Abstract
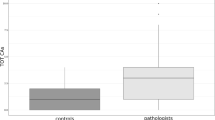
Benzene exposure causes acute myeloid leukemia and hematotoxicity, shown as suppression of mature blood and myeloid progenitor cell numbers. As the leukemia-related aneuploidies monosomy 7 and trisomy 8 previously had been detected in the mature peripheral blood cells of exposed workers, we hypothesized that benzene could cause leukemia through the induction of these aneuploidies in hematopoietic stem and progenitor cells. We measured loss and gain of chromosomes 7 and 8 by fluorescence in situ hybridization in interphase colony-forming unit–granulocyte-macrophage (CFU-GM) cells cultured from otherwise healthy benzene-exposed (n=28) and unexposed (n=14) workers. CFU-GM monosomy 7 and 8 levels (but not trisomy) were significantly increased in subjects exposed to benzene overall, compared with levels in the control subjects (P=0.0055 and P=0.0034, respectively). Levels of monosomy 7 and 8 were significantly increased in subjects exposed to <10 p.p.m. (20%, P=0.0419 and 28%, P=0.0056, respectively) and ⩾10 p.p.m. (48%, P=0.0045 and 32%, 0.0354) benzene, compared with controls, and significant exposure–response trends were detected (Ptrend=0.0033 and 0.0057). These data show that monosomies 7 and 8 are produced in a dose-dependent manner in the blood progenitor cells of workers exposed to benzene, and may be mechanistically relevant biomarkers of early effect for benzene and other leukemogens.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Johnson ES, Langard S, Lin YS . A critique of benzene exposure in the general population. Sci Total Environ 2007; 374: 183–198.
Smith MT . Advances in understanding benzene health effects and susceptibility. Annu Rev Public Health 2010; 31: 133–148.
Aksoy M . Hematotoxicity and carcinogenicity of benzene. Environ Health Perspect 1989; 82: 193–197.
Lan Q, Zhang L, Li G, Vermeulen R, Weinberg RS, Dosemeci M et al. Hematotoxicity in workers exposed to low levels of benzene. Science 2004; 306: 1774–1776.
Sheltzer JM, Blank HM, Pfau SJ, Tange Y, George BM, Humpton TJ et al. Aneuploidy drives genomic instability in yeast. Science 2011; 333: 1026–1030.
Solomon DA, Kim T, Diaz-Martinez LA, Fair J, Elkahloun AG, Harris BT et al. Mutational inactivation of STAG2 causes aneuploidy in human cancer. Science 2011; 333: 1039–1043.
Johansson B, Mertens F, Heim S, Kristoffersson U, Mitelman F . Cytogenetics of secondary myelodysplasia (sMDS) and acute nonlymphocytic leukemia (sANLL). Eur J Haematol 1991; 47: 17–27.
Davico L, Sacerdote C, Ciccone G, Pegoraro L, Kerim S, Ponzio G et al. Chromosome 8, occupational exposures, smoking, and acute nonlymphocytic leukemias: a population-based study. Cancer Epidemiol Biomarkers Prev 1998; 7: 1123–1125.
Smith MT, Zhang L, Wang Y, Hayes RB, Li G, Wiemels J et al. Increased translocations and aneusomy in chromosomes 8 and 21 among workers exposed to benzene. Cancer Res 1998; 58: 2176–2181.
Zhang L, Eastmond DA, Smith MT . The nature of chromosomal aberrations detected in humans exposed to benzene. Crit Rev Toxicol 2002; 32: 1–42.
Zhang L, Lan Q, Guo W, Hubbard AE, Li G, Rappaport SM et al. Chromosome-wide aneuploidy study (CWAS) in workers exposed to an established leukemogen, benzene. Carcinogenesis 2011; 32: 605–612.
Zhang L, Lan Q, Guo W, Li G, Yang W, Hubbard AE et al. Use of OctoChrome fluorescence in situ hybridization to detect specific aneuploidy among all 24 chromosomes in benzene-exposed workers. Chem Biol Interact 2005; 153–154: 117–122.
Zhang L, Rothman N, Wang Y, Hayes RB, Li G, Dosemeci M et al. Increased aneusomy and long arm deletion of chromosomes 5 and 7 in the lymphocytes of Chinese workers exposed to benzene. Carcinogenesis 1998; 19: 1955–1961.
Stillman WS, Varella-Garcia M, Irons RD . The benzene metabolite, hydroquinone, selectively induces 5q31- and -7 in human CD34+CD19- bone marrow cells. Exp Hematol 2000; 28: 169–176.
Zhang L, Wang Y, Shang N, Smith MT . Benzene metabolites induce the loss and long arm deletion of chromosomes 5 and 7 in human lymphocytes. Leuk Res 1998; 22: 105–113.
Smith MT, Zhang L, Jeng M, Wang Y, Guo W, Duramad P et al. Hydroquinone, a benzene metabolite, increases the level of aneusomy of chromosomes 7 and 8 in human CD34-positive blood progenitor cells. Carcinogenesis 2000; 21: 1485–1490.
Vermeulen R, Li G, Lan Q, Dosemeci M, Rappaport SM, Bohong X et al. Detailed exposure assessment for a molecular epidemiology study of benzene in two shoe factories in China. Ann Occup Hyg 2004; 48: 105–116.
Waidyanatha S, Rothman N, Fustinoni S, Smith MT, Hayes RB, Bechtold W et al. Urinary benzene as a biomarker of exposure among occupationally exposed and unexposed subjects. Carcinogenesis 2001; 22: 279–286.
Zhang L, Rothman N, Wang Y, Hayes RB, Yin S, Titenko-Holland N et al. Benzene increases aneuploidy in the lymphocytes of exposed workers: a comparison of data obtained by fluorescence in situ hybridization in interphase and metaphase cells. Environ Mol Mutagen 1999; 34: 260–268.
Ihaka R, Gentleman R . R: A language for data analysis and graphics. J Comput Graph Stat 1996; 5: 299–314.
Hastie TJ, Tibshirani RJ . Generalized Additive Models. CRC Press: London, 1990.
Zhang L, Rothman N, Li G, Guo W, Yang W, Hubbard AE et al. Aberrations in chromosomes associated with lymphoma and therapy-related leukemia in benzene-exposed workers. Environ Mol Mutagen 2007; 48: 467–474.
Zhang L, Rothman N, Wang Y, Hayes RB, Bechtold W, Venkatesh P et al. Interphase cytogenetics of workers exposed to benzene. Environ Health Perspect 1996; 104 (Suppl 6): 1325–1329.
Subrahmanyam VV, Kolachana P, Smith MT . Metabolism of hydroquinone by human myeloperoxidase: Mechanisms of stimulation by other phenolic compounds. Arch BiochemBiophys 1991; 286: 76–84.
Smith MT . The mechanism of benzene-induced leukemia: a hypothesis and speculations on the causes of leukemia. Environ Health Perspect 1996; 104 (Suppl 6): 1219–1225.
Irons RD, Pfeifer RW, Aune TM, Pierce CW . Soluble immune response suppressor (SIRS) inhibits microtubule function in vivo and microtubule assembly in vitro. J Immunol 1984; 133: 2032–2036.
Irons RD, Dent JG, Baker TS, Rickert DE . Benzene is metabolized and covalently bound in bone marrow in situ. Chem Biol Interact 1980; 30: 241–245.
Zhang L, Venkatesh P, Creek ML, Smith MT . Detection of 1,2,4-benzenetriol induced aneuploidy and microtubule disruption by fluorescence in situ hybridization and immunocytochemistry. Mutat Res 1994; 320: 315–327.
Herrera LA, Prada D, Andonegui MA, Duenas-Gonzalez A . The epigenetic origin of aneuploidy. Curr Genomics 2008; 9: 43–50.
Ji Z, Zhang L, Peng V, Ren X, McHale CM, Smith MT . A comparison of the cytogenetic alterations and global DNA hypomethylation induced by the benzene metabolite, hydroquinone, with those induced by melphalan and etoposide. Leukemia 2010; 24: 986–991.
Shiga T, Suzuki H, Yamamoto A, Yamamoto H, Yamamoto K . Hydroquinone, a benzene metabolite, induces Hog1-dependent stress response signaling and causes aneuploidy in Saccharomyces cerevisiae. J Radiat Res (Tokyo) 2010; 51: 405–415.
Acknowledgements
We thank the participants for taking part in this study. This study was supported by NIH grants RO1ES06721, P42ES04705 and P30ES01896 (MTS), P42ES05948 and P30ES10126 (SMR), and NIH contract N01-CO-12400 with SAIC-Frederick, Inc.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
MTS and SMR have received consulting and expert testimony fees from law firms representing both plaintiffs and defendants in cases involving exposure to benzene. GL has received funds from the American Petroleum Institute for consulting on benzene-related health research. The remaining authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Zhang, L., Lan, Q., Ji, Z. et al. Leukemia-related chromosomal loss detected in hematopoietic progenitor cells of benzene-exposed workers. Leukemia 26, 2494–2498 (2012). https://doi.org/10.1038/leu.2012.143
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2012.143
Keywords
This article is cited by
-
The association of three DNA repair genes polymorphisms on the frequency of chromosomal alterations detected by fluorescence in situ hybridization
International Archives of Occupational and Environmental Health (2021)
-
Benzene poisoning, clinical and blood abnormalities in two Brazilian female gas station attendants: two case reports
BMC Research Notes (2017)