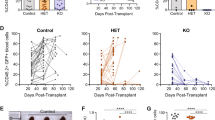
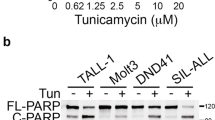
Abstract
Activation of the Notch pathway occurs commonly in T acute lymphoblastic leukemia (T-ALL) because of mutations in Notch1 or Fbw7 and is involved in the regulation of cell proliferation and survival. Deregulated Notch3 signalling has also been shown to promote leukemogenesis in transgenic mice, but the targets of Notch3 in human T-ALL cells remain poorly characterized. Here, we show that Notch3 controls levels of mitogen-activated protein kinase (MAPK) phosphatase 1 (MKP-1). In a model of T-ALL cell dormancy, both Notch3 activation and MKP-1 expression were upregulated in aggressive compared with dormant tumors, and this inversely correlated with the levels of phosphorylated p38 and extracellular signal-regulated kinase1/2 (ERK1/2) MAPKs, two canonical MKP-1 targets. We demonstrate that MKP-1 protein levels are regulated by Notch3 in T-ALL cell lines because its silencing by RNA interference or treatment with γ-secretase inhibitors induced strong MKP-1 reduction whereas activation of Notch3 signalling had the opposite effect. Furthermore, MKP-1 has an important role in T-ALL cell survival because its attenuation by short hairpin RNA significantly increased cell death under stress conditions. This protective function has a key role in vivo, as MKP-1-deficient cells showed impaired tumorigenicity. These results elucidate a novel mechanism downstream of Notch3 that controls the survival of T-ALL cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Grabher C, von Boehmer H, Look AT . Notch 1 activation in the molecular pathogenesis of T-cell acute lymphoblastic leukaemia. Nat Rev Cancer 2006; 6: 347–359.
Weng AP, Ferrando AA, Lee W, Morris IV JP, Silverman LB, Sanchez-Irizarry C et al. Activating mutations of NOTCH1 in human T cell acute lymphoblastic leukemia. Science 2004; 306: 269–271.
Matsuoka S, Oike Y, Onoyama I, Iwama A, Arai F, Takubo K et al. Fbxw7 acts as a critical fail-safe against premature loss of hematopoietic stem cells and development of T-ALL. Genes Dev 2008; 22: 986–991.
Thompson BJ, Buonamici S, Sulis ML, Palomero T, Vilimas T, Basso G et al. The SCFFBW7 ubiquitin ligase complex as a tumor suppressor in T cell leukemia. J Exp Med 2007; 204: 1825–1835.
Palomero T, Sulis ML, Cortina M, Real PJ, Barnes K, Ciofani M et al. Mutational loss of PTEN induces resistance to NOTCH1 inhibition in T-cell leukemia. Nat Med 2007; 13: 1203–1210.
Dohda T, Maljukova A, Liu L, Heyman M, Grander D, Brodin D et al. Notch signaling induces SKP2 expression and promotes reduction of p27Kip1 in T-cell acute lymphoblastic leukemia cell lines. Exp Cell Res 2007; 313: 3141–3152.
Palomero T, Lim WK, Odom DT, Sulis ML, Real PJ, Margolin A et al. NOTCH1 directly regulates c-MYC and activates a feed-forward-loop transcriptional network promoting leukemic cell growth. Proc Natl Acad Sci USA 2006; 103: 18261–18266.
Weng AP, Millholland JM, Yashiro-Ohtani Y, Arcangeli ML, Lau A, Wai C et al. c-Myc is an important direct target of Notch1 in T-cell acute lymphoblastic leukemia/lymphoma. Genes Dev 2006; 20: 2096–2109.
Aifantis I, Vilimas T, Buonamici S . Notches, NFkappaBs and the making of T cell leukemia. Cell cycle 2007; 6: 403–406.
Liu WH, Hsiao HW, Tsou WI, Lai MZ . Notch inhibits apoptosis by direct interference with XIAP ubiquitination and degradation. EMBO J 2007; 26: 1660–1669.
Mungamuri SK, Yang X, Thor AD, Somasundaram K . Survival signaling by Notch1: mammalian target of rapamycin (mTOR)-dependent inhibition of p53. Cancer Res 2006; 66: 4715–4724.
Bellavia D, Campese AF, Alesse E, Vacca A, Felli MP, Balestri A et al. Constitutive activation of NF-kappaB and T-cell leukemia/lymphoma in Notch3 transgenic mice. EMBO J 2000; 19: 3337–3348.
Aifantis I, Raetz E, Buonamici S . Molecular pathogenesis of T-cell leukaemia and lymphoma. Nat Rev Immunol 2008; 8: 380–390.
Lee SH, Jeong EG, Yoo NJ, Lee SH . Mutational analysis of NOTCH1, 2, 3 and 4 genes in common solid cancers and acute leukemias. Apmis 2007; 115: 1357–1363.
Indraccolo S, Minuzzo S, Masiero M, Pusceddu I, Persano L, Moserle L et al. Cross-talk between tumor and endothelial cells involving the Notch3-Dll4 interaction marks escape from tumor dormancy. Cancer Res 2009; 69: 1314–1323.
Vacca A, Felli MP, Palermo R, Di Mario G, Calce A, Di Giovine M et al. Notch3 and pre-TCR interaction unveils distinct NF-kappaB pathways in T-cell development and leukemia. EMBO J 2006; 25: 1000–1008.
Aguirre-Ghiso JA . Models, mechanisms and clinical evidence for cancer dormancy. Nat Rev Cancer 2007; 7: 834–846.
Aguirre Ghiso JA, Kovalski K, Ossowski L . Tumor dormancy induced by downregulation of urokinase receptor in human carcinoma involves integrin and MAPK signaling. J Cell Biol 1999; 147: 89–104.
Aguirre-Ghiso JA, Estrada Y, Liu D, Ossowski L . ERK(MAPK) activity as a determinant of tumor growth and dormancy; regulation by p38(SAPK). Cancer Res 2003; 63: 1684–1695.
Boutros T, Chevet E, Metrakos P . Mitogen-activated protein (MAP) kinase/MAP kinase phosphatase regulation: roles in cell growth, death, and cancer. Pharmacol Rev 2008; 60: 261–310.
Indraccolo S, Stievano L, Minuzzo S, Tosello V, Esposito G, Piovan E et al. Interruption of tumor dormancy by a transient angiogenic burst within the tumor microenvironment. Proc Natl Acad Sci USA 2006; 103: 4216–4221.
Keyaerts M, Verschueren J, Bos TJ, Tchouate-Gainkam LO, Peleman C, Breckpot K et al. Dynamic bioluminescence imaging for quantitative tumour burden assessment using IV or IP administration of D: -luciferin: effect on intensity, time kinetics and repeatability of photon emission. Eur J Nucl Med Mol Imaging 2008; 35: 999–1007.
Indraccolo S, Tisato V, Tosello V, Habeler W, Esposito G, Moserle L et al. Interferon-alpha gene therapy by lentiviral vectors contrasts ovarian cancer growth through angiogenesis inhibition. Hum Gene Therapy 2005; 16: 957–970.
Indraccolo S, Habeler W, Tisato V, Stievano L, Piovan E, Tosello V et al. Gene transfer in ovarian cancer cells: a comparison between retroviral and lentiviral vectors. Cancer Res 2002; 62: 6099–6107.
Sansone P, Storci G, Tavolari S, Guarnieri T, Giovannini C, Taffurelli M et al. IL-6 triggers malignant features in mammospheres from human ductal breast carcinoma and normal mammary gland. J Clin Invest 2007; 117: 3988–4002.
Wu JJ, Bennett AM . Essential role for mitogen-activated protein (MAP) kinase phosphatase-1 in stress-responsive MAP kinase and cell survival signaling. J Biol Chem 2005; 280: 16461–16466.
Brondello JM, Pouyssegur J, McKenzie FR . Reduced MAP kinase phosphatase-1 degradation after p42/p44MAPK-dependent phosphorylation. Science 1999; 286: 2514–2517.
Kassel O, Sancono A, Kratzschmar J, Kreft B, Stassen M, Cato AC . Glucocorticoids inhibit MAP kinase via increased expression and decreased degradation of MKP-1. EMBO J 2001; 20: 7108–7116.
Li M, Zhou JY, Ge Y, Matherly LH, Wu GS . The phosphatase MKP1 is a transcriptional target of p53 involved in cell cycle regulation. J Biol Chem 2003; 278: 41059–41068.
Lin YW, Yang JL . Cooperation of ERK and SCFSkp2 for MKP-1 destruction provides a positive feedback regulation of proliferating signaling. J Biol Chem 2006; 281: 915–926.
Yaglom J, O'Callaghan-Sunol C, Gabai V, Sherman MY . Inactivation of dual-specificity phosphatases is involved in the regulation of extracellular signal-regulated kinases by heat shock and hsp72. Mol Cell Biol 2003; 23: 3813–3824.
Chattopadhyay S, Machado-Pinilla R, Manguan-Garcia C, Belda-Iniesta C, Moratilla C, Cejas P et al. MKP1/CL100 controls tumor growth and sensitivity to cisplatin in non-small-cell lung cancer. Oncogene 2006; 25: 3335–3345.
Liu Y, Shepherd EG, Nelin LD . MAPK phosphatases—regulating the immune response. Nat Rev Immunol 2007; 7: 202–212.
Keyse SM . Dual-specificity MAP kinase phosphatases (MKPs) and cancer. Cancer Metastasis Rev 2008; 27: 253–261.
Zhang Y, Reynolds JM, Chang SH, Martin-Orozco N, Chung Y, Nurieva RI et al. MKP-1 is necessary for T cell activation and function. J Biol Chem 2009; 284: 30815–30824.
Bulavin DV, Fornace Jr AJ . p38 MAP kinase's emerging role as a tumor suppressor. Adv Cancer Res 2004; 92: 95–118.
Kennedy NJ, Cellurale C, Davis RJ . A radical role for p38 MAPK in tumor initiation. Cancer Cell 2007; 11: 101–103.
Bellavia D, Campese AF, Vacca A, Gulino A, Screpanti I . Notch3, another Notch in T cell development. Semin Immunol 2003; 15: 107–112.
Rothenberg EV . Notchless T cell maturation? Nature Immunol 2001; 2: 189–190.
Berset T, Hoier EF, Battu G, Canevascini S, Hajnal A . Notch inhibition of RAS signaling through MAP kinase phosphatase LIP-1 during C. elegans vulval development. Science 2001; 291: 1055–1058.
Kondoh K, Sunadome K, Nishida E . Notch signaling suppresses p38 MAPK activity via induction of MKP-1 in myogenesis. J Biol Chem 2007; 282: 3058–3065.
Sarmento LM, Huang H, Limon A, Gordon W, Fernandes J, Tavares MJ et al. Notch1 modulates timing of G1-S progression by inducing SKP2 transcription and p27 Kip1 degradation. J Exp Med 2005; 202: 157–168.
Acknowledgements
We are grateful to Annunciata Vecchi (Mario Negri Institute) for providing SIEC, Adolfo Ferrando (Columbia University) for providing T-ALL cell lines, Minhong Yan (Genentech) for the YW152F mAb, Micol Silic-Benussi (University of Padova) for technical help with some experiments. This work was supported in part by grants from Associazione Italiana per la Ricerca sul Cancro (AIRC) and Fondazione Italiana per la Ricerca sul Cancro (FIRC); Ministry of University and Research, 60% and PRIN; Ministry of Health, Oncology Program 2006 and Ricerca Finalizzata 2007; Banca Popolare di Verona; Foundation CARIPARO Projects of Excellence Program 2008.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Leukemia website
Rights and permissions
About this article
Cite this article
Masiero, M., Minuzzo, S., Pusceddu, I. et al. Notch3-mediated regulation of MKP-1 levels promotes survival of T acute lymphoblastic leukemia cells. Leukemia 25, 588–598 (2011). https://doi.org/10.1038/leu.2010.323
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2010.323
Keywords
This article is cited by
-
Molecular effects of genistein, as a potential anticancer agent, on CXCR-4 and VEGF pathway in acute lymphoblastic leukemia
Molecular Biology Reports (2022)
-
Histone deacetylase 6 controls Notch3 trafficking and degradation in T-cell acute lymphoblastic leukemia cells
Oncogene (2018)
-
NOTCH3 inactivation increases triple negative breast cancer sensitivity to gefitinib by promoting EGFR tyrosine dephosphorylation and its intracellular arrest
Oncogenesis (2018)
-
The EphB6 receptor is overexpressed in pediatric T cell acute lymphoblastic leukemia and increases its sensitivity to doxorubicin treatment
Scientific Reports (2017)
-
The critical role of macrophages in the pathogenesis of hidradenitis suppurativa
Inflammation Research (2017)