Abstract
Cholangiopathies are characterized by dysregulation of the balance between biliary growth and loss. We have shown that histamine (HA) stimulates biliary growth via autocrine mechanisms. To evaluate the paracrine effects of mast cell (MC) stabilization on biliary proliferation, sham or BDL rats were treated by IP-implanted osmotic pumps filled with saline or cromolyn sodium (24 mg/kg BW/day (inhibits MC histamine release)) for 1 week. Serum, liver blocks and cholangiocytes were collected. Histidine decarboxylase (HDC) expression was measured using real-time PCR in cholangiocytes. Intrahepatic bile duct mass (IBDM) was evaluated by IHC for CK-19. MC number was determined using toluidine blue staining and correlated to IBDM. Proliferation was evaluated by PCNA expression in liver sections and purified cholangiocytes. We assessed apoptosis using real-time PCR and IHC for BAX. Expression of MC stem factor receptor, c-kit, and the proteases chymase and tryptase were measured by real-time PCR. HA levels were measured in serum by EIA. In vitro, MCs and cholangiocytes were treated with 0.1% BSA (basal) or cromolyn (25 μM) for up to 48 h prior to assessing HDC expression, HA levels and chymase and tryptase expression. Supernatants from MCs treated with or without cromolyn were added to cholangiocytes before measuring (i) proliferation by MTT assays, (ii) HDC gene expression by real-time PCR and (iii) HA release by EIA. In vivo, cromolyn treatment decreased BDL-induced: (i) IBDM, MC number, and biliary proliferation; (ii) HDC and MC marker expression; and (iii) HA levels. Cromolyn treatment increased cholangiocyte apoptosis. In vitro, cromolyn decreased HA release and chymase and tryptase expression in MCs but not in cholangiocytes. Cromolyn-treated MC supernatants decreased biliary proliferation and HA release. These studies provide evidence that MC histamine is key to biliary proliferation and may be a therapeutic target for the treatment of cholangiopathies.
Similar content being viewed by others
Main
Cholangiopathies, such as primary biliary cirrhosis (PBC) and primary sclerosing cholangitis (PSC), are characterized by chronic inflammation1 and either abnormal growth or loss of cholangiocytes, the cells lining the intra- and extra-hepatic ducts of the biliary tree.2, 3, 4 If not treated properly these features can lead to bile duct cholestasis, a known precursor of hepatic failure, fibrosis and hyperplasia, which can then transform into cholangiocarcinoma.1
Bile duct ligation (BDL), an experimental model of cholestasis, markedly alters the function of cholagiocytes, causing the proliferation of these otherwise dormant cells.5, 6 We have demonstrated that after BDL there is an increase in biliary histamine secretion and proliferation,7 suggesting an important autocrine regulation of histamine within cholangiocytes; however, the paracrine regulation (by mast cells) of biliary proliferation is yet to be determined.
Mast cells develop from the pluripotent progenitor cell lineage located in the bone marrow, circulate throughout the body as immature cells and mature once they arrive to their target tissue.8 Upon maturation, mast cells release and express a wide variety of factors, including the stem cell factor receptor, c-kit9, 10 and the proteases, chymase and tryptase.11 It has been shown that mast cells can reside in a variety of tissues and organs, including cardiac tissue,12, 13 the gastrointestinal tract14, 15 and the liver.16, 17 Hepatic mast cell number increases with the progression of liver diseases, such as cirrhosis, fibrosis, hepatitis and cholangiopathies.18, 19, 20, 21
Cromolyn sodium has been studied for its anti-asthmatic, anti-histamine and mast cell-stabilizing properties for the past 30 years.22, 23 Although the mechanism of action is not completely known, it is thought that one potential way that cromolyn sodium inhibits the release of histamine from mast cells is by blocking calcium channels located on the surface of the mast cell.24 The major aim of this work is to evaluate the potential paracrine role that mast cells and mast cell-derived histamine have on biliary proliferation.
MATERIALS AND METHODS
All reagents and kits were purchased from Sigma-Aldrich (St Louis, MO, USA) unless otherwise indicated. The antibodies used for immunohistochemistry and immunoblots were obtained from Santa Cruz Biotechnology (Santa Cruz, San Diego, CA, USA) unless specified otherwise. The commercially available kits for immunohistochemistry (IHC) were purchased from Vector Laboratories (Burlingame, CA, USA). Histamine EIA kits were purchased from Cayman Chemical (Ann Arbor, MI, USA). All primers and reagents for real-time PCR were purchased from SABiosciences (Fredrick, MD, USA).
In Vivo Models
Male Fisher 544 rats were subjected to sham or BDL25, 26 and congruently implanted with IP osmotic pumps to deliver saline or cromolyn sodium (inhibits mast cell histamine release, 24 mg/kg BW/day27) for 7 days before killing. Animals were housed at the Scott and White Hospital Animal Facility and given free access to drinking water and standard chow. All animals were kept in a temperature-controlled environment with a 12:12 light/dark cycle, and all protocols were strictly adhered to as set forth by the local IACUC committee.
From these animals, we collected serum, liver blocks (frozen and paraffin-embedded), and cholangiocytes.25, 28 Virtually pure cholangiocytes (verified by γ-glutamyltranspetidase histochemistry29) were isolated as described by us25, 26 using a monoclonal antibody (a mouse IgG2a, a gift from Dr R Faris, Brown University, Providence, RI, USA) against an unidentified antigen expressed by all rat cholangiocytes. Cell viability was evaluated using trypan blue.
Evaluation of Biliary Mass, Proliferation and Apoptosis
In sham-operated and BDL rats treated with saline or cromolyn sodium, we measured intrahepatic bile duct mass (IBDM) and cholangiocyte proliferation by (i) immunohistochemistry for CK-19 (a specific marker of cholangiocytes) and PCNA, respectively;7, 28 (ii) real-time PCR for CK-19 and PCNA expression in total liver and isolated cholangiocytes; and (iii) PCNA expression by immunoblots in isolated cholangiocytes. Apoptosis was evaluated by real-time PCR for the apoptotic markers, BAX and pro-caspase 3, and the anti-apoptotic marker, Bcl-2, in RNA obtained from isolated cholangiocytes.30
Immunohistochemistry was performed on liver sections obtained from the selected animal models. Liver sections (4–5 μm) were incubated with specific antibodies against PCNA, CK-19 or BAX (1:50 dilution) at 4 °C overnight.26 Reactions were detected with the Vector Laboratories, DAB Kit (Burlingame, CA, USA). After staining, the slides were scanned at × 20 magnification using the Leica SCN400 system (Leica Microsystems, Buffalo Grove, IL, USA), and pictures were obtained at × 20 magnification using the Leica SCN400 Image Viewer software.
Real-time PCR was performed for CK-19, PCNA, Bcl-2, BAX and pro-caspase 3 in total liver or isolated cholangiocytes from all the groups using rat primers (SABiosciences), and a ΔΔCT (delta delta threshold cycle) analysis was performed for real-time PCR as previously described.31, 32 One microgram of total cellular RNA was used for all PCR reactions. In total liver RNA, CK-19 was utilized to determine the ratio of expression per cholangiocytes; all reactions were compared with glyceraldehyde-3-phosphate dehydrogenase to ensure proper loading.33
Western blotting was performed in isolated cholangiocytes for PCNA expression with 20 μg of protein; β-actin was used as the housekeeping gene.28, 34 Band intensity was determined on the Odyssey system from LI-COR Biosciences (Lincoln, NE, USA), Model no. 9120.
Evaluation of Hepatic Mast Cell Presence and Mast Cell Markers
To quantify the number of mast cells found in the liver, toluidine blue staining was performed on liver sections obtained from the animal models.14, 35 Paraffin-embedded liver sections (4–5 μm) were fixed in 10% buffered formalin, deparaffinized and stained with toluidine blue working solution (pH∼2.3). The working solution was made from a toluidine blue stock solution containing 1 g of Toluidine blue O (Sigma-Aldrich) and 100 ml of 70% isopropanol, mixed with 1% sodium chloride. After staining with the toluidine blue working solution, the slides were rinsed with distilled water, dehydrated with 95% and then 100% EtOH and cleared with xylene before being mounted with a resinous medium. After staining, the slides were scanned at × 20 magnification using the Leica SCN400 system (Leica Microsystems, Buffalo Grove, IL, USA), and pictures were captured with the Leica SCN400 Image Viewer software. The number of hepatic mast cells was determined by manually counting the number of toluidine blue-positive cells in up to 10 random fields. Mast cell number was correlated to bile duct mass (percentage of CK-19-positive bile ducts), which was quantified using the Pearson’s correlation coefficient (r) to determine the linear relationship between hepatic mast cell number and bile duct mass. The following scale was used to report the Pearson’s correlation coefficient (r): high correlation (0.5 to 1.0), medium correlation (−0.3 to 0.5), and low correlation (−0.1 to −0.3).36
Mast cell markers were measured by real-time PCR in total liver. We evaluated the expression levels of stem cell factor receptor, c-kit and the mast cell proteases, chymase and tryptase.9, 35, 37 Real-time PCR was performed as described above.
Evaluation of histidine decarboxylase (HDC) Expression and Histamine Secretion
HDC expression was evaluated by real-time PCR in isolated cholangiocytes from all groups of animals as described above. Histamine secretion was measured in serum by commercially available histamine EIA kits (Cayman Chemical, Ann Arbor, MI, USA).7, 32
In Vitro Analysis
Cell culture
In vitro, we utilized a mast cell line derived from fetal mouse liver (Amerian Type Culture Collection (ATCC), Manassas, VA, USA) and cultured murine cholangiocytes that were SV-40 transformed.38 All cells were maintained according to previous protocols39, 40 or ATCC recommendations. Prior to performing experimental analysis, the mast cell line was characterized by evaluating the expression of typical mast cell markers (HDC, MAO-B, chymase, tryptase and c-kit9, 35, 37) by real-time PCR.
To determine whether the effects of cromolyn sodium were specific to mast cells, we treated cholangiocytes and mast cells with 0.1% BSA (basal) or cromolyn (25 μM) for up to 48 h prior to assessing HDC, chymase and tryptase expression, histamine secretion and biliary proliferation. Cholangiocyte apoptosis was measured after cromolyn sodium (25 μM) treatment by real-time PCR for BAX and pro-caspase 3. In separate experiments, supernatants from mast cells treated with or without cromolyn sodium (25 μM) were added to cholangiocyte cultures before measuring HDC expression by real-time PCR, histamine secretion by EIA and PCNA gene expression by real-time PCR.
Statistical Analysis
All data are expressed as mean±s.e.m. Differences between groups were analyzed by Student’s unpaired t-test when two groups were analyzed and ANOVA when more than two groups were analyzed, followed by an appropriate post hoc test.
RESULTS
Treatment with Cromolyn Sodium Decreases BDL-Induced IBDM and Biliary Proliferation
As we have previously shown,25 after BDL there is a marked increase in intrahepatic biliary mass and cholangiocyte proliferation compared with sham-operated rats. Following treatment with cromolyn sodium, IBDM was significantly decreased as shown by immunohistochemistry (quantified in the bottom left panel) and real-time PCR for CK-19 (Figure 1). Similarly, treatment with cromolyn sodium for 1 week significantly decreased cholangiocyte proliferation when compared with BDL alone as shown by the decrease in (i) the number of PCNA-positive cholangiocytes; (ii) gene expression shown by real-time PCR; and (iii) protein expression shown by immunoblotting analysis (Figure 2). Intracellular cAMP signaling is a functional marker of cholangiocyte proliferation;41, 42 we found that, in cholangiocytes isolated from cromolyn sodium-treated BDL rats, basal cAMP levels were significantly decreased versus BDL cholangiocytes (0.398±0.042 pmol/1 × 106 cells (BDL) vs 0.216±0.029 pmol/1 × 106 cells (BDL+cromolyn)). In addition, we evaluated the effects of cromolyn sodium on Ca2+ signaling by measuring intracellular IP3 levels and found that cromolyn sodium treatment decreased IP3 levels compared with BDL (50.37±12.112 nmol/1 × 106 cells (BDL) vs 28.11±6.933 nmol/ 1 × 106 cells (BDL+cromolyn)), although this was not a significant change.
Evaluation of intrahepatic biliary mass. IBDM was evaluated in liver sections from all groups and by real-time PCR in isolated cholangiocytes. Following BDL, there was a significant increase in IBDM and CK-19 gene expression that is decreased in BDL+cromolyn-treated rats. Original magnification × 20. Data are mean±s.e. of six experiments. *P<0.05 versus sham-operated rat.
Determination of biliary proliferation. PCNA expression was measured by immunohistochemistry (a), real-time PCR (b) and western blots (c). (a) The number of PCNA-positive cholangiocytes was increased in BDL rats compared with sham-operated but was decreased in BDL+cromolyn-treated rats. Original magnification × 20. (b) By real-time PCR, gene expression of PCNA was significantly increased in BDL cholangiocytes compared with normal but was drastically decreased in BDL+cromolyn-treated cholangiocytes. (c) Immunoblots for PCNA show that PCNA protein expression was significantly increased in BDL cholangiocytes but reduced in BDL+cromolyn-treated cholangiocytes. Data are mean±s.e. of 6 experiments for real-time PCR and 12 experiments for immunoblotting analysis. *P<0.05 versus sham-operated rat; #P<0.05 versus BDL.
Cromolyn sodium treatment increased biliary BDL-induced apoptosis as shown by real-time PCR and BAX IHC in Figure 3. After BDL, BAX and pro-caspase 3 expression significantly increased compared with sham-operated rat cholangiocytes; these parameters were further increased in cholangiocytes from BDL+cromolyn-treated rats as shown by real-time PCR (Figures 3a and b). Conversely, Bcl-2 expression in cholangiocytes was unchanged after BDL when compared with sham-operated rats but was significantly decreased in cholangiocytes collected from BDL+cromolyn-treated rats as shown by real-time PCR (Figure 3c). IHC for BAX reveals that the expression of BAX is increased in BDL liver sections primarily in cholangiocytes, and this was further enhanced in the sections from BDL+cromolyn-treated rats (Figure 3d).
Evaluation of apoptosis. Biliary apoptosis was measured by real-time PCR for BAX, pro-caspase 3 and Bcl-2 and IHC for BAX. After BDL, cholangiocyte apoptosis increased compared with sham-operated and in cholangiocytes from BDL+cromolyn; the expression of (a) BAX and (b) pro-caspase 3 was significantly greater than sham-operated and BDL. After BDL, cholangiocyte expression of (c) Bcl-2 was unchanged compared with sham-operated, but cholangiocytes from BDL+cromolyn had significantly decreased expression when compared with sham-operated and BDL. (d) BAX staining confirmed that apoptosis was significantly increased in BDL+cromolyn liver compared with BDL alone. Data are mean±s.e. of nine experiments. *P<0.05 versus sham-operated rat; #P<0.05 versus BDL.
Increased Hepatic Mast Cell Number Correlates with Increased Bile Duct Mass and Cromolyn Sodium Decreases Hepatic Mast Cell Number
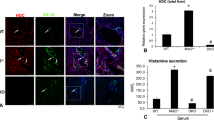
By toluidine blue staining, the number of infiltrating hepatic mast cells increased after BDL by almost 10-fold when compared with sham-operated rat liver (Figure 4a). Hepatic mast cells (red arrows) were found in close proximity to bile ducts (black arrows), and following treatment with cromolyn sodium, there was a marked decrease in the number of hepatic mast cells (Figure 4b). Using Pearson’s coefficient, we found that increased mast cell number positively correlates with increased bile duct mass (R=0.950) in BDL rats (Figure 4c).
Evaluation of hepatic mast cell infiltration and correlation with IBDM. By toluidine blue staining, the number of mast cells significantly increased in BDL compared with sham-operated but were reduced in BDL+cromolyn (a and b). Mast cells (marked by red arrows) were found in close proximity to bile ducts (black arrows), and the increase in mast cells positively correlated (R=0.950) to the increase in IBDM (c). Data are mean±s.e. of six experiments. *P<0.05 versus sham-operated rat; #P<0.05 versus BDL.
Inhibition of Mast Cell Histamine by Cromolyn Sodium Decreases the Expression of Hepatic c-Kit, Chymase and Tryptase
Using real-time PCR in total liver RNA, we found that the expression of c-kit, chymase and tryptase are all increased following BDL for 1 week (Figure 5). These data are consistent with the increase of hepatic mast cell infiltration as seen in Figure 4. In BDL rats treated with cromolyn sodium, there was a marked decrease in c-kit, chymase and tryptase expression (Figure 5), which also correlates with the decrease in hepatic mast cell number.
Determination of mast cell markers. In the total liver, the expression of c-kit, chymase and tryptase was determined by real-time PCR. Following BDL, c-kit, chymase and tryptase expression significantly increased compared with sham-operated, whereas treatment with cromolyn significantly downregulated the expression of all three compared with both sham-operated and BDL. Data are mean±s.e. of 12 experiments. *P<0.05 versus sham-operated rat; #P<0.05 versus BDL.
Treatment with Cromolyn Sodium Decreases Biliary HDC Expression and Histamine Secretion
Similar to our previously published data in a mouse model,7 we found that the expression of HDC was significantly increased in BDL rat cholangiocytes compared with sham-operated rats (Figure 6a). HDC expression was markedly decreased in cholangiocytes from BDL+cromolyn treatment compared with sham-operated and BDL cholangiocytes, suggesting that mast cell histamine influences biliary HDC levels via a paracrine interaction (Figure 6a). Histamine secretion was measured in serum from normal, BDL and BDL+cromolyn-treated rats. We found (similar to our previous studies in mice7) that circulating histamine levels are increased in BDL rats compared with sham-operated rats, whereas treatment with cromolyn sodium significantly reduces the amount of circulating histamine compared with both sham-operated and BDL rat serum (Figure 6b).
Evaluation of HDC expression and histamine secretion. (a) By real-time PCR, HDC expression is significantly increased in BDL cholangiocytes compared with sham-operated cholangiocytes, and in BDL+cromolyn, there is a significant decrease in the expression of HDC. (b) Circulating levels of histamine measured by EIA were increased in serum from BDL rats compared with sham-operated, and treatment with cromolyn significantly decreased histamine secretion compared with both sham-operated and BDL. Data are mean±s.e. of six experiments for real-time PCR and three experiments for histamine EIA. *P<0.05 versus sham-operated rat; #P<0.05 versus BDL.
Treatment with Cromolyn Sodium In Vitro Decreases Mast Cell (i) HDC Expression, (ii) Histamine Release and (iii) Chymase and Tryptase Expression
Prior to performing in vitro studies, we characterized our cultured mast cell line by measuring typical markers of mast cells by real-time PCR, including: HDC, MAO-B (enzyme responsible for the breakdown of histamine43), chymase, tryptase, and c-kit. We also evaluated the expression of CK-19 (the cholangiocyte-specific marker25, 28) by real-time PCR. Our cultured mast cell line expressed high levels of HDC, MAO-B, chymase, tryptase and c-kit but had virtually no expression of CK-19 (Figure 7). We compared the results from the mast cells with our cultured mouse cholangiocytes that we verified expressed increased levels of CK-19.
Characterization of mast cell line. By real-time PCR, the expression of HDC, MAO-B, chymase, tryptase and c-kit was increased in cultured mast cells compared with cultured cholangiocytes. Mast cells were void of CK-19 expression that was increased in cultured cholangiocytes. Data are mean±s.e. of six experiments. Data are expressed as fold over basal (set to 1).
To determine that cromolyn sodium treatment affects mast cells specifically, we treated mast cell lines with cromolyn (25 μM for up to 48 h) and measured HDC expression, histamine release and chymase and tryptase expression. As shown in Figure 8, in mast cells treated with cromolyn sodium there was a significant reduction of HDC expression (Figure 8a), histamine secretion (Figure 8b) and chymase and tryptase expression (Figures 8c and d).
Evaluation of cromolyn sodium treatment on mast cells in vitro. Mast cells were treated with cromolyn sodium (25 μM) for up to 48 h. (a) HDC expression was measured by real-time PCR. After treatment with cromolyn sodium, HDC expression significantly decreased at 24 and 48 h compared with basal-treated. (b) Histamine secretion was measured by EIA, and after cromolyn sodium treatment, histamine secretion was significantly blocked compared with basal at 24 and 48 h. (c and d) The expression of chymase and tryptase was measured by real-time PCR, and we found that cromolyn sodium significantly decreased the expression of these mast cell proteases at 24 and 48 h. Data are mean±s.e. of six experiments for real-time PCR and three experiments for histamine EIA. *P<0.05 versus basal (0.1% BSA) treatment.
To further determine whether cromolyn sodium directly affects cholangiocytes, we measured HDC, chymase and tryptase expression by real-time PCR, histamine secretion by EIA and proliferation by MTT assay in cholangiocytes treated with or without cromolyn sodium (25 μM). Further, we measured markers of apoptosis, including BAX and pro-caspase 3, by real-time PCR in cholangiocytes treated with or without cromolyn sodium (25 μM). We found that treatment with cromolyn sodium (24 and 48 h) did not significantly alter cholangiocyte: HDC, chymase and tryptase expression (Supplementary Figure S1A and B), histamine secretion (Supplementary Figure S1C), and proliferation (Supplementary Figure S1D). Cromolyn sodium did not induce apoptosis in cultured cholangiocytes as shown by the results for pro-caspase 3 and BAX at both 24 and 48 h (Supplementary Figure S2). These data demonstrate that cromolyn sodium acts primarily on mast cells.
Inhibition of Mast Cell Histamine Decreases Biliary (i) HDC Expression, (ii) Histamine Secretion and (iii) Proliferation In Vitro
In our final experiments, we determined the effects of mast cell histamine on cholangiocyte proliferation in vitro. After treating mast cell lines with cromolyn sodium, we collected the culture medium and stimulated our cholangiocyte cell line and measured HDC expression, histamine secretion and proliferation. In Figure 9, when cholangiocytes were treated with basal-treated mast cell supernatants there was a significant increase in biliary (i) HDC expression shown by real-time PCR (Figure 9a), (ii) histamine secretion (primarily released by mast cells) by EIA (Figure 9b) and (iii) proliferation as shown by PCNA gene expression (Figure 9c). In contrast, when mast cells were pretreated with cromolyn sodium (24 and 48 h), these parameters were all downregulated compared with cholangiocytes treated with mast cell basal-treated supernatants.
Determination of the effects of mast cell histamine on cholangiocytes in vitro. Cholangiocytes were treated with supernatants from mast cells treated in the absence or presence of cromolyn sodium (25 μM, 24 and 48 h), and HDC expression was measured by real-time PCR, histamine secretion by EIA and proliferation by real-time PCR for PCNA. (a) In cholangiocytes treated with mast cell basal-treated supernatants, there was an increase in HDC expression, whereas treatment with cromolyn-treated mast cell supernatants decreased biliary HDC expression at both 24 and 48 h. (b) Biliary histamine secretion was significantly increased after treatment with mast cell basal-treated supernatants compared with basal-treated cholangiocytes, which was significantly reduced in cholangiocytes treated with mast cell supernatants previously treated with cromolyn. (c) PCNA expression was significantly increased after treatment with mast cell basal-treated supernatants compared with basal-treated cholangiocytes, which was significantly reduced in cholangiocytes treated with mast cell supernatants treated with cromolyn. Data are mean±s.e. of nine experiments for real-time PCR and six experiments for histamine EIA. *P<0.05 versus basal (0.1% BSA) treated cholangiocytes. #P<0.05 versus cholangiocytes stimulated with basal-treated mast cell supernatants.
DISCUSSION
Our study demonstrates that: (i) mast cells infiltrate the liver during BDL-induced liver damage; and (ii) the number of mast cells positively correlates with increased IBDM. Inhibition of mast cell-derived histamine (via cromolyn sodium treatment) decreases IBDM and biliary proliferation supporting the paracrine role that mast cells have on biliary disease. In total liver, the expression of mast cell markers was downregulated after treatment with cromolyn sodium. The fact that biliary HDC and histamine secretion was diminished supports the notion that mast cell histamine may aid in regulating biliary function. In vitro, we demonstrated that cromolyn sodium acts on mast cells directly but not on cholangiocytes. These studies provide novel evidence to support the paracrine function that mast cells and mast cell-derived histamine have during cholangiopathies and add to our previous findings of histamine-mediated autocrine regulation of biliary proliferation.
Autocrine/paracrine regulation of biliary proliferation is highly complex and involves numerous factors, including vascular endothelial growth factor (VEGF), serotonin, secretin and histamine.26, 34, 44, 45 After BDL, the biliary epithelium proliferates and cholangiocytes secrete abundant amounts of these neuroendocrine factors to support the increased demands of the proliferating biliary tree.26, 34, 44 We have previously shown that (i) histamine treatment increases normal rat cholangiocyte proliferation;28 and (ii) after BDL, histamine levels are increased in both serum and cholangiocyte supernatant.7 Autocrine regulation by histamine has been demonstrated in both biliary hyperplasia and neoplasia, as evidenced by our finding that inhibition of HDC decreases biliary growth.7, 32 Our current studies demonstrate for the first time that a paracrine mechanism (modulated by factors released by mast cells) that regulates biliary proliferation also exists. Few studies show the anti-proliferative effects of cromolyn sodium; however, in pancreatic cancer a PEGylated liposomal formulation of cromolyn was found to decrease proliferation both in vitro and in vivo.46 Another study has shown that: (i) mast cells are present in human thyroid cancer; and (ii) treatment with cromolyn sodium decreased thyroid carcinoma growth and invasion potential, suggesting that inhibition of mast cell histamine blocks tumor growth.47 Herein, we found that treatment with cromolyn sodium decreased IBDM, proliferation and intracellular cAMP levels (but not significantly altering IP3 levels, suggesting that cromolyn sodium may regulate its effects via cAMP-dependent signaling specifically targeting large cholangiocytes, which are the primary cells that proliferate following BDL4), thereby increasing cholangiocyte apoptosis in vivo. These findings are likely linked directly to the effects of cromolyn sodium on mast cells as our study revealed that, in vitro, cromolyn sodium had no direct effect on cholangiocyte proliferation or apoptosis.
Besides their interaction with cholangiocytes, mast cells are likely to influence other cell types in the liver, including hepatic stellate cells (HSCs) and hepatocytes, as the presence of mast cells is increased in diseases targeting these cell types.48, 49, 50 Our present study focused primarily on the paracrine influence induced by mast cells on cholangiocytes; however, in data not shown here, we found that the proliferative response of hepatocytes was decreased in BDL+cromolyn sodium-treated rats compared with BDL alone, and HSC activation is diminished after BDL+cromolyn sodium treatment, suggesting that mast cell stabilization can also influence other liver cell types in vivo.
Mast cells are found in almost all tissues in relatively low numbers.35, 51 In the liver, mast cells infiltrate after damage or during disease progression. Histological analysis has demonstrated that mast cells are present in cirrhosis, fibrosis, hepatitis and cholangiopathies.18, 49, 50 Mast cells are found in various areas of the liver depending upon the etiology. For example, and in support of our current findings, in both PSC and PBC, mast cells are found surrounding damaged bile ducts, portal triads and portal tracts.50, 52 In hepatic fibrosis and hepatitis, mast cells are located near hepatic sinusoids and HSCs.49 We found that mast cells were primarily adjacent to bile ducts, and as IBDM increased, hepatic mast cell numbers increased as shown by the positive correlation. In our study, we found that treatment with cromolyn sodium decreased mast cell number, which was likely a consequence of the decrease in IBDM.
Circulating histamine levels are increased in patients with PBC and PSC53 and in the cholestatic BDL model;7 however, the source of increased histamine is not known. We have previously shown that cholangiocytes synthesize and secrete histamine in response to damage such as BDL, and our current study reveals that, in animals treated with cromolyn sodium, there is a decreased level of circulating histamine indicating that mast cells also have a role in the production of histamine during liver BDL-induced cholestasis. To support our findings, it has been demonstrated that cromolyn sodium treatment inhibits plasma histamine release in a model of airway inflammation and thrombosis.54 Our in vitro studies demonstrated that treatment with cromolyn sodium did not affect cholangiocyte histamine secretion, suggesting that the in vivo effects of histamine secretion are directly related to the inhibition of mast cell-derived histamine.
Mast cells express c-kit and the proteases, chymase and tryptase.9, 37 In total liver, after BDL there was an increase in these markers, which correlates with the increase in the number of mast cells in the liver. In rats treated with cromolyn sodium, the expression of these markers was significantly downregulated. In a model of intestinal-reperfusion, cromolyn sodium treatment decreased mast cell count and tryptase expression in intestinal tissues,55 which further supports our findings. Although the exact mechanisms by which cromolyn sodium blocks histamine release are currently unknown, we speculate that one potential mechanism is by the direct inhibition of histamine synthesis. To support this, we found that, in our cultured mast cells, cromolyn sodium decreased HDC levels, which would suggest that cromolyn sodium directly interacts with and alters histamine synthesis; however, it is unclear via which mechanism this occurs. Further, we found in vitro that cromolyn sodium had no significant impact on cholangiocyte proliferation, HDC expression or histamine secretion indicating that the effects of cromolyn sodium might be attributed solely to mast cells.
Besides histamine, mast cells release numerous mediators that may influence biliary proliferation, including VEGF and serotonin.26, 44, 45 These factors are known to regulate biliary proliferation by autocrine/paracrine mechanisms; however, our studies directly implicate histamine as a key contributor to biliary growth as cromolyn sodium is well known to inhibit histamine but not other mediators.22 The actions of cromolyn sodium are not completely known, but the effect on blocking histamine release has been widely studied in allergy-mediated reactions or diseases.56, 57 Further, in data not shown here, we found that cromolyn sodium treatment in vivo decreased biliary VEGF levels; however, when this assay was performed in vitro on cholangiocytes treated with cromolyn sodium, there was no change in biliary VEGF expression suggesting that cromolyn sodium may not interact with biliary function directly but via a paracrine interaction induced by mast cells. The biliary changes in VEGF levels in vivo are likely linked to the overall decrease in IBDM, reduced mast cell number and decreased cholangiocyte proliferation. Further studies are needed to determine the full effects of cromolyn sodium on biliary cells and other cell types within the liver parenchyma.
Our current study demonstrates that stabilization of mast cells reduces histamine release, which, in turn, decreases biliary proliferation; however, mast cells release other inflammatory mediators that can impact biliary proliferation.9, 14 Preliminary data from our lab demonstrates that treatment with cromolyn sodium in vivo also decreases the biliary expression of inflammatory cytokines such as interleukin-6 (IL-6) and IL-10 suggesting an important role for mast cells and mast cell-derived histamine during cholangiopathic-driven inflammation. The decrease in IL-6/IL-10 is likely mediated by decreased mast cell histamine release as cromolyn sodium is a specific blocker of histamine and not other mediators that are released from mast cells. Further studies are warranted to fully understand the effects of mast cell stabilization during biliary diseases.
Clinically, inhibition of mast cell histamine may not only be a therapeutic target for cholangiopathies but may also alleviate symptoms that are typically associated with liver disease, including pruritus.58 In support of this, it has been shown that in nephritic diseases exposure to cromolyn sodium decreases the incidences of pruritus.59 Further, it has been shown that bile acids (that increase in content during liver disease) can regulate mast cell histamine release60 and may also be a source of increased pruritus.61, 62 This suggests that manipulation of mast cell histamine by cromolyn sodium or bile acid therapy might also be an option for patients suffering from cholangiopathies or other liver diseases.
We have demonstrated the novel finding that mast cell-derived histamine induces a paracrine effect on biliary proliferation. Our studies reveal that (i) mast cells are present in the liver of BDL-induced cholestasis; (ii) inhibition of mast cell degranulation and histamine release decreases biliary proliferation and (iii) in vitro, cromolyn sodium has no direct effect on cholangiocytes (Figure 10). Therefore we conclude that histamine-induced biliary proliferation is regulated by both autocrine (by cholangiocytes) and paracrine (by mast cells) mechanisms.
References
Park SM . The crucial role of cholangiocytes in cholangiopathies. Gut liver 2012;6:295–304.
Alpini G, Glaser S, Alvaro D et al. Bile acid depletion and repletion regulate cholangiocyte growth and secretion by a phosphatidylinositol 3-kinase-dependent pathway in rats. Gastroenterology 2002;123:1226–1237.
Alpini G, Roberts S, Kuntz SM et al. Morphological, molecular, and functional heterogeneity of cholangiocytes from normal rat liver. Gastroenterology 1996;110:1636–1643.
Alpini G, Ulrich C, Roberts S et al. Molecular and functional heterogeneity of cholangiocytes from rat liver after bile duct ligation. Am J Physiol 1997;272 (2 Pt 1):G289–G297.
Alvaro D, Mancino MG, Glaser S et al. Proliferating cholangiocytes: a neuroendocrine compartment in the diseased liver. Gastroenterology 2007;132:415–431.
LeSage G, Glaser S, Alpini G . Regulation of cholangiocyte proliferation. Liver 2001;21:73–80.
Meng F, Onori P, Hargrove L et al. Regulation of the histamine/VEGF axis by miR-125b during cholestatic liver injury in mice. Am J Pathol 2014;184:662–673.
Ishizaka T, Mitsui H, Yanagida M et al. Development of human mast cells from their progenitors. Curr Opin Immunol 1993;5:937–943.
Galli SJ, Tsai M, Wershil BK . The c-kit receptor, stem cell factor, and mast cells. What each is teaching us about the others. Am J Pathol 1993;142:965–974.
Shiohara M, Koike K . Regulation of mast cell development. Chem Immunol Allergy 2005;87:1–21.
Jeziorska M, McCollum C, Woolley DE . Mast cell distribution, activation, and phenotype in atherosclerotic lesions of human carotid arteries. J Pathol 1997;182:115–122.
Patella V, Marino I, Lamparter B et al. Human heart mast cells. Isolation, purification, ultrastructure, and immunologic characterization. J Immunol 1995;154:2855–2865.
Sperr WR, Bankl HC, Mundigler G et al. The human cardiac mast cell: localization, isolation, phenotype, and functional characterization. Blood 1994;84:3876–3884.
Bischoff SC . Physiological and pathophysiological functions of intestinal mast cells. Semin Immunopathol 2009;31:185–205.
Traver E, Torres R, de Mora F et al. Mucosal mast cells mediate motor response induced by chronic oral exposure to ovalbumin in the rat gastrointestinal tract. Neurogastroenterol Motil 2010;22:e34–e43.
Chan A, Cooley MA, Collins AM . Mast cells in the rat liver are phenotypically heterogeneous and exhibit features of immaturity. Immunol Cell Biol 2001;79:35–40.
Irani AA, Schechter NM, Craig SS et al. Two types of human mast cells that have distinct neutral protease compositions. Proc Natl Acad Sci USA 1986;83:4464–4468.
Koda W, Harada K, Tsuneyama K et al. Evidence of the participation of peribiliary mast cells in regulation of the peribiliary vascular plexus along the intrahepatic biliary tree. Lab Invest 2000;80:1007–1017.
Matsunaga Y, Terada T . Mast cell subpopulations in chronic inflammatory hepatobiliary diseases. Liver 2000;20:152–156.
Nakamura A, Yamazaki K, Suzuki K et al. Increased portal tract infiltration of mast cells and eosinophils in primary biliary cirrhosis. Am J Gastroenterol 1997;92:2245–2249.
Stoyanova II . Relevance of mast cells and hepatic lobule innervation to liver injury. Rom J Gastroenterol 2004;13:203–209.
Herzig DJ, Kusner EJ . Effect of cromolyn sodium and deuterium oxide on anaphylactic histamine release from rat peritoneal mast cells in vitro. J Pharmacol Exp Ther 1975;194:457–462.
Murphy S, Kelly HW . Cromolyn sodium: a review of mechanisms and clinical use in asthma. Drug Intell Clin Pharm 1987;21 (1 Pt 1):22–35.
Janssen LJ, Wattie J, Betti PA . Effects of cromolyn and nedocromil on ion currents in canine tracheal smooth muscle. Eur Respir J 1998;12:50–56.
Francis H, Franchitto A, Ueno Y et al. H3 histamine receptor agonist inhibits biliary growth of BDL rats by downregulation of the cAMP-dependent PKA/ERK1/2/ELK-1 pathway. Lab Invest 2007;87:473–487.
Marzioni M, Glaser S, Francis H et al. Autocrine/paracrine regulation of the growth of the biliary tree by the neuroendocrine hormone serotonin. Gastroenterology 2005;128:121–137.
Brower GL, Chancey AL, Thanigaraj S et al. Cause and effect relationship between myocardial mast cell number and matrix metalloproteinase activity. Am J Physiol Heart Circ Physiol 2002;283:H518–H525.
Francis HL, Demorrow S, Franchitto A et al. Histamine stimulates the proliferation of small and large cholangiocytes by activation of both IP3/Ca2+ and cAMP-dependent signaling mechanisms. Lab Invest 2012;92:282–294.
Rutenburg AM, Kim H, Fischbein JW et al. Histochemical and ultrastructural demonstration of gamma-glutamyl transpeptidase activity. J Histochem Cytochem 1969;17:517–526.
Albamonte MI, Albamonte MS, Stella I et al. The infant and pubertal human ovary: Balbiani's body-associated VASA expression, immunohistochemical detection of apoptosis-related BCL2 and BAX proteins, and DNA fragmentation. Hum Reprod 2013;28:698–706.
DeMorrow S, Francis H, Gaudio E et al. Anandamide inhibits cholangiocyte hyperplastic proliferation via activation of thioredoxin 1/redox factor 1 and AP-1 activation. Am J Physiol Gastrointest Liver Physiol 2008;294:G506–G519.
Francis H, Demorrow S, Venter J et al. Inhibition of histidine decarboxylase ablates the autocrine tumorigenic effects of histamine in human cholangiocarcinoma. Gut 2012;61:753–764.
Xue JL, Cheng XW . Using host 28S ribosomal RNA as a housekeeping gene for quantitative real-time reverse transcription-PCR (qRT-PCR) in virus-infected animal cells. Curr Protoc Microbiol 2010, Chapter 1:Unit1D.2.
Glaser S, Meng F, Han Y et al. Secretin stimulates biliary cell proliferation by regulating expression of microRNA 125b and microRNA let7a in mice. Gastroenterology 2014;146:1795–808 e12.
Beaven MA . Our perception of the mast cell from Paul Ehrlich to now. Eur J Immunol 2009;39:11–25.
Islam MA, Sumon SM, Rahman MW et al. Comparison of creatinine clearance estimates with routine measured creatinine clearance in hospitalized chronic kidney disease patients. Mymensingh Med J 2014;23:213–220.
Galli SJ, Zsebo KM, Geissler EN . The kit ligand, stem cell factor. Adv Immunol 1994;55:1–96.
Ueno Y, Alpini G, Yahagi K et al. Evaluation of differential gene expression by microarray analysis in small and large cholangiocytes isolated from normal mice. Liver Int 2003;23:449–459.
Francis H, Glaser S, Demorrow S et al. Small mouse cholangiocytes proliferate in response to H1 histamine receptor stimulation by activation of the IP3/CaMK I/CREB pathway. Am J Physiol Cell Physiol 2008;295:C499–C513.
Glaser S, Wang M, Ueno Y et al. Differential transcriptional characteristics of small and large biliary epithelial cells derived from small and large bile ducts. Am J Physiol Gastrointest Liver Physiol 2010;299:G769–G777.
Francis H, Glaser S, Ueno Y et al. cAMP stimulates the secretory and proliferative capacity of the rat intrahepatic biliary epithelium through changes in the PKA/Src/MEK/ERK1/2 pathway. J Hepatol 2004;41:528–537.
Minagawa N, Nagata J, Shibao K et al. Cyclic AMP regulates bicarbonate secretion in cholangiocytes through release of ATP into bile. Gastroenterology 2007;133:1592–1602.
Rosenthaler J, Guirard BM, Chang GW et al. Purification and properties of histidine decarboxylase from Lactobacillus 30a. Proc Natl Acad Sci USA 1965;54:152–158.
Gaudio E, Barbaro B, Alvaro D et al. Administration of r-VEGF-A prevents hepatic artery ligation-induced bile duct damage in bile duct ligated rats. Am J Physiol Gastrointest Liver Physiol 2006;291:G307–G317.
Gaudio E, Barbaro B, Alvaro D et al. Vascular endothelial growth factor stimulates rat cholangiocyte proliferation via an autocrine mechanism. Gastroenterology 2006;130:1270–1282.
Kim CE, Lim SK, Kim JS . In vivo antitumor effect of cromolyn in PEGylated liposomes for pancreatic cancer. J Control Release 2012;157:190–195.
Melillo RM, Guarino V, Avilla E et al. Mast cells have a protumorigenic role in human thyroid cancer. Oncogene 2010;29:6203–6215.
Akiyoshi H, Terada T . Mast cell, myofibroblast and nerve terminal complexes in carbon tetrachloride-induced cirrhotic rat livers. J Hepatol 1998;29:112–119.
Bardadin KA, Scheuer PJ . Mast cells in acute hepatitis. J Pathol 1986;149:315–325.
Farrell DJ, Hines JE, Walls AF et al. Intrahepatic mast cells in chronic liver diseases. Hepatol 1995;22 (4 Pt 1):1175–1181.
Rao KN, Brown MA . Mast cells: multifaceted immune cells with diverse roles in health and disease. Ann NY Acad Sci 2008;1143:83–104.
Yamashiro M, Kouda W, Kono N et al. Distribution of intrahepatic mast cells in various hepatobiliary disorders. An immunohistochemical study. Virchows Arch 1998;433:471–479.
Gittlen SD, Schulman ES, Maddrey WC . Raised histamine concentrations in chronic cholestatic liver disease. Gut 1990;31:96–99.
Nemmar A, Hoet PH, Vermylen J et al. Pharmacological stabilization of mast cells abrogates late thrombotic events induced by diesel exhaust particles in hamsters. Circulation 2004;110:1670–1677.
Liu DZ, Chen ZG, Ge M et al. Effects of cromolyn sodium on intestinal ischemia-reperfusion injury by inhibiting PAR-2 expression in rats. Zhonghua Yi Xue Za Zhi 2012;92:2597–2600.
Kim HH, Park SB, Lee S et al. Inhibitory effect of putranjivain A on allergic inflammation through suppression of mast cell activation. Toxicol Appl Pharmacol 2014;274:455–461.
Volcheck GW, O'Connell EJ . Anti-inflammatory drugs for controlling asthma. Postgrad Med 1998;104:127–136 139.
Bolier R, Oude Elferink RP, Beuers U . Advances in pathogenesis and treatment of pruritus. Clin Liver Dis 2013;17:319–329.
Vessal G, Sagheb MM, Shilian S et al. Effect of oral cromolyn sodium on CKD-associated pruritus and serum tryptase level: a double-blind placebo-controlled study. Nephrol Dial Transplant 2010;25:1541–1547.
Quist RG, Ton-Nu HT, Lillienau J et al. Activation of mast cells by bile acids. Gastroenterology 1991;101:446–456.
Bunchorntavakul C, Reddy KR . Pruritus in chronic cholestatic liver disease. Clin Liver Dis 2012;16:331–346.
Schaefer B, Schaefer F, Wittmer D et al. Molecular Adsorbents Recirculating System dialysis in children with cholestatic pruritus. Pediatr Nephrol 2012;27:829–834.
Acknowledgements
This work was supported by a Career Development Award from the Veteran’s Affairs (IK2 BX001760) and funds from the Primary Sclerosing Cholangitis Foundation to Dr Francis and a VA Merit award to Dr Meng.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest. This research was supported in part by the Veterans Health Administration. The views expressed are those of the authors and do not necessarily reflect the views of the Department of Veterans Affairs.
Additional information
Supplementary Information accompanies the paper on the Laboratory Investigation website
This paper demonstrates that during biliary hyperplasia mast cells are recruited to the liver and histamine levels are significantly increased. Stabilization of mast cells with cromolyn reduces mast cell number and proliferation. Manipulation of mast cell-derived histamine may be a therapeutic target for the treatment of cholangiopathies.
Rights and permissions
About this article
Cite this article
Kennedy, L., Hargrove, L., Graf, A. et al. Inhibition of mast cell-derived histamine secretion by cromolyn sodium treatment decreases biliary hyperplasia in cholestatic rodents. Lab Invest 94, 1406–1418 (2014). https://doi.org/10.1038/labinvest.2014.129
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2014.129
This article is cited by
-
Ursodeoxycholate inhibits mast cell activation and reverses biliary injury and fibrosis in Mdr2−/− mice and human primary sclerosing cholangitis
Laboratory Investigation (2018)
-
Isolation and characterization of hepatic mast cells from cholestatic rats
Laboratory Investigation (2016)
-
Knockout of microRNA-21 reduces biliary hyperplasia and liver fibrosis in cholestatic bile duct ligated mice
Laboratory Investigation (2016)