato T, Murphy KM, Stine A, Rajkhowa T, Levis M . FLT3-mutant allelic burden and clinical status are predictive of response to FLT3 inhibitors in AML. Blood 2010; 115: 1425–1432.
Zarrinkar PP, Gunawardane RN, Cramer MD, Gardner MF, Brigham D, Belli B et al. AC220 is a uniquely potent and selective inhibitor of FLT3 for the treatment of acute myeloid leukemia (AML). Blood 2009; 114: 2984–2992.
Manning G, Whyte DB, Martinez R, Hunter T, Sudarsanam S . The protein kinase complement of the human genome. Science 2002; 298: 1912–1934.
Dubreuil P, Letard S, Ciufolini M, Gros L, Humbert M, Casteran N et al. Masitinib (AB1010), a potent and selective tyrosine kinase inhibitor targeting KIT. PLoS One 2009; 4: e7258.
Levis M, Allebach J, Tse KF, Zheng R, Baldwin BR, Smith BD et al. A FLT3-targeted tyrosine kinase inhibitor is cytotoxic to leukemia cells in vitro and in vivo. Blood 2002; 99: 3885–3891.
Stone RM, DeAngelo DJ, Klimek V, Galinsky I, Estey E, Nimer SD et al. Patients with acute myeloid leukemia and an activating mutation in FLT3 respond to a small-molecule FLT3 tyrosine kinase inhibitor, PKC412. Blood 2005; 105: 54–60.
Bagrintseva K, Schwab R, Kohl TM, Schnittger S, Eichenlaub S, Ellwart JW et al. Mutations in the tyrosine kinase domain of FLT3 define a new molecular mechanism of acquired drug resistance to PTK inhibitors in FLT3-ITD-transformed hematopoietic cells. Blood 2004; 103: 2266–2275.
Clark JJ, Cools J, Curley DP, Yu JC, Lokker NA, Giese NA et al. Variable sensitivity of FLT3 activation loop mutations to the small molecule tyrosine kinase inhibitor MLN518. Blood 2004; 104: 2867–2872.
Grundler R, Thiede C, Miething C, Steudel C, Peschel C, Duyster J . Sensitivity toward tyrosine kinase inhibitors varies between different activating mutations of the FLT3 receptor. Blood 2003; 102: 646–651.
Breitenbuecher F, Markova B, Kasper S, Carius B, Stauder T, Bohmer FD et al. A novel molecular mechanism of primary resistance to FLT3-kinase inhibitors in AML. Blood 2009; 113: 4063–4073.
Levis M, Ravandi F, Wang ES, Baer MR, Perl A, Coutre S et al. Results from a randomized trial of salvage chemotherapy followed by lestaurtinib for patients with FLT3 mutant AML in first relapse. Blood 2009; 117: 3294–3301.
Sato T, Yang X, Knapper S, White P, Smith BD, Galkin S et al. FLT3 ligand impedes the efficacy of FLT3 inhibitors in vitro and in vivo. Blood 2011; 117: 3286–3293.
Knapper S, Mills KI, Gilkes AF, Austin SJ, Walsh V, Burnett AK . The effects of lestaurtinib (CEP701) and PKC412 on primary AML blasts: the induction of cytotoxicity varies with dependence on FLT3 signaling in both FLT3-mutated and wild-type cases. Blood 2006; 108: 3494–3503.
Chalandon Y, Schwaller J . Targeting mutated protein tyrosine kinases and their signaling pathways in hematologic malignancies. Haematologica 2005; 90: 949–968.
Jiang BH, Liu LZ . Role of mTOR in anticancer drug resistance: perspectives for improved drug treatment. Drug Resist Updat 2008; 11: 63–76.
Mohi MG, Boulton C, Gu TL, Sternberg DW, Neuberg D, Griffin JD et al. Combination of rapamycin and protein tyrosine kinase (PTK) inhibitors for the treatment of leukemias caused by oncogenic PTKs. Proc Natl Acad Sci USA 2004; 101: 3130–3135.
Stommel JM, Kimmelman AC, Ying H, Nabioullin R, Ponugoti AH, Wiedemeyer R et al. Coactivation of receptor tyrosine kinases affects the response of tumor cells to targeted therapies. Science 2007; 318: 287–290.
Linger RM, DeRyckere D, Brandao L, Sawczyn KK, Jacobsen KM, Liang X et al. Mer receptor tyrosine kinase is a novel therapeutic target in pediatric B-cell acute lymphoblastic leukemia. Blood 2009; 114: 2678–2687.
Acknowledgements
We thank all the biologists and clinicians of the LALA/GRAALL adult and FRALLE pediatric French ALL groups for providing T-ALL samples and results. This work was supported by grants from the Association Cent Pour Sang La Vie and the association Laurette Fugain. We thank the Necker-Enfants-Malades department of pediatric cardiac surgery for providing thymic material.
LL and RBA performed research, data analysis and wrote the paper. PV, ML, NB and ASB performed research. VG performed statistical data analysis. HD and OH performed data analysis. EM and VA oversaw conceptual development of the project and wrote the paper.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Author contributions
LL and RBA performed research, data analysis and wrote the paper. PV, ML, NB and ASB performed research. VG performed statistical data analysis. HD and OH performed data analysis. EM and VA oversaw conceptual development of the project and wrote the paper.
Supplementary Information accompanies the paper on the Leukemia website
Supplementary information
Rights and permissions
About this article
Cite this article
Lhermitte, L., Ben Abdelali, R., Villarèse, P. et al. Receptor kinase profiles identify a rationale for multitarget kinase inhibition in immature T-ALL. Leukemia 27, 305–314 (2013). https://doi.org/10.1038/leu.2012.177
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2012.177
Keywords
This article is cited by
-
Hhex regulates Kit to promote radioresistance of self-renewing thymocytes in Lmo2-transgenic mice
Leukemia (2015)
-
Cytotoxic activity of the casein kinase 2 inhibitor CX-4945 against T-cell acute lymphoblastic leukemia: targeting the unfolded protein response signaling
Leukemia (2014)
-
Activity of the pan-class I phosphoinositide 3-kinase inhibitor NVP-BKM120 in T-cell acute lymphoblastic leukemia
Leukemia (2014)