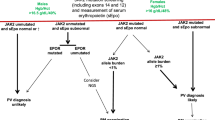
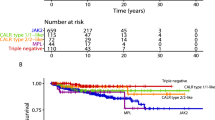
Abstract
Major causes of morbidity and mortality in polycythemia vera (PV) and essential thrombocythemia (ET) are represented by thrombosis and bleeding, progression to myelofibrosis and transformation to acute leukemia. Myelosuppressive therapy, preferentially with hydroxyurea, can reduce the rate of vascular complications, but there is some concern about an increased rate of leukemic transformation with this agent. Therefore, management of these disorders poses a significant challenge, and a risk-oriented therapeutic approach should be strictly followed to avoid inappropriate exposure to cytotoxic drugs on one side or suboptimal treatment on the other. Established risk factors for cardiovascular events are represented by older age and previous thrombosis, whereas impact of novel biological factors, including leukocytosis and JAK2V617F mutational status and/or mutational burden, is under active investigation. Low-risk PV patients should be managed only with phlebotomy and aspirin, whereas high-risk patients should also receive cytotoxic therapy. Regarding the management of ET, there is no clear indication for intervention in low-risk patients, whereas high-risk patients should be managed with chemotherapy. Other therapeutic options, such as interferon-α or anagrelide, may find place in selected patients including those who are resistant/intolerant to hydroxyurea. Finally, there is great expectation for novel drugs targeting the constitutively active JAK2/STAT pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Tefferi A, Vardiman JW . Classification and diagnosis of myeloproliferative neoplasms: the 2008 World Health Organization criteria and point-of-care diagnostic algorithms. Leukemia 2008; 22: 14–22.
Baxter EJ, Scott LM, Campbell PJ, East C, Fourouclas N, Swanton S et al. Acquired mutation of the tyrosine kinase JAK2 in human myeloproliferative disorders. Lancet 2005; 365: 1054–1061.
Levine RL, Wadleigh M, Cools J, Ebert BL, Wernig G, Huntly BJ et al. Activating mutation in the tyrosine kinase JAK2 in polycythemia vera, essential thrombocythemia, and myeloid metaplasia with myelofibrosis. Cancer Cell 2005; 7: 387–397.
James C, Ugo V, Le Couedic JP, Staerk J, Delhommeau F, Lacout C et al. A unique clonal JAK2 mutation leading to constitutive signalling causes polycythaemia vera. Nature 2005; 434: 1144–1148.
Kralovics R, Passamonti F, Buser AS, Teo SS, Tiedt R, Passweg JR et al. A gain-of-function mutation of JAK2 in myeloproliferative disorders. N Engl J Med 2005; 352: 1779–1790.
Scott LM, Tong W, Levine RL, Scott MA, Beer PA, Stratton MR et al. JAK2 exon 12 mutations in polycythemia vera and idiopathic erythrocytosis. N Engl J Med 2007; 356: 459–468.
Pardanani A, Lasho TL, Finke C, Hanson CA, Tefferi A . Prevalence and clinicopathologic correlates of JAK2 exon 12 mutations in JAK2 V617F-negative polycythemia vera. Leukemia 2007; 21: 1960–1963.
Pietra D, Li S, Brisci A, Passamonti F, Rumi E, Theocharides A et al. Somatic mutations of JAK2 exon 12 in patients with JAK2 (V617F)-negative myeloproliferative disorders. Blood 2008; 111: 1686–1689.
Pardanani AD, Levine RL, Lasho T, Pikman Y, Mesa RA, Wadleigh M et al. MPL515 mutations in myeloproliferative and other myeloid disorders: a study of 1182 patients. Blood 2006; 108: 3472–3476.
Beer PA, Campbell P, Scott LM, Bench AJ, Erber WN, Bareford D et al. MPL mutations in myeloproliferative disorders: analysis of the PT-1 cohort. Blood 2008 May 1 [Epub ahead of print].
Tefferi A, Thiele J, Orazi A, Kvasnicka HM, Barbui T, Hanson CA et al. Proposals and rationale for revision of the World Health Organization diagnostic criteria for polycythemia vera, essential thrombocythemia, and primary myelofibrosis: recommendations from an ad hoc international expert panel. Blood 2007; 110: 1092–1097.
Berk PD, Goldberg JD, Donovan PB, Fruchtman SM, Berlin NI, Wasserman LR . Therapeutic recommendations in polycythemia vera based on Polycythemia Vera Study Group protocols. Semin Hematol 1986; 23: 132–143.
Haanen C, Mathe G, Hayat M . Treatment of polycythemia vera by radiophosphorus or busulphan. A randomized clinical trial. Br J Cancer 1981; 44: 75.
Najean Y, Rain JD for the French Polycythemia Study Group Treatment of polycythemia vera: use of 32P alone or in combination with maintenance therapy using hydroxyurea in 461 patients greater than 65 years of age. Blood 1997; 89: 2319–2327.
Najean Y, Rain JD for the French Polycythemia Study Group Treatment of polycythemia vera: the use of hydroxyurea and pipobroman in 292 patients under the age of 65 years. Blood 1997; 90: 3370–3377.
Tartaglia AP, Goldberg JD, Berk PD, Wasserman LR . Adverse effects of antiaggregating platelet therapy in the treatment of polycythemia vera. Semin Hematol 1986; 23: 172–176.
Gruppo Italiano Studio Policitemia (GISP). Polycythemia vera: the natural history of 1213 patients followed over 20 years. Ann Int Med 1995; 123: 656–664.
Landolfi R, Marchioli R, Kutti J, Gisslinger H, Tognoni G, Patrono C et al. Efficacy and safety of low-dose aspirin in polycythemia vera. N Engl J Med 2004; 350: 114–124.
Cortelazzo S, Finazzi G, Ruggeri M, Vestri O, Galli M, Rodeghiero F et al. Hydroxyurea in the treatment of patients with essential thrombocythemia at high risk of thrombosis: a prospective randomized trial. N Engl J Med 1995; 332: 1132–1136.
Harrison CN, Campbell PJ, Buck G, Weathley K, East CL, Marsden JT et al. Hydroxyurea compared with anagrelide in high-risk essential thrombocythemia. N Engl J Med 2005; 353: 33–45.
Gisslinger H, Kralovics R, Gotic M, Holowiecki J, Penka M, Widmann R et al. Non-inferiority of anagrelide compared to hydroxyurea in newly diagnosed patients with essential thrombocythemia. The ANAHYDRET-Study. Blood 2007; 110: 3547 (abstr.).
Finazzi G, Barbui T . How I treat patients with polycythemia vera. Blood 2007; 109: 5104–5111.
Barbui T, Barosi G, Grossi A, Gugliotta L, Liberato LN, Marchetti M et al. Practice guidelines for the therapy of essential thrombocythemia. A statement from the Italian Society of Hematology, the Italian Society of Experimental Hematology and the Italian Group for Bone Marrow Transplantation. Haematologica 2004; 89: 215–232.
Marchioli R, Finazzi G, Landolfi R, Kutti J, Gisslinger H, Patrono C et al. Vascular and neoplastic risk in a large cohort of patients with polycythemia vera. J Clin Oncol 2005; 23: 2224–2232.
Cortelazzo S, Viero P, Finazzi G, D'Emilio A, Rodeghiero F, Barbui T . Incidence and risk factors for thrombotic complications in a historical cohort of 100 patients with essential thrombocythemia. J Clin Oncol 1990; 8: 556–562.
Besses C, Cervantes F, Pereira A, Florensa L, Solé F, Herdandez-Boluda JC et al. Major vascular complications in essential thrombocythemia: a study of the predictive factors in a series of 148 patients. Leukemia 1999; 13: 150–154.
Chim CS, Kwong YL, Lie AKW, Ma SK, Chan CC, Wong LG et al. Long-term outcome of 231 patients with essential thrombocythemia. Arch Intern Med 2005; 165: 2651–2658.
Jantunen R, Juvonen E, Ikkala E, Oksanen K, Anttila P, Ruutu T . The predictive value of vascular risk factors and gender for the development of thrombotic complications in essential thrombocythemia. Ann Hematol 2001; 80: 74–78.
Bazzan M, Tamponi G, Schinco P, Vaccarino A, Foli C, Gallone G et al. Thrombosis-free survival and life expectancy in 187 consecutive patients with essential thrombocythemia. Ann Hematol 1999; 78: 539–543.
Wolanskyj AP, Schwager SM, McClure RF, Larson DR, Tefferi A . Essential thrombocythemia beyond the first decade: life expectancy, long-term complication rates, and prognostic factors. Mayo Clin Proc 2006; 81: 159–166.
Carobbio A, Finazzi G, Guerini V, Spinelli O, Delaini F, Guerini V et al. Leukocytosis is a risk factor for thrombosis in essential thrombocythemia: interaction with treatment, standard risk factors and JAK2 mutation status. Blood 2007; 109: 2310–2313.
Alvarez-Larran A, Cervantes F, Bellosillo B, Giralt M, Julia A, Hernandez-Boluda JC et al. Essential thrombocythemia in young individuals: frequency and risk factors for vascular events and evolution to myelofibrosis in 126 patients. Leukemia 2007; 21: 1218–1223.
Radaelli F, Colombi M, Calori R, Zilioli VR, Bramanti S, Iurlo A et al. Analysis of risk factors predicting thrombotic and/or haemorrhagic complications in 306 patients with essential thrombocythemia. Hematol Oncol 2007; 25: 115–120.
Tefferi A, Gangat N, Wolanskyi A . The interaction between leukocytosis and other risk factors for thrombosis in essential thrombocythemia. Blood 2007; 109: 4105.
VanGenderen PJJ, Michiels JJ . Erythromelalgic, thrombotic and hemorrhagic manifestations of thrombocythemia. Presse Med 1994; 23: 73–77.
Fenaux P, Simon M, Caulier MT, Lai JL, Goudemand J, Bauters F . Clinical course of essential thrombocythemia in 147 cases. Cancer 1990; 66: 549–556.
Budde U, van Genderen PJ . Acquired vonWillebrand disease in patients with high platelet counts. Semin Thromb Hemost 1997; 23: 425–431.
Tefferi A, Gangat N, Wolanskyi AP . Management of extreme thrombocytosis in otherwise low-risk essential thrombocythemia: does number matter? Blood 2006; 108: 2493–2494.
Landolfi R, Di Gennaro L, Barbui T, De Stefano V, Finazzi G, Marfisi R et al. Leukocytosis as a major thrombotic risk factor in patients with polycythemia vera. Blood 2007; 109: 2446–2452.
Gangat N, Strand J, Li CY, Wu W, Pardanani A, Tefferi A . Leucocytosis in polycythemia vera predicts both inferior survival and leukaemic transformation. Br J Haematol 2007; 138: 354–358.
Gangat N, Wolanskyj AP, McClure RF, Li CY, Schwager S, Wu W et al. Risk stratification for survival and leukemic transformation in essential thrombocythemia: a single institutional study of 605 patients. Leukemia 2007; 21: 270–276.
Carobbio A, Antonioli E, Guglielmelli P, Vannucchi A, Delaini F, Guerini V et al. Leukocytosis and risk stratification assessment in essential thrombocythemia. J Clin Oncol 2008; 26: 2732–2736.
Arellano-Rodrigo E, Alvarez-Larran A, Reverter JC, Villamor N, Colomer D, Cervantes F . Increased platelet and leukocyte activation as contributing mechanisms for thrombosis in essential thrombocythemia and correlation with the JAK2 mutational status. Haematologica 2006; 91: 169–175.
Falanga A, Marchetti M, Vignoli A, Balducci D, Russo L, Guerini V et al. V617F JAK2 mutation in patients with essential thrombocythemia: relation to platelet, granulocyte, and plasma hemostatic and inflammatory molecules. Exp Hematol 2007; 35: 702–711.
Passamonti F, Rumi E, Pietra D, Della Porta MG, Boveri E, Pascutto C et al. Relation between JAK2 (V617F) mutation status, granulocyte activation, and constitutive mobilization of CD34+ cells into peripheral blood in myeloproliferative disorders. Blood 2006; 107: 3676–3682.
Vannucchi AM, Antonioli E, Guglielmelli P, Longo G, Pancrazzi A, Ponziani V et al. Prospective identification of high-risk polycythemia vera patients based on JAK2(V617F) allele burden. Leukemia 2007; 21: 1952–1959.
Silver RT, Vandris K, Wang LY, Christos PJ, D'Adriano F, Jones AV et al. JAK2V617F mutational load in patients with polycythemia vera measured by peripheral blood DNA is associated with disease severity. Blood 2007; 110: 2530A.
Tefferi A, Strand JJ, Lasho TL, Knudson RA, Finke CM, Gangat N et al. Bone marrow JAK2V617F allele burden and clinical correlates in polycythemia vera. Leukemia 2007; 21: 2074–2075.
Campbell PJ, Scott LM, Buck G, Wheatley K, East CL, Marsden JT et al. Definition of subtypes of essential thrombocythaemia and relation to polycythemia vera based on JAK2 V617F mutation status: a prospective study. Lancet 2005; 366: 1945–1953.
Cheung B, Radia D, Pantedelis P, Yadergarfar G, Harrison C . The presence of the JAK2 V617F mutation is associated with a higher haemoglobin and increased risk of thrombosis in essential thrombocythemia. Br J Haematol 2005; 132: 244–250.
Finazzi G, Rambaldi A, Guerini V, Carobbio A, Barbui T . Risk of thrombosis in patients with essential thrombocythemia and polycythemia vera according to JAK2 V617F mutation. Haematologica 2007; 92: 135–136.
Antonioli E, Guglielmelli P, Pancrazzi A, Bogani C, Verrucci M, Ponziani V et al. Clinical implications of the JAK2 V617F mutation in essential thrombocythemia. Leukemia 2005; 19: 1847–1849.
Wolanskyj AP, Lasho TL, Schwager SM, McClure RF, Wadleigh M, Lee SJ et al. JAK2 V617F mutation in essential thrombocythemia: clinical associations and long-term prognostic relevance. Br J Haematol 2005; 131: 208–213.
Kittur J, Knudson RA, Lasho TL, Finke CM, Gangat N, Wolanskyj AP et al. Clinical correlates of JAK2V617F allele burden in essential thrombocythemia. Cancer 2007; 109: 2279–2284.
Antonioli E, Guglielmelli P, Poli G, Bogani C, Pancrazzi A, Longo G et al. Influence of JAK2V617F allele burden on phenotype in essential thrombocythemia. Haematologica 2008; 93: 41–48.
Vannucchi AM, Antonioli E, Guglielmelli P, Pardanani A, Tefferi A . Clinical correlates of JAK2V617F presence or allele burden in myeloproliferative neoplasms: a critical reappraisal. Leukemia 2008 May 22 [Epub ahead of print].
Pearson TC, Wetherley-Mein G . Vascular occlusive episodes and venous haematocrit in primary proliferative polycythaemia. Lancet 1978; 2: 1219–1222.
Di Nisio M, Barbui T, Di Gennaro L, Borrelli G, Finazzi G, Landolfi R et al. The hematocrit and platelet target in polycythemia vera. Br J Haematol 2007; 136: 249–259.
Van Genderen PJJ, Mulder PGH, Waleboer M, van de Moesdijk D, Michiels JJ . Prevention and treatment of thrombotic complications in essential thrombocythemia: efficacy and safety of low-dose aspirin. Br J Haematol 1997; 97: 179–184.
Barbui T, Finazzi G . When and how to treat essential thrombocythemia. N Engl J Med 2005; 353: 85–86.
Barbui T, Finazzi G . Myeloproliferative disease in pregnancy and other management issues. Hematology Am Soc Hematol Educ Program 2006, 246–252.
Passamonti F, Randi ML, Rumi E, Pungolino E, Elena C, Pietra D et al. Increased risk of pregnancy complications in patients with essential thrombocythemia carrying the JAK2 (V617F) mutation. Blood 2007; 110: 485–489.
Fruchtman SM, Mack K, Kaplan ME, Peterson P, Berk PD, Wasserman LR . From efficacy to safety: a polycythemia vera study group report on hydroxyurea in patients with polycythemia vera. Semin Hematol 1997; 34: 17–23.
Finazzi G, Caruso V, Marchioli R, Capnist G, Chisesi T, Finelli C et al. Acute leukemia in polycythemia vera. An analysis of 1638 patients enrolled in a prospective observational study. Blood 2005; 105: 2664–2670.
Passamonti F, Rumi E, Pungolino E, Malabarba L, Bertazzoni P, Valentini M et al. Life expectancy and prognostic factors for survival in patients with polycythemia vera and essential thrombocythemia. Am J Med 2004; 117: 755–761.
Sterkers Y, Preudhomme C, Lai J-L, Demory JL, Caulier MT, Wattel E et al. Acute myeloid leukemia and myelodyslastic syndromes following essential thrombocythemia treated with hydroxyurea: high proportion of cases with 17p deletion. Blood 1998; 91: 616–622.
Finazzi G, Ruggeri M, Rodeghiero F, Barbui T . Second malignancies in patients with essential thrombocythemia treated with busulphan and hydroxyurea: long-term follow-up of a randomized clinical trial. Br J Haematol 2000; 110: 577–583.
Finazzi G, Ruggeri M, Rodeghiero F, Barbui T . Efficacy and safety of long-term use of hydroxyurea in young patients with essential thrombocythemia and a high risk of thrombosis. Blood 2003; 101: 3749.
Martyre MC . Critical review of pathogenetic mechanisms in myelofibrosis with myeloid metaplasia. Curr Hematol Rep 2003; 2: 257–263.
Lengfelder E, Berger U, Hehlmann R . Interferon alpha in the treatment of polycythemia vera. Ann Hematol 2000; 79: 103–109.
Silver RT . Long-term effects of the treatment of polycythemia vera with recombinant interferon-alpha. Cancer 2006; 107: 451–458.
Lengfelder E, Griesshammer M, Hehlmann R . Interferon-alpha in the treatment of essential thrombocythemia. Leuk Lymphoma 1996; 22 (Suppl 1): 135–142.
Kiladjian JJ, Cassinat B, Turlure P, Cambier N, Roussel M, Bellucci S et al. High molecular response rate of polycythemia vera patients treated with pegylated interferon alpha-2a. Blood 2006; 108: 2037–2040.
Samuelsson J, Mutschler M, Birgegard G, Gram-Hansen P, Bjorkholm M, Pahl HL et al. Limited effects on JAK2 mutational status after pegylated interferon α-2b therapy in polycythemia vera and essential thrombocythemia. Haematologica 2006; 91: 1281–1282.
Fruchtman SM, Petitt RM, Gilbert HS, Fiddler G, Lyne A . Anagrelide: analysis of long term efficacy, safety and leukemogenic potential in myeloproliferative diseases. Leuk Res 2005; 5: 481–491.
Barosi G, Besses C, Birgegard G, Briere J, Cervantes F, Finazzi G et al. A unified definition of clinical resistance/intolerance to hydroxyurea in essential thrombocythemia: results of a consensus process by an International Working Group. Leukemia 2007; 21: 277–280.
Verstovsek S, Pardanani AD, Shah NP, Sokol L, Wadleigh M, Gilliland DG et al. A phase I study of XL019, a selective JAK2 inhibitor, in patients with primary myelofibrosis and post-polycythemia vera/essential thrombocythemia myelofibrosis. Blood 2007; 110, abstr. 553.
Guerini V, Barbui V, Spinelli O, Salvi A, Dellacasa C, Carobbio A et al. The histone deacetylase inhibitor ITF2357 selectively targets cells bearing mutated JAK2V617F. Leukemia 2008; 22: 740–747.
Verstovsek S, Kantarjian H, Thomas APD, Cortes J, Mesa R, Staschen JRCM et al. INCB018424 an oral, selective JAK2 inhibitor, shows significant clinical activity in a phase I/II study in patients with primary myelofibrosis (PMF) and post-polycythemia vera/essential thrombocythemia myelofibrosis (post PV/ET MF). Blood 2007; 110, abstr. 558.
Pardanani A, Hood J, Lasho T, Levine RL, Martin MB, Noronha G et al. TG101209, a small molecule JAK2-selective kinase inhibitor potently inhibits myeloproliferative disorder-associated JAK2 V617F and MPL515L/K mutations. Leukemia 2007; 21: 1658–1668.
Lasho T, Tefferi A, Hood JD, Verstovsek S, Gilliland DG, Pardanani A . TG101348, a JAK2-selective antagonist, inhibits primary hematopoietic cells derived from myeloproliferative disorder patients with JAK2V617F, MPL515L/K or JAK2 exon 12 mutations as well as mutation negative patients. Leukemia 2008, March 20 [Epub ahead of print].
Wernig G, Kharas MG, Okabe R, Moore SA, Leeman DS, Cullen DE et al. Efficacy of TG101348, a selective JAK2 inhibitor, in treatment of a murine model of JAK2V617F-induced polycythemia vera. Cancer Cell 2008; 13: 311–320.
Geron I, Abrahamsson AE, Barroga CF, Kavalerchik E, Gotlib J, Hood JD et al. Selective inhibition of JAK2-driven erythroid differentiation of polycythemia vera progenitors. Cancer Cell 2008; 13: 321–330.
Pardanani A . JAK2 inhibitor therapy in myeloproliferative disorders: rationale, preclinical studies and ongoing clinical trials. Leukemia 2008; 22: 23–30.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Finazzi, G., Barbui, T. Evidence and expertise in the management of polycythemia vera and essential thrombocythemia. Leukemia 22, 1494–1502 (2008). https://doi.org/10.1038/leu.2008.177
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/leu.2008.177
Keywords
This article is cited by
-
Cytoreductive treatment in real life: a chart review analysis on 1440 patients with polycythemia vera
Journal of Cancer Research and Clinical Oncology (2022)
-
A distinct molecular mutational profile and its clinical impact in essential thrombocythemia and primary myelofibrosis patients
BMC Cancer (2020)
-
Epidemiology of polycythemia vera in a Mexican population
memo - Magazine of European Medical Oncology (2020)
-
Real-world, retrospective study evaluating thromboembolic events, associated risk factors, and health-care resource utilization in Japanese patients with polycythemia vera
International Journal of Hematology (2020)
-
Cell autonomous expression of CXCL-10 in JAK2V617F-mutated MPN
Journal of Cancer Research and Clinical Oncology (2017)