Abstract
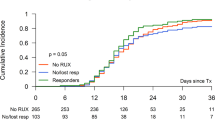
In a previous study, we reported on the safety and efficacy of low-dose (0.5 mg) pomalidomide and prednisone and pomalidomide alone (2 mg/day), for the treatment of anemia associated with myelofibrosis (MF). The current study examined the value of low-dose pomalidomide alone. The main eligibility criterion was transfusion-dependency or hemoglobin <10 gm per 100 ml. Anemia response was assessed by International Working Group criteria. Pomalidomide (0.5 mg/day) was given to 58 patients (median age 68 years); 46 (79%) were transfusion-dependent and 42 were JAK2V617F positive. Anemia response was documented only in the presence of JAK2V617F (24 vs 0%; P=0.03) but was not further affected by mutant allele burden (P=0.39); 9 of the 10 anemia responders became transfusion independent. Anemia response in JAK2V617F-positive patients was predicted by the presence of pomalidomide-induced basophilia in the first month of therapy (38 vs 6%; P=0.02) or absence of marked splenomegaly (38 vs 11%; P=0.05). A total of 14 (58%) of 24 patients with a platelet count of ⩽100 × 109 cells/l experienced a >50% increment in platelet count. There were no spleen responses. Grade 3 or 4 thrombocytopenia/neutropenia occurred in 2%/0% of patients. Low-dose pomalidomide is effective in the treatment of anemia associated with JAK2V617F-positive MF; response is predicted by early drug-induced basophilia.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Bartlett JB, Dredge K, Dalgleish AG . The evolution of thalidomide and its IMiD derivatives as anticancer agents. Nat Rev Cancer 2004; 4: 314–322.
Weber DM, Chen C, Niesvizky R, Wang M, Belch A, Stadtmauer EA et al. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med 2007; 357: 2133–2142.
Dimopoulos M, Spencer A, Attal M, Prince HM, Harousseau JL, Dmoszynska A et al. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med 2007; 357: 2123–2132.
Rajkumar SV, Hayman S, Gertz MA, Dispenzieri A, Lacy MQ, Greipp PR et al. Combination therapy with thalidomide plus dexamethasone for newly diagnosed myeloma. J Clin Oncol 2002; 20: 4319–4323.
List A, Dewald G, Bennett J, Giagounidis A, Raza A, Feldman E et al. Lenalidomide in the myelodysplastic syndrome with chromosome 5q deletion. N Engl J Med 2006; 355: 1456–1465.
Barosi G, Grossi A, Comotti B, Musto P, Gamba G, Marchetti M . Safety and efficacy of thalidomide in patients with myelofibrosis with myeloid metaplasia. Br J Haematol 2001; 114: 78–83.
Abgrall JF, Guibaud I, Bastie JN, Flesch M, Rossi JF, Lacotte-Thierry L et al. Thalidomide versus placebo in myeloid metaplasia with myelofibrosis: a prospective, randomized, double-blind, multicenter study. Haematologica 2006; 91: 1027–1032.
Ochonisky S, Verroust J, Bastuji-Garin S, Gherardi R, Revuz J . Thalidomide neuropathy incidence and clinico-electrophysiologic findings in 42 patients. Arch Dermatol 1994; 130: 66–69.
Elliott MA, Mesa RA, Li CY, Hook CC, Ansell SM, Levitt RM et al. Thalidomide treatment in myelofibrosis with myeloid metaplasia. Br J Haematol 2002; 117: 288–296.
Mesa RA, Steensma DP, Pardanani A, Li CY, Elliott M, Kaufmann SH et al. A phase 2 trial of combination low-dose thalidomide and prednisone for the treatment of myelofibrosis with myeloid metaplasia. Blood 2003; 101: 2534–2541.
Barosi G, Elliott M, Canepa L, Ballerini F, Piccaluga PP, Visani G et al. Thalidomide in myelofibrosis with myeloid metaplasia: a pooled-analysis of individual patient data from five studies. Leuk Lymphoma 2002; 43: 2301–2307.
Marchetti M, Barosi G, Balestri F, Viarengo G, Gentili S, Barulli S et al. Low-dose thalidomide ameliorates cytopenias and splenomegaly in myelofibrosis with myeloid metaplasia: a phase II trial. J Clin Oncol 2004; 22: 424–431.
Barosi G . Myelofibrosis with myeloid metaplasia. Hematol Oncol Clin North Am 2003; 17: 1211–1226.
Tefferi A, Cortes J, Verstovsek S, Mesa RA, Thomas D, Lasho TL et al. Lenalidomide therapy in myelofibrosis with myeloid metaplasia. Blood 2006; 108: 1158–1164.
Mesa RA, Yao X, Cripe LD, Li CY, Litzow M, Paietta E et al. Lenalidomide and prednisone for myelofibrosis: Eastern Cooperative Oncology Group (ECOG) phase-2 trial E4903. Blood 2010. (in press).
Quintas-Cardama A, Kantarjian HM, Manshouri T, Thomas D, Cortes J, Ravandi F . et al. Lenalidomide plus prednisone results in durable clinical, histopathologic, and molecular responses in patients with myelofibrosis. J Clin Oncol 2009; 27: 4760–4766.
Tefferi A, Lasho TL, Mesa RA, Pardanani A, Ketterling RP, Hanson CA . Lenalidomide therapy in del(5)(q31)-associated myelofibrosis: cytogenetic and JAK2V617F molecular remissions. Leukemia 2007; 21: 1827–1828.
Schey SA, Fields P, Bartlett JB, Clarke IA, Ashan G, Knight RD et al. Phase I study of an immunomodulatory thalidomide analog, CC-4047, in relapsed or refractory multiple myeloma. J Clin Oncol 2004; 22: 3269–3276.
Mesa RA, Pardanani AD, Hussein K, Wu W, Schwager S, Litzow MR et al. Phase1/-2 study of pomalidomide in myelofibrosis. Am J Hematol 2010; 85: 129–130.
Lacy MQ, Hayman SR, Gertz MA, Dispenzieri A, Buadi F, Kumar S et al. Pomalidomide (CC4047) plus low-dose dexamethasone as therapy for relapsed multiple myeloma. J Clin Oncol 2009; 27: 5008–5014.
Tefferi A, Verstovsek S, Barosi G, Passamonti F, Roboz GJ, Gisslinger H et al. Pomalidomide is active in the treatment of anemia associated with myelofibrosis. J Clin Oncol 2009; 27: 4563–4569.
Swerdlow SH, Campo E, NL H et al. WHO Classification of Tumors of Haematopoietic and Lymphoid Tissues. 4th edn. International Agency for research on Cancer (IARC): Lyon, France, 2008.
Tefferi A, Barosi G, Mesa RA, Cervantes F, Deeg HJ, Reilly JT et al. International Working Group (IWG) consensus criteria for treatment response in myelofibrosis with myeloid metaplasia, for the IWG for Myelofibrosis Research and Treatment (IWG-MRT). Blood 2006; 108: 1497–1503.
Passamonti F, Cervantes F, Vannucchi AM, Morra E, Rumi E, Pereira A et al. A dynamic prognostic model to predict survival in primary myelofibrosis: a study by the IWG-MRT (International Working Group for Myeloproliferative Neoplasms Research and Treatment). Blood 2010; 115: 1703–1708.
Hussein K, Pardanani AD, Van Dyke DL, Hanson CA, Tefferi A . International Prognostic Scoring System-independent cytogenetic risk categorization in primary myelofibrosis. Blood 2010; 115: 496–499.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Begna, K., Mesa, R., Pardanani, A. et al. A phase-2 trial of low-dose pomalidomide in myelofibrosis. Leukemia 25, 301–304 (2011). https://doi.org/10.1038/leu.2010.254
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2010.254
Keywords
This article is cited by
-
Management of Myelofibrosis-Related Cytopenias
Current Hematologic Malignancy Reports (2018)
-
Philadelphia chromosome-negative classical myeloproliferative neoplasms: revised management recommendations from European LeukemiaNet
Leukemia (2018)
-
Pomalidomide in myeloproliferative neoplasm-associated myelofibrosis
Leukemia (2017)
-
A randomized study of pomalidomide vs placebo in persons with myeloproliferative neoplasm-associated myelofibrosis and RBC-transfusion dependence
Leukemia (2017)
-
Pharmacotherapy of Myelofibrosis
Drugs (2017)