Abstract
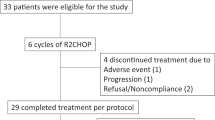
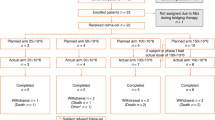
Lenalidomide was shown to have significant single-agent activity in relapsed aggressive non-Hodgkin's lymphoma (NHL). We conducted a phase I trial to establish the maximum tolerated dose of lenalidomide that could be combined with R-CHOP (rituximab–cyclophosphamide, doxorubicin, vincristine, and prednisone). Eligible patients were adults with newly diagnosed, untreated CD20 positive diffuse large cell or follicular grade III NHL. Patients received oral lenalidomide on days 1–10 with standard dose R-CHOP every 21 days. All patients received pegfilgrastim on day 2 of the cycle and aspirin prophylaxis. The lenalidomide dose levels tested were 15, 20 and 25 mg. A total of 24 patients were enrolled. The median age was 65 (35–82) years and 54% were over 60 years. Three patients received 15 mg, 3 received 20 mg and 18 received 25 mg of lenalidomide. No dose limiting toxicity was found, and 25 mg on days 1–10 is the recommended dose for phase II. The incidence of grade IV neutropenia and thrombocytopenia was 67% and 21%, respectively. Febrile neutropenia was rare (4%) and there were no toxic deaths. The overall response rate was 100% with a complete response rate of 77%. Lenalidomide at the dose of 25 mg/day administered on days 1 to 10 of 21-day cycle can be safely combined with R-CHOP in the initial chemotherapy of aggressive B-cell lymphoma.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Coiffier B, Lepage E, Briere J, Herbrecht R, Tilly H, Bouabdallah R et al. CHOP chemotherapy plus rituximab compared with CHOP alone in elderly patients with diffuse large-B-cell lymphoma. N Engl J Med 2002; 346: 235–242.
Habermann TM, Weller EA, Morrison VA, Gascoyne RD, Cassileth PA, Cohn JB et al. Rituximab-CHOP versus CHOP alone or with maintenance rituximab in older patients with diffuse large B-cell lymphoma. J Clin Oncol 2006; 24: 3121–3127.
Sehn LH, Donaldson J, Chhanabhai M, Fitzgerald C, Gill K, Klasa R et al. Introduction of combined CHOP plus rituximab therapy dramatically improved outcome of diffuse large B-cell lymphoma in British Columbia. J Clin Oncol 2005; 23: 5027–5033.
Pfreundschuh M, Trumper L, Osterborg A, Pettengell R, Trneny M, Imrie K et al. CHOP-like chemotherapy plus rituximab versus CHOP-like chemotherapy alone in young patients with good-prognosis diffuse large-B-cell lymphoma: a randomised controlled trial by the MabThera International Trial (MInT) Group. Lancet Oncol 2006; 7: 379–391.
Feugier P, Van Hoof A, Sebban C, Solal-Celigny P, Bouabdallah R, Ferme C et al. Long-term results of the R-CHOP study in the treatment of elderly patients with diffuse large B-cell lymphoma: a study by the Groupe d’Etude des Lymphomes de l’Adulte. J Clin Oncol 2005; 23 (18): 4117–4126.
Klimo P, Connors JM . MACOP-B chemotherapy for the treatment of diffuse large-cell lymphoma. Ann Intern Med 1985; 102: 596–602.
Fisher RI, Gaynor ER, Dahlberg S, Oken MM, Grogan TM, Mize EM et al. Comparison of a standard regimen (CHOP) with three intensive chemotherapy regimens for advanced non-Hodgkin's lymphoma. N Engl J Med 1993; 328: 1002–1006.
Gordon LI, Harrington D, Andersen J, Colgan J, Glick J, Neiman R et al. Comparison of a second-generation combination chemotherapeutic regimen (m-BACOD) with a standard regimen (CHOP) for advanced diffuse non-Hodgkin's lymphoma. N Eng J Med 1992; 327: 1342–1349.
Cooper IA, Wolf MM, Robertson TI, Fox RM, Matthews JP, Stone JM et al. Randomized comparison of MACOP-B with CHOP in patients with intermediate-grade non-Hodgkin's lymphoma. The Australian and New Zealand Lymphoma Group. J Clin Oncol 1994; 12: 769–778.
Sertoli MR, Santini G, Chisesi T, Congiu AM, Rubagotti A, Contu A et al. MACOP-B versus ProMACE-MOPP in the treatment of advanced diffuse non-Hodgkin's lymphoma: results of a prospective randomized trial by the non-Hodgkin's Lymphoma Cooperative Study Group. J Clin Oncol 1994; 12: 1366–1374.
Wiernik PH, Lossos IS, Tuscano JM, Justice G, Vose JM, Cole CE et al. Lenalidomide monotherapy in relapsed or refractory aggressive non-Hodgkin's lymphoma. J Clin Oncol 2008; 26: 4952–4957.
Ivanov V, Tabouret E, Chuto G, Chetaille B, Fezoui H, Coso D et al. Rituximab-lenalidomide-dexamethasone induces complete and durable remission in relapsed refractory diffuse large B-cell non-Hodgkin lymphoma. Leuk Lymphoma 2010; 51: 1758–1760.
Witzig TE, Vose JM, Zinzani PL, Reeder CB, Buckstein R, Polikoff JA et al. An international phase II trial of single-agent lenalidomide for relapsed or refractory aggressive B-Cell non-Hodgkin lymphoma. Ann Oncol 2011, (e-pub ahead of print).
Haslett PA, Hanekom WA, Muller G, Kaplan G . Thalidomide and a thalidomide analogue drug costimulate virus-specific CD8+ T cells in vitro. J Infect Dis 2003; 187: 946–955.
Zhang H, Vakil V, Braunstein M, Smith EL, Maroney J, Chen L et al. Circulating endothelial progenitor cells in multiple myeloma: implications and significance. Blood 2005; 105: 3286–3294.
Pellagatti A, Jadersten M, Forsblom AM, Cattan H, Christensson B, Emanuelsson EK et al. Lenalidomide inhibits the malignant clone and up-regulates the SPARC gene mapping to the commonly deleted region in 5q- syndrome patients. Proc Natl Acad Sci USA 2007; 104: 11406–11411.
Cheson BD, Pfistner B, Juweid ME, Gascoyne RD, Specht L, Horning SJ et al. Revised response criteria for malignant lymphoma. J Clin Oncol 2007; 25: 579–586.
Zhang L, Qian Z, Cai Z, Sun L, Wang H, Bartlett JB et al. Synergistic antitumor effects of lenalidomide and rituximab on mantle cell lymphoma in vitro and in vivo. Am J Hematol 2009; 84: 553–559.
Richardson PG, Schlossman RL, Weller E, Hideshima T, Mitsiades C, Davies F et al. Immunomodulatory drug CC-5013 overcomes drug resistance and is well tolerated in patients with relapsed multiple myeloma. Blood 2002; 100: 3063–3067.
Lu L, Payvandi F, Wu L, Zhang LH, Hariri RJ, Man HW et al. The anti-cancer drug lenalidomide inhibits angiogenesis and metastasis via multiple inhibitory effects on endothelial cell function in normoxic and hypoxic conditions. Mic Res 2009; 77: 78–86.
Wu L, Adams M, Carter T, Chen R, Muller G, Stirling D et al. Lenalidomide enhances natural killer cell and monocyte-mediated antibody-dependent cellular cytotoxicity of rituximab-treated CD20+ tumor cells. Clin Cancer Res 2008; 14: 4650–4657.
Smith TJ, Khatcheressian J, Lyman GH, Ozer H, Armitage JO, Balducci L et al. 2006 update of recommendations for the use of white blood cell growth factors: an evidence-based clinical practice guideline. J Clin Oncol 2006; 24: 3187–3205.
Czuczman MS . CHOP plus rituximab chemoimmunotherapy of indolent B-cell lymphoma. Semin oncol 1999; 26 (5 Suppl): 88–96.
Rajkumar SV, Blood E . Lenalidomide and venous thrombosis in multiple myeloma. N Eng J Med 2006; 354: 2079–2080.
Zonder JA, Barlogie B, Durie BG, McCoy J, Crowley J, Hussein MA . Thrombotic complications in patients with newly diagnosed multiple myeloma treated with lenalidomide and dexamethasone: benefit of aspirin prophylaxis. Blood 2006; 108: 403, author reply 404.
Caruso V, Di Castelnuovo A, Meschengieser S, Lazzari MA, de Gaetano G, Storti S et al. Thrombotic complications in adult patients with lymphoma: a meta-analysis of 29 independent cohorts including 18018 patients and 1149 events. Blood 2010; 115: 5322–5328.
Morton LM, Wang SS, Devesa SS, Hartge P, Weisenburger DD, Linet MS . Lymphoma incidence patterns by WHO subtype in the United States, 1992–2001. Blood 2006; 107: 265–276.
Habermann TM, Lossos IS, Justice G, Vose JM, Wiernik PH, McBride K et al. Lenalidomide oral monotherapy produces a high response rate in patients with relapsed or refractory mantle cell lymphoma. Br J Haematol 2009; 145: 344–349.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Nowakowski, G., LaPlant, B., Habermann, T. et al. Lenalidomide can be safely combined with R-CHOP (R2CHOP) in the initial chemotherapy for aggressive B-cell lymphomas: phase I study. Leukemia 25, 1877–1881 (2011). https://doi.org/10.1038/leu.2011.165
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2011.165
Keywords
This article is cited by
-
Efficacy and safety of lenalidomide in diffuse large B-cell lymphoma: a meta-analysis of randomized controlled trials
Clinical and Experimental Medicine (2022)
-
Targeting of inflammatory pathways with R2CHOP in high-risk DLBCL
Leukemia (2021)
-
B-cell non-Hodgkin lymphoma: importance of angiogenesis and antiangiogenic therapy
Angiogenesis (2020)
-
Genetic alterations and their clinical implications in DLBCL
Nature Reviews Clinical Oncology (2019)
-
Integrating precision medicine through evaluation of cell of origin in treatment planning for diffuse large B-cell lymphoma
Blood Cancer Journal (2019)