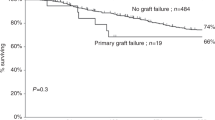
Abstract
The role of graft-versus-malignancy reactivity in the effects of allogeneic hematopoietic stem cell transplantation and donor lymphocyte infusion (DLI) for myelodysplastic syndromes is as yet not well established. Clinical data are limited and animal models are scarce. Here, we report on the effects of allogeneic bone marrow transplantation (alloBMT) and DLI in a novel model of irradiation-induced murine myelodysplastic/myeloproliferation syndrome (MD/MPS). Total body irradiation with 8.5 Gy in SJL/J mice gave rise to a lethal wasting syndrome in 60% of mice, characterized by 1° normocellular bone marrow with dysplastic features in erythroid, myeloid and megakaryocytic cell lineages, 2° lymphosplenomegaly with spleens harboring a prominent extramedullary hematopoiesis with erythroid, myeloid and megakaryocytic lineages exhibiting dysplastic features, and foci of dysplastic hematomyelopoiesis in the liver, 3° peripheral thrombocytopenia and 4° evidence of disseminated infection or leukemic transformation in selected animals. This clinicopathological picture was consistent with a murine form of MD/MPS. Syngeneic or allogeneic (BALB/c) T cell-depleted BMT could not prevent the occurrence of lethal MD/MPS. In contrast, DLI at weeks 2–4 after BMT led to restoration of the dysbalanced hematomyelopoiesis. However, severe DLI-induced acute graft-versus-host disease occurred, precluding a survival advantage. We present evidence of the existence of a post-alloBMT DLI-induced graft-versus-MD/MPS effect in murine irradiation-induced MD/MPS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Mufti GJ . Pathobiology, classification, and diagnosis of myelodysplastic syndrome. Best Pract Res Clin Haematol 2004; 17: 543–557.
Vardiman JW . Myelodysplastic syndromes, chronic myeloproliferative diseases, and myelodysplastic/myeloproliferative diseases. Semin Diagn Pathol 2003; 20: 154–179.
Van Etten RA, Shannon KM . Focus on myeloproliferative diseases and myelodysplastic syndromes. Cancer Cell 2004; 6: 547–552.
Larson RA . Myelodysplasia: when to treat and how. Best Pract Res Clin Haematol 2006; 19: 293–300.
Schiffer CA . Myelodysplasia: the good, the fair and the ugly. Best Pract Res Clin Haematol 2007; 20: 49–55.
Anderson JE, Appelbaum FR, Fisher LD, Schoch G, Shulman H, Anasetti C et al. Allogeneic bone marrow transplantation for 93 patients with myelodysplastic syndrome. Blood 1993; 82: 677–681.
Runde V, de Witte T, Arnold R, Gratwohl A, Hermans J, van Biezen A et al. Bone marrow transplantation from HLA-identical siblings as first-line treatment in patients with myelodysplastic syndromes: early transplantation is associated with improved outcome. Chronic Leukemia Working Party of the European Group for Blood and Marrow Transplantation. Bone Marrow Transplant 1998; 21: 255–261.
Deeg HJ, Storer B, Slattery JT, Anasetti C, Doney KC, Hansen JA et al. Conditioning with targeted busulfan and cyclophosphamide for hemopoietic stem cell transplantation from related and unrelated donors in patients with myelodysplastic syndrome. Blood 2002; 100: 1201–1207.
Castro-Malaspina H, Harris RE, Gajewski J, Ramsay N, Collins R, Dharan B et al. Unrelated donor marrow transplantation for myelodysplastic syndromes: outcome analysis in 510 transplants facilitated by the National Marrow Donor Program. Blood 2002; 99: 1943–1951.
Ho AY, Pagliuca A, Kenyon M, Parker JE, Mijovic A, Devereux S et al. Reduced-intensity allogeneic hematopoietic stem cell transplantation for myelodysplastic syndrome and acute myeloid leukemia with multilineage dysplasia using fludarabine, busulphan, and alemtuzumab (FBC) conditioning. Blood 2004; 104: 1616–1623.
Deeg HJ . Transplant strategies for patients with myelodysplastic syndromes. Curr Opin Hematol 2006; 13: 61–66.
Appelbaum FR . Haematopoietic cell transplantation as immunotherapy. Nature 2001; 411: 385–389.
Collins Jr RH, Shpilberg O, Drobyski WR, Porter DL, Giralt S, Champlin R et al. Donor leukocyte infusions in 140 patients with relapsed malignancy after allogeneic bone marrow transplantation. J Clin Oncol 1997; 15: 433–444.
Porter DL, Collins Jr RH, Hardy C, Kernan NA, Drobyski WR, Giralt S et al. Treatment of relapsed leukemia after unrelated donor marrow transplantation with unrelated donor leukocyte infusions. Blood 2000; 95: 1214–1221.
Levine JE, Braun T, Penza SL, Beatty P, Cornetta K, Martino R et al. Prospective trial of chemotherapy and donor leukocyte infusions for relapse of advanced myeloid malignancies after allogeneic stem-cell transplantation. J Clin Oncol 2002; 20: 405–412.
Depil S, Deconinck E, Milpied N, Sutton L, Witz F, Jouet JP et al. Donor lymphocyte infusion to treat relapse after allogeneic bone marrow transplantation for myelodysplastic syndrome. Bone Marrow Transplant 2004; 33: 531–534.
Bethge WA, Hegenbart U, Stuart MJ, Storer BE, Maris MB, Flowers ME et al. Adoptive immunotherapy with donor lymphocyte infusions after allogeneic hematopoietic cell transplantation following nonmyeloablative conditioning. Blood 2004; 103: 790–795.
Campregher PV, Gooley T, Scott BL, Moravec C, Sandmaier B, Martin PJ et al. Results of donor lymphocyte infusions for relapsed myelodysplastic syndrome after hematopoietic cell transplantation. Bone Marrow Transplant 2007; 40: 965–971.
Pollyea DA, Artz AS, Stock W, Daugherty C, Godley L, Odenike OM et al. Outcomes of patients with AML and MDS who relapse or progress after reduced intensity allogeneic hematopoietic cell transplantation. Bone Marrow Transplant 2007; 40: 1027–1032.
Buonamici S, Li D, Chi Y, Zhao R, Wang X, Brace L et al. EVI1 induces myelodysplastic syndrome in mice. J Clin Invest 2004; 114: 713–719.
Futterer A, Campanero MR, Leonardo E, Criado LM, Flores JM, Hernandez JM et al. Dido gene expression alterations are implicated in the induction of hematological myeloid neoplasms. J Clin Invest 2005; 115: 2351–2362.
Lin YW, Slape C, Zhang Z, Aplan PD . NUP98-HOXD13 transgenic mice develop a highly penetrant, severe myelodysplastic syndrome that progresses to acute leukemia. Blood 2005; 106: 287–295.
Putz G, Rosner A, Nuesslein I, Schmitz N, Buchholz F . AML1 deletion in adult mice causes splenomegaly and lymphomas. Oncogene 2006; 25: 929–939.
Haran-Ghera N, Resnitzky P, Krautghamer R, Tartakovsky B . Multiphase process involved in radiation induced murine AML. Leukemia 1992; 6 (Suppl 3): s123–s125.
Verhoef GE, Demuynck H, Zachee P, Boogaerts MA . Myelodysplastic syndrome evolving into a myeloproliferative disorder: one disease or two? Leukemia 1994; 8: 714–715.
Sefrioui H, Billiau AD, Waer M . Graft-versus-leukemia effect in minor antigen mismatched chimeras given delayed donor leucocyte infusion: immunoregulatory aspects and role of donor T and ASGM1-positive cells. Transplantation 2000; 70: 348–353.
Billiau AD, Fevery S, Rutgeerts O, Landuyt W, Waer M . Transient expansion of Mac1+Ly6-G+Ly6-C+ early myeloid cells with suppressor activity in spleens of murine radiation marrow chimeras: possible implications for the graft-versus-host and graft-versus-leukemia reactivity of donor lymphocyte infusions. Blood 2003; 102: 740–748.
Fevery S, Billiau AD, Sprangers B, Rutgeerts O, Lenaerts C, Goebels J et al. CTLA-4 blockade in murine bone marrow chimeras induces a host-derived antileukemic effect without graft-versus-host disease. Leukemia 2007; 21: 1451–1459.
Kogan SC, Ward JM, Anver MR, Berman JJ, Brayton C, Cardiff RD et al. Bethesda proposals for classification of nonlymphoid hematopoietic neoplasms in mice. Blood 2002; 100: 238–245.
Murphy ED . SJLJ/J, a new inbred strain of mouse with a high early incidence of reticulum cell neoplasma. Proc Am Assoc Cancer Res 1963; 4: 46.
Johnson BD, McCabe C, Hanke CA, Truitt RL . Use of anti-CD3 epsilon F(ab’)2 fragments in vivo to modulate graft-versus-host disease without loss of graft-versus-leukemia reactivity after MHC-matched bone marrow transplantation. J Immunol 1995; 154: 5542–5554.
Miconnet I, Roger T, Seman M, Bruley-Rosset M . Critical role of endogenous Mtv in acute lethal graft-versus-host disease. Eur J Immunol 1995; 25: 364–368.
Cilloni D, Messa E, Messa F, Carturan S, Defilippi I, Arruga F et al. Genetic abnormalities as targets for molecular therapies in myelodysplastic syndromes. Ann NY Acad Sci 2006; 1089: 411–423.
Trakhtenbrot L, Krauthgamer R, Resnitzky P, Haran-Ghera N . Deletion of chromosome 2 is an early event in the development of radiation-induced myeloid leukemia in SJL/J mice. Leukemia 1988; 2: 545–550.
Haran-Ghera N, Krautghamer R, Lapidot T, Peled A, Dominguez MG, Stanley ER . Increased circulating colony-stimulating factor-1 (CSF-1) in SJL/J mice with radiation-induced acute myeloid leukemia (AML) is associated with autocrine regulation of AML cells by CSF-1. Blood 1997; 89: 2537–2545.
Sprangers B, Van Wijmeersch B, Fevery S, Waer M, Billiau AD . Experimental and clinical approaches for optimization of the graft-versus-leukemia effect. Nat Clin Pract Oncol 2007; 4: 404–414.
Porter D, Levine JE . Graft-versus-host disease and graft-versus-leukemia after donor leukocyte infusion. Semin Hematol 2006; 43: 53–61.
Mapara MY, Kim YM, Wang SP, Bronson R, Sachs DH, Sykes M . Donor lymphocyte infusions mediate superior graft-versus-leukemia effects in mixed compared to fully allogeneic chimeras: a critical role for host antigen-presenting cells. Blood 2002; 100: 1903–1909.
Billiau AD, Fevery S, Rutgeerts O, Landuyt W, Waer M . Crucial role of timing of donor lymphocyte infusion in generating dissociated graft-versus-host and graft-versus-leukemia responses in mice receiving allogeneic bone marrow transplants. Blood 2002; 100: 1894–1902.
Merad M, Hoffmann P, Ranheim E, Slaymaker S, Manz MG, Lira SA et al. Depletion of host Langerhans cells before transplantation of donor alloreactive T cells prevents skin graft-versus-host disease. Nat Med 2004; 10: 510–517.
Reddy P, Maeda Y, Liu C, Krijanovski OI, Korngold R, Ferrara JL . A crucial role for antigen-presenting cells and alloantigen expression in graft-versus-leukemia responses. Nat Med 2005; 11: 1244–1249.
Chakraverty R, Sykes M . The role of antigen-presenting cells in triggering graft-versus-host disease and graft-versus-leukemia. Blood 2007; 110: 9–17.
Acknowledgements
Gregor Verhoef and Michel Delforge are gratefully acknowledged for critical reading of the manuscript. This work was supported by grants from the Flanders Fund for Scientific Research, the Belgian Federation against Cancer and the WOMS (Scientific Research Multiple Sclerosis) Foundation. Bénédicte Dubois is supported by the University Research Council of the University of Leuven, Belgium. Ben Sprangers and Bart Van Wijmeersch are doctoral fellows, An D Billiau a postdoctoral fellow of the Flanders Fund for Scientific Research.
Author information
Authors and Affiliations
Corresponding author
Additional information
Statement of authorship
BS, BVW and ADB designed and performed all experiments, some of which assisted by AL and LD, and analyzed all data. OR and CL provided technical assistance. WL performed irradiation. AL and NB performed analysis of CBC and BM smears. XS and CD-P performed pathological studies. BS and ADB wrote the manuscript. BD and MW oversaw the study and critically revised the manuscript.
Rights and permissions
About this article
Cite this article
Sprangers, B., Van Wijmeersch, B., Luyckx, A. et al. Allogeneic bone marrow transplantation and donor lymphocyte infusion in a mouse model of irradiation-induced myelodysplastic/myeloproliferation syndrome (MD/MPS): evidence for a graft-versus-MD/MPS effect. Leukemia 23, 340–349 (2009). https://doi.org/10.1038/leu.2008.298
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.298