Abstract
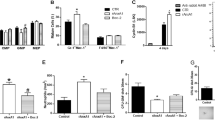
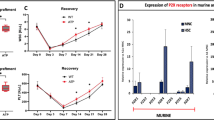
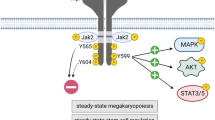
We have recently identified a human homolog of a fungal nuclear migration protein (hNUDC) that binds specifically with the extracellular domain of thrombopoietin receptor (Mpl). Preliminary studies with human CD34+ cells cultured in serum-free medium and normal mice showed that hNUDC appears to act as a cytokine, triggering many of the same responses as thrombopoietin (TPO). More intriguingly, recent data gained using a NIH 3T3 system have demonstrated that hNUDC exerts its biological activities through activation of Mpl. In this study, we further compared the biological functions of hNUDC with TPO in an EPO-dependent UT-7 cell line that was engineered to express the thrombopoietin receptor (Mpl). These Mpl-expressing cells following stimulation by either hNUDC or TPO exhibited overlapping patterns of megakaryocytic proliferation and differentiation, manifested by cell morphological change, polyploidy and expression of CD41+. Similar with TPO, hNUDC induced a sustained activation of the extracellular signal-regulated protein kinases-1 and -2 (ERK1/2) as well as p38 mitogen-activated kinase (p38 MAPK) pathways and these activations were inhibited in the presence of PD98059 or SB203580. Further evidence is provided that PD98059 or SB203580 inhibited hNUDC- or TPO-induced cell proliferation and differentiation, suggesting that ERK1/2 and p38 MAPK pathways are necessary in megakaryocyte development.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Ellis MH, Avraham H, Groopman JE . The regulation of megakaryocytopoiesis. Blood Rev 1995; 9: 1–6.
de Sauvage FJ, Hass PE, Spencer SD, Malloy BE, Gurney AL, Spencer SA et al. Stimulation of megakaryocytopoiesis and thrombopoiesis by the c-Mpl ligand. Nature 1994; 369: 533–538.
Lok S, Kaushansky K, Holly RD, Kuijper JL, Lofton-Day CE, Oort PJ et al. Cloning and expression of murine thrombopoietin cDNA and stimulation of platelet production in vivo. Cloning and expression of murine thrombopoietin cDNA and stimulation of platelet production in vivo. Nature 1994; 369: 565–568.
Bartley TD, Bogenberger J, Hunt P, Li YS, Lu HS, Martin F et al. Identification and cloning of a megakaryocyte growth and development factor that is a ligand for the cytokine receptor Mpl. Identification and cloning of a megakaryocyte growth and development factor that is a ligand for the cytokine receptor Mpl. Cell 1994; 77: 1117–1124.
Wendling F, Maraskovsky E, Debili N, Florindo C, Teepe M, Titeux M et al. cMpl ligand is a humoral regulator of megakaryocytopoiesis. Nature 1994; 369: 571–574.
de Sauvage FJ, Carver-Moore K, Luoh SM, Ryan A, Dowd M, Eaton DL et al. Physiological regulation of early and late stages of megakaryocytopoiesis by thrombopoietin. J Exp Med 1996; 183: 651–656.
Nagahisa H, Nagata Y, Ohnuki T, Osada M, Nagasawa T, Abe T et al. Bone marrow stromal cells produce thrombopoietin and stimulate megakaryocyte growth and maturation but suppress proplatelet formation. Blood 1996; 87: 1309–1316.
Bunting S, Widmer R, Lipari T, Rangell L, Steinmetz H, Garver-Moore K et al. Normal platelets and megakaryocytes are produced in vivo in the absence of thrombopoietin. Blood 1997; 90: 3423–3429.
Williams JL, Pipia GG, Datta NS, Long MW . Thrombopoietin requires additional megakaryocyte-active cytokines for optimal ex vivo expansion of megakaryocyte precursor cells. Blood 1998; 91: 4118–4126.
Pan RM, Yang Y, Wei MX, Yu XB, Ge YC, Xu P . A microtubule associated protein, hNUDC, binds to the extracellular domain of thrombopoietin receptor (Mpl). J Cell Biochem 2005; 96: 741–750.
Wei MX, Yang Y, Ge YC, Xu P . Functional characterization of hNUDC as a novel accumulator that specifically acts on in vitro megakaryocytopoiesis and in vivo platelet production. J Cell Biochem 2006; 98: 429–439.
Zhang YP, Tang YS, Chen XS, Xu P . Regulation of cell differentiation by hNUDC via a Mpl-dependent mechanism in NIH 3T3 cells. Exp Cell Res 2007; 313: 3210–3221.
Komatsu N, Yamamoto M, Fujita H, Miwa A, Hatake K, Endo T et al. Establishment and characterization of an erythropoietin-dependent subline, UT-7/Epo, derived from human leukemia cell line, UT-7. Blood 1993; 82: 456–464.
Takatoku M, Kametaka M, Miura Y, Komatsu N . Identification of functional domains of the human thrombopoietin receptor required for growth and differentiation of megakaryocytic cells. J Biol Chem 1997; 272: 7259–7263.
Vigon I, Mornon JP, Cocault L, Mitjavila MT, Tambourin P, Gsselbrecht S et al. Molecular cloning and characterization of MPL, the human homolog of the v-mpl oncogene: identification of a member of the hematopoietic growth factor receptor superfamily. Proc Natl Acad Sci USA 1992; 89: 5640–5644.
Melemed AS, Ryder JW, Vik TA . Activation of the mitogen-activated protein kinase pathway is involved in and sufficient for megakaryocytic differentiation of CMK cells. Blood 1997; 90: 3462–3470.
Rouyez MC, Boucheron C, Gisselbrecht S, Dusanter-Fourt I, Porteu F . Control of thrombopoietin-induced megakaryocytic differentiation by the mitogen-activated protein kinase pathway. Mol Cell Biol 1997; 17: 4991–5000.
Garcia J, de Gunzburg J, Eychène A, Gisselbrecht S, Porteu F . Thrombopoietin-mediated sustained activation of extracellular signal-regulated kinase in UT-7-Mpl cells requires both Ras-Raf-1- and Rap1-B-Raf-dependent pathways. Mole Cell Biol 2001; 21: 2659–2670.
Nagata Y, Moriguchi T, Nishida E, Todokoro K . Activation of p38 MAP kinase pathway by erythropoietin and interleukin-3. Blood 1997; 90: 929–934.
Foltz IN, Lee JC, Young PR, Schrader JW . Hemopoietic growth factors with the exception of interleukin-4 activate the p38 mitogen-activated protein kinase pathway. J Biol Chem 1997; 272: 3296–3301.
Nagata Y, Takahashi N, Davis RJ, Todokoro K . Activation of p38 MAP kinase and JNK but not ERK is required for erythropoietin-induced erythroid differentiation. Blood 1998; 92: 1859–1869.
Acknowledgements
This research was supported by China National Science Foundation Grant 30670769 and ‘863’ Grant 2007AA21009.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tang, YS., Zhang, YP. & Xu, P. hNUDC promotes the cell proliferation and differentiation in a leukemic cell line via activation of the thrombopoietin receptor (Mpl). Leukemia 22, 1018–1025 (2008). https://doi.org/10.1038/leu.2008.20
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.20
Keywords
This article is cited by
-
Establishment of a novel mesenchymal stem cell-based regimen for chronic myeloid leukemia differentiation therapy
Cell Death & Disease (2021)
-
Emerging roles of NudC family: from molecular regulation to clinical implications
Science China Life Sciences (2016)
-
The additive effects of combined murine nuclear migration protein with murine thrombopoietin in vitro and in vivo on normal and myelosuppressed mice
International Journal of Hematology (2011)
-
The mammalian NudC-like genes: a family with functions other than regulating nuclear distribution
Cellular and Molecular Life Sciences (2009)