Abstract
Type 2 diabetes is hallmarked by insulin resistance and insufficient β-cell function. Islets of type 2 diabetes patients have been shown to have decreased hypoxia-inducible factor (HIF)-1α/β expression. Target genes of the HIF pathway are involved in angiogenesis, survival, proliferation, and energy metabolism, and von Hippel-Lindau protein (VHL) is a negative regulator of this pathway. We hypothesized that increased HIF-mediated gene transcription by VHL deletion in the β-cells would increase β-cell mass and function. We generated β-cell-specific VHL-knockout mice using the Cre-loxP recombination system driven by the rat insulin promoter to assess the role of VHL in glucose homeostasis and β-cell function. VHL deletion in the pancreatic β-cells led to impaired glucose tolerance due to defects in glucose-stimulated insulin secretion and β-cell mass with age. VHL-knockout islets had decreased GLUT2, but increased glucose transporter 1 and vascular endothelial growth factor expression. Furthermore, there were significant aberrations in islet morphology in the VHL-knockout mice, likely due to increased islet vasculature. Given that erythropoietin (EPO) is a target gene of the HIF pathway, which is not expressed in islets, we tested whether activating EPO signaling by systemic administration with recombinant human EPO (rHuEPO) can overcome the β-cell defects that occurred with VHL loss. We observed improved glucose tolerance and restoration of GLUT2 expression in VHL-deficient β-cells in response to rHuEPO. Contrary to our hypothesis, loss of VHL and increased transcription of HIF-target genes resulted in impaired β-cell function and mass, which can be overcome with exogenous EPO. Our results indicate a critical role for VHL in β-cell function and mass, and that EPO administration improved β-cell function making it a potential strategy for diabetes treatment.
Similar content being viewed by others
Main
Type 2 diabetes is a disease that is now considered epidemic, as it affects over 200 million people globally, and its prevalence continues to rise despite advances in treatment.1, 2 The two hallmark characteristics of type 2 diabetes are insulin resistance and β-cell dysfunction.3, 4 Under insulin-resistant conditions, β-cells undergo a compensation process by expanding β-cell mass and enhancing β-cell function. When β-cell compensation can adequately meet the insulin demands of the body, normoglycemia is maintained.5, 6, 7 However, in susceptible individuals perhaps with genetic defects and/or environmental insults, the β-cells cannot meet the metabolic demands, ultimately resulting in type 2 diabetes.8, 9, 10, 11
A previous study by Gunton et al12 reported results of gene expression profiling analyses on islets of humans with type 2 diabetes, and found a significant decrease in aryl-hydrocarbon-receptor nuclear translocator (ARNT)/hypoxia-inducible factor-1β (HIF-1β) expression in these islets.12 Concomitant with the decrease in ARNT expression was a decrease in HIF-1α expression in the islets of deceased humans with type 2 diabetes.12
HIFs are the key mediators of cellular adaptation in response to hypoxia. Under conditions of adequate oxygen supply, von Hippel-Lindau tumor suppressor protein (VHL), which makes up a part of the E3-ubiquitin ligase complex, targets HIF-α for ubiquitination and subsequent proteasomal degradation. During hypoxia, the HIF-α subunit is stabilized and translocated to the nucleus, where it binds to the HIF-β/ARNT subunit. The HIF-α/ARNT complex then binds to DNA, resulting in increased transcription of HIF-target genes important for angiogenesis, proliferation, survival, and energy metabolism.13, 14, 15, 16 It is, therefore, plausible that activating HIF may improve cell survival and also promote beneficial adaptive changes, such as increased angiogenesis. The concept is supported by observations that exposure to hypoxia protects tissues, including the heart, brain, and kidney, from subsequent ischemic injury.17, 18, 19 We hypothesized that enhanced HIF-α stabilization and HIF-dependent pro-survival gene activation, including vascular endothelial growth factor (Vegf), glucose transporter 1 (Glut1), and erythropoietin (Epo), in the pancreatic β-cells would enhance β-cell mass and improve glucose homeostasis in mice.
MATERIALS AND METHODS
Mouse Protocol
RIPcre+ mice20 were bred to vhlhfl/fl mice21 (vhlh is the mouse homolog for VHL; Jackson Laboratories) to generate RIPcre+vhlh+/fl mice, which were then interbred to generate RIPcre−, RIPcre+vhlhfl/fl RIPcre+vhlh+/fl, and RIPcre+ vhlh+/+ mice. All mice were maintained on a mixed 129J/C57BL/6 background, and only RIPcre+vhlh+/+ littermates were used as controls. Genotypes for the cre and Vhlh genes were determined by PCR using tail DNA. Primer sequences are available on request. Mice were maintained on a 12-h light–dark cycle with free access to water and standard irradiated rodent chow (5% fat; Harlan Teklad, Indianapolis, IN, USA) and housed in pathogen-free barrier facilities at the central animal facility at the Ontario Cancer Institute (Toronto, Ontario, Canada). All animal experiments were approved by the Ontario Cancer Institute Animal Care Facility.
Metabolic Studies and Hormone Measurements
Blood glucose levels were determined from tail venous blood with an automated glucose monitor (Precision Xtra, Abbott Laboratories, Saint-Laurent, Quebec, Canada). Overnight fasting was approximately 14–16 h in duration. Glucose tolerance tests were performed on overnight-fasted animals by intraperitoneal (i.p.) injection of glucose at a dose of 1 g/kg of body weight, and blood glucose levels were measured at 0, 15, 30, 45, 60, and 120 min post-glucose challenge. Insulin tolerance tests were performed using human regular insulin (Novo Nordisk, Toronto, Ontario, Canada) at a dose of 1 U/kg of body weight, and blood glucose levels were measured at 0, 15, 30, 45, 60, and 120 min after the injection of insulin. Glucose-stimulated insulin secretion in vivo was carried out on overnight-fasted animals after an i.p. injection of glucose at a dose of 3 g/kg of body weight, with tail vein blood collected at 0, 2, 10, and 30 min after the injection for insulin measurements. Insulin levels were measured by an enzyme-linked immunosorbent assay (ELISA) kit using a rat insulin standard (Crystal Chem, Downers Grove, IL, USA). Islet insulin content was measured by ELISA after acid-ethanol extraction.
Islet Morphometry, Immunohistochemistry, and Immunofluorescent Staining
Pancreatic tissue was isolated and fixed in 4% paraformaldehyde in 0.1 M phosphate-buffered saline (pH 7.4). Samples were dehydrated and prepared as paraffin blocks. Thick sections (7 μm) were obtained at 150-μm intervals on three levels and stained for insulin, Ki67, factor VIII (DAKO), cleaved caspase-3 (Cell Signaling), GLUT2 (Millipore), and glucagon (Sigma). Immunofluorescent-stained sections were visualized using a Zeiss inverted fluorescent microscope (Advanced Optical Microscopy Facility, Toronto, Ontario, Canada). Pancreatic sections immunostained for insulin, cleaved caspase-3, and factor VIII were scanned at × 20 magnification using a ScanScope ImageScope system. The digital images were analyzed with ImageScope version 9.0.19.1516 software (Aperio Technologies, Vista, CA, USA). β-cell area per total pancreatic area was determined in insulin-immunostained pancreatic sections. Endothelial cell immunostaining in the islets with anti-factor VIII and sections immunostained for cleaved caspase-3 were calculated using an automated positive pixel count algorithm and normalized per total islet area. Ki67-positive cells were calculated as a percentage of total islet cells.
Islet Isolation
Pancreatic islets were isolated as previously described.22 Briefly, 3 ml collagenase (3 mg/ml; Sigma) was injected into the common bile duct to inflate the pancreas. The pancreatic tissue was then removed and digested in collagenase solution at 37°C with shaking for 30 min. The digestion was stopped by ice-cold Hanks’ balanced salt solution and passed through a filter. Islets were then handpicked under a dissecting microscope.
Western Blotting
Islets, fat, hypothalamus, liver, and muscle tissues were isolated, and protein lysates were obtained as previously described.23 Lysates were separated by sodium dodecyl sulfate-10% polyacrylamide gel electrophoresis, and immunoblotted with antibodies for VHL (BD Bioscience), Pdx-1, and α-tubulin (Cell Signaling). Western blot signal densities were analyzed using ImageJ software. Protein levels were normalized to α-tubulin levels and expressed in arbitrary units relative to littermate control levels.
mRNA Measurements by Reverse Transcription-PCR (RT-PCR) and Quantitative Real-Time PCR
mRNA was extracted from isolated islets by TRIzol following the manufacturer's protocol (Invitrogen, Toronto, Ontario, Canada) and treated with RNase-free DNase (Invitrogen). Semiquantitative RT-PCR amplification was performed with a one-step RT-PCR kit (Invitrogen). Epo, Vegf, and Glut1 were amplified by PCR using specific primers. Densitometric analysis was performed using ImageJ software and normalized to β-actin. Quantitative real-time PCR was performed and quantified by Applied Biosystems 7900 HT Fast Real-Time PCR system (Life Technologies Corporation, Carlsbad, CA, USA). Primer sequences are provided in Supplementary Table S1.
Recombinant Human EPO (rHuEPO) Treatment
rHuEPO (Epoetin-α or Eprex; Ortho-Biotech) was used in all experiments. rHuEPO was diluted in 0.9% NaCl, and mice were injected intraperitoneally three times per week at a dose of 50 μg per kg body weight24 between 0900 and 1000 hours for 4 weeks.
Statistical Analysis
Data are presented as means±standard errors of the mean, and were analyzed by two-tailed independent samples t-test and one-way analysis of variance with the post hoc Tukey least significant difference test, as appropriate, using the statistical software SPSS (version 17.0 for Windows). P-values of <0.05 were accepted as statistically significant.
RESULTS
Generation of RIPcre+vhlhfl/fl Mice and Efficient Deletion of vhlh in the Pancreatic β-cells
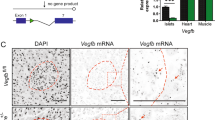
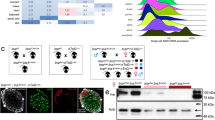
We confirmed efficient deletion of VHL in the pancreatic β-cells by western blot on isolated islets from RIPcre+vhlhfl/fl mice (Figure 1a), whereas the expression of VHL in other tissues was unaffected (Figure 1b). Body weight was significantly lower in the RIPcre+vhlhfl/fl mice compared with RIPcre+vhlh+/+ control littermates (Figure 1c). Though we did not detect a significant decrease in VHL expression in the lysates of whole hypothalami in the RIPcre+vhlhfl/fl mice, the reduced body weight is likely attributed to the VHL deletion in the small subset of insulin-transcribing neurons in the hypothalamus.25, 26 Indeed, another report, using the same genetic strategy, show that the proportionate growth defect in the RIPcre+vhlhfl/fl mice is due to reduced pituitary growth hormone production.27 To confirm that, VHL deletion resulted in enhanced HIF-mediated gene transcription, we performed RT-PCR analyses. HIF-target genes, including Glut1, Vegf, and Epo, were assessed by RT-PCR. Glut1 and Vegf expression levels were increased in the mutant mice compared with controls (Figure 2a). Epo gene expression was not present in the islets of either RIPcre+vhlhfl/fl mice or control littermates (Figure 2b). Other HIF-regulated genes were examined by quantitative real-time PCR, including Epo receptor (EpoR), glucokinase (Gck), pyruvate dehydrogenase kinase 1 (Pdk1), and lactate dehydrogenase (Ldha) in islets of RIPcre+vhlhfl/fl and wild-type mice. In keeping with previous reports,28, 29 gene expression of EpoR, Pdk1, and Ldha were upregulated, whereas Gck trended lower in islets of RIPcre+vhlhfl/fl mice compared with wild-type mice (Figure 9).
Efficient deletion of VHL in the pancreatic β-cells. (a) VHL deletion in the β-cells of the mutant mice as shown by western blot, n=3 per genotype. (b) Hypothalamus, liver, and muscle tissues were unaffected, n=3 per genotype. (c) Decreased weight in the RIPcre+vhlhfl/fl mice (white bars) compared with controls (black bars), n=15 per genotype. *P<0.05. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
VHL deletion leads to the upregulation of HIF-target genes. (a) RT-PCR analyses show increased Glut1 and Vegf expression in the islets of RIPcre+vhlhfl/fl mice (white bars) compared with controls (black bars). *P<0.05. Results represent means±s.e. (b) There was no Epo gene expression detected in the islets of both genotypes, n=3 per genotype; K, kidney. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
RIPcre+vhlhfl/fl Mice have Impaired Glucose Tolerance
To assess the effect of VHL deletion in the pancreatic β-cells on glucose homeostasis, we measured monthly random and fasting blood glucose levels from 1 to 4 months of age. RIPcre+vhlhfl/fl mice had slightly increased random (Figure 3a) and fasting (Figure 3b) blood glucose levels compared with control RIPcre+vhlh+/+ mice. We then assessed glucose tolerance in 2-month-old mice by performing glucose tolerance tests, which showed impairment in the RIPcre+vhlhfl/fl mice compared with controls (Figure 3c). Next, we investigated whether the glucose intolerance in the RIPcre+vhlhfl/fl mice was due to peripheral insulin resistance or impaired β-cell function. Insulin tolerance tests showed similar glucose-lowering effects by insulin between RIPcre+vhlhfl/fl and RIPcre+vhlh+/+ mice (Figure 3d), which suggests that the glucose intolerance in the mutant mice was not due to changes in peripheral insulin sensitivity. Rather, in vivo glucose-stimulated insulin secretion tests showed impaired β-cell function in the RIPcre+vhlhfl/fl mice compared with control mice (Figure 3e). Both the baseline fasting serum insulin levels as well as glucose responsive insulin secretion were significantly decreased in the RIPcre+vhlhfl/fl mice compared with their control counterparts, which likely explains the glucose intolerance in the VHL mutant mice.
RIPcre+vhlhfl/fl mice have impaired glucose tolerance. Slight differences were found between the mutant (white bars) and control mice (black bars) in (a) random and (b) fasting blood glucose levels, n=15 per genotype. (c) RIPcre+vhlhfl/fl mice (white squares) were severely glucose intolerant compared with littermate controls (black squares), as assessed by glucose tolerance tests, n=14 per genotype. (d) Insulin tolerance tests showed no differences between the mutant (white squares) and controls (black squares), n=10 per genotype. (e) In vivo glucose-stimulated insulin secretion tests showed defects in the mutant mice (white bars) compared with control mice (black bars) at first-phase and second-phase secretion, n=5 per genotype. All experiments were performed on 2-month-old mice. *P<0.05. **P<0.01. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
VHL Deletion in the β-Cells Leads to a Reduction in β-Cell Mass with Age
To explore the causes of the impaired β-cell function in the RIPcre+vhlhfl/fl mice, we first analyzed insulin-immunostained pancreatic sections for changes in β-cell area. In 2-month-old RIPcre+vhlhfl/fl mice, we found no differences in β-cell area per pancreatic area compared with RIPcre+vhlh+/+ mice (Figure 4a). In addition, the number of islets per pancreas area was not significantly different between the mutant and wild-type mice (Figure 4b). We assessed for changes in β-cell proliferation and apoptosis. We immunostained pancreatic sections for Ki67, a marker of proliferation, and found that there was no significant difference in Ki67-positive islet cells between the genotypes (Figure 4c). Cleaved (activated) caspase-3-immunostained sections also showed no differences in the degrees of apoptosis in the islets between RIPcre+vhlhfl/fl mice and their control counterparts (Figure 4d).
VHL deletion in the β-cells has no effects on β-cell mass. (a) Quantification analyses showed no changes in β-cell area per pancreatic area ratio in RIPcre+vhlhfl/fl mice compared with control mice at 2 months of age, n=5 per genotype. (b) Islet number per pancreatic area was unaffected, n=5 per genotype. (c) Ki67 staining showed no significant differences between the genotypes (original magnification × 40), n=3 per genotype. (d) Cleaved caspase-3 staining on pancreatic sections showing increased immunostaining in RIPcre+vhlhfl/fl mice (original magnification × 20), n=3 per genotype. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
β-Cell mass increases with aging as evidenced by the increase in β-cell area per pancreatic area in the RIPcre+vhlh+/+ mice in the older 7- to 9-month-old mice compared with their younger counterpart. This age-dependent increase in β-cell mass occurred to a lesser extent in the RIPcre+vhlhfll/fl mice, resulting in a significantly smaller β-cell area per pancreatic area in the mutant mice compared with RIPcre+vhlh+/+ mice in the older age group (Figure 5a). There was an increase in cleaved caspase-3-positive islet cells in the older RIPcre+vhlhfl/fl mice compared with their control littermates (Figure 5b). In addition, we assessed islet cell proliferation in these mice by immunostaining for Ki67 on pancreatic sections. We observed a trend toward a decrease in proliferating β-cells in the RIPcre+vhlhfll/fl mice compared with RIPcre+vhlh+/+ mice (Figure 5c). Importantly, in either the 2-month-old or the older 7- to 9-month-old RIPcre+vhlhfll/fl mice, there was no evidence of pancreatic or islet tumors.
RIPcre+vhlhfl/fl mice have decreased β-cell mass with increasing age. (a) Insulin-immunostained pancreatic sections revealed a decrease in β-cell area per pancreatic area ratio in RIPcre+vhlhfl/fl mice compared with control mice at 7–9 months of age, n=5 per genotype. (b) Cleaved caspase-3 staining on pancreatic sections showing increased immunostaining in RIPcre+vhlhfl/fl mice (original magnification × 20), n=5 per genotype. (c) Ki67 immunostaining showed a trend toward a decrease in islet proliferation in the mutant mice compared with controls, n=3 per genotype. *P<0.05. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
Loss of VHL Leads to Decreased GLUT2 Expression in the Islets
We examined whether the defects in β-cell function observed in the RIPcre+vhlhfl/fl mice were due to reduced insulin or Pdx-1 expression. Interestingly, insulin content of the islets, as assessed by ELISA, showed no differences between the two genotypes (Figure 6a). In addition, Pdx-1 protein expression was similar in the islets of RIPcre+vhlhfl/fl mice compared with control mice (Figure 6b). However, we observed a significantly reduced expression of GLUT2 at the membrane of the β-cells in the RIPcre+vhlhfl/fl mice compared with control mice, which suggests that the abnormal glucose tolerance and glucose-stimulated insulin secretion may be due to a defect in the glucose-sensing mechanisms (Figure 6c).
Loss of VHL does not lead to changes islet insulin or Pdx-1 levels, but decreases the expression of GLUT2. (a) Islet insulin content measurement of 20 size-matched isolated islets by ELISA showed no difference between the genotypes, n=3 per genotype. (b) Pdx-1 levels assessed by western blot showed no changes in protein expression between the mutant and control islets, n=3 per genotype. (c) Islet GLUT-2 expression on pancreatic sections was decreased in the mutant mice compared with controls (original magnification × 40), n=3 per genotype. Mice were analyzed at 2 months of age. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
VHL-Deficient Islets have Enhanced Angiogenesis and Disrupted Morphology
Murine islets have a characteristic architecture, in which the core of the islets consists of insulin producing β-cells, whereas other cell types, α, δ, and PP cells are localized around the islet periphery.30 Interestingly, insulin and glucagon co-immunostaining by immunofluorescence showed disrupted islet morphology with glucagon-positive α cells scattered throughout the core of the islets in RIPcre+vhlhfl/fl mice (Figure 7a). A target gene downstream of the HIF pathway is VEGF, which is important in facilitating tissue oxygenation.31 RT-PCR analyses showed increased Vegf expression in the islets of the RIPcre+vhlhfl/fl mice (Figure 2a). Indeed, when we immunostained pancreatic sections for factor VIII, a marker of endothelial cells, we found increased vasculature occupying a significant space within the islets of the RIPcre+vhlhfl/fl mice compared with RIPcre+vhlhfl/fl mice (Figure 7b).
VHL-deficient islets have enhanced angiogenesis and disrupted morphology. (a) Immunofluorescent staining for insulin and glucagon show disrupted islet morphology in the mutant mice compared with wild-type mice (original magnification × 20), n=5 per genotype. (b) Factor VIII immunostaining showed increased islet microvasculature in 2-month-old mutant mice (original magnification × 40), n=3 per genotype. *P<0.05. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
rHuEPO Administration Rescues the Glucose Intolerance and β-Cell Dysfunction in RIPcre+vhlhfl/fl Mice
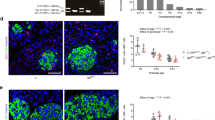
The induction of hypoxia leads to the upregulation of various hypoxia-inducible genes, including Epo, Vegf, and Glut1, which by-in-large provide pro-survival functions.13, 14, 15, 16, 31, 32 Accordingly, RT-PCR analyses on isolated islets from the RIPcre+vhlhfl/fl mice showed enhanced gene expression of Vegf and Glut1 in the RIPcre+vhlhfl/fl mice compared with controls (Figure 2a). Epo gene expression is well documented to be produced by the peritubular fibroblasts of the kidney33 and the hepatocytes.34 In addition, recent studies have shown its production in other tissues, including the brain, heart, and uterus.35 However, Epo gene expression was not present in the islets of RIPcre+vhlh+/+ mice, and furthermore, no upregulation was observed in the RIPcre+vhlhfl/fl mice (Figure 2b). Given that EPO has been shown to promote pro-survival and proliferative signaling,36 and that EPO receptor is present in pancreatic β cells,37 we tested whether providing exogenous EPO through systemic rHuEPO treatment could overcome the defects in glucose homeostasis in the RIPcre+vhlhfl/fl mice. Indeed, glucose tolerance in the RIPcre+vhlhfl/fl mice improved when treated with rHuEPO for 4 weeks compared with saline-treated RIPcre+vhlhfl/fl controls (Figure 8a). There was an increase in β-cell area per total pancreatic area and Ki67-positive islet cells in the rHuEPO-treated RIPcre+vhlh+/+ mice compared with saline-treated RIPcre+vhlh+/+ mice, whereas no significant increases in these parameters occurred in rHuEPO-treated RIPcre+vhlhfl/fl mice compared with saline-treated RIPcre+vhlhfl/fl mice (Figure 8b and c).
rHuEPO treatment improves glucose tolerance in RIPcre+vhlhfl/fl mice. (a) Glucose tolerance tests show improved glucose tolerance in the rHuEPO-treated RIPcre+vhlhfl/fl mice compared with saline-treated RIPcre+vhlhfl/fl mice, n=6 per genotype. *P<0.05; +,+/+ (saline; black squares) vs +,+/+ (rHuEPO; black circles). **P<0.05; +,−/− (saline; white squares) vs +,−/− (rHuEPO; white circles). #P<0.05; +,−/− (saline; white squares) vs +,+/+ (saline; black squares). (b) β-Cell area per pancreatic area as assessed by insulin immunostaining and (c) β-Cell proliferation as assessed by Ki67 immunostaining on pancreatic sections, n=4–6 per treatment group. *P<0.05. (d) GLUT2 immunostaining showed enhanced GLUT2 expression in the islets of rHuEPO-treated RIPcre+vhlhfl/fl mice compared with saline-treated RIPcre+vhlhfl/fl mice, n=3 per treatment group, × 20 original magnification. (e) Insulin level after 30 min of glucose challenge (3 g/kg of body weight), n=5 per treatment group. Saline-treated RIPcre+vhlhfl/fl mice decreased insulin secretion compared with saline-treated RIPcre+vhlh+/+ mice *P<0.05. rHuEPO-treated RIPcre+vhlhfl/fl mice increased insulin secretion compared with saline-treated RIPcre+vhlhfl/fl mice ##P<0.01. Treatments were started on 8-week-old mice. Mice were killed after 4 weeks on their respective treatments. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
We next assessed for the effects of rHuEPO on insulin secretion. The insulin level after glucose challenge was decreased in saline-treated RIPcre+vhlhfl/fl mice compared with saline-treated RIPcre+vhlh+/+ mice. Interestingly, improved insulin secretion was shown in rHuEPO-treated RIPcre+vhlhfl/fl mice compared with saline-treated RIPcre+vhlh+/+ mice (Figure 8e), which suggests that the improved glucose tolerance observed in rHuEPO-treated RIPcre+vhlhfl/fl mice may be due to enhanced insulin secretion rather than an increase in β-cell mass.
To assess whether the enhanced insulin secretion is because of the improvement in glucose-sensing abilities in rHuEPO-treated RIPcre+vhlhfl/fl mice, we examined their GLUT2 expression. Interestingly, GLUT2 expression was restored in the islets of rHuEPO-treated RIPcre+vhlhfl/fl mice as assessed by immunofluorescence (Figure 8d). Gene expression measurements using quantitative real-time PCR also showed increased levels of Glut2 in rHuEPO-treated RIPcre+vhlhfl/fl mice compared with saline-treated RIPcre+vhlhfl/fl mice (Figure 9). Effects of rHuEPO on other HIF-regulated genes were also measured. Interestingly, rHuEPO treatment completely reversed some transcriptional changes that were present in islets of RIPcre+vhlhfl/fl mice, such as EpoR and Pdk1. Gck on the other hand was increased in both RIPcre+vhlhfl/fl and wild-type mice after rHuEPO treatment, whereas partial normalization was achieved with Glut2 and Ldha in islets of RIPcre+vhlhfl/fl. These results show that rHuEPO may exert effects in pancreatic islet function through alterations in HIF-regulated genes in RIPcre+vhlhfl/fl mice.
rHuEPO treatment reversed gene expression changes associated with VHL deletion in pancreatic islets. Epo receptor expression level was increased in saline-treated RIPcre+vhlhfl/f mice compared with saline-treated controls (*P<0.05), whereas this was attenuated by rHuEPO-treatment (#P<0.05). With VHL deletion, Glut2 gene expression was dramatically decreased in saline-treated RIPcre+vhlhfl/f islets compared with saline-treated RIPcre+vhlh+/+ mice (***P<0.001), and this effect was partially rescued by rHuEPO (###P<0.001). Saline-treated RIPcre+vhlh+/+ mice showed decreased Gck expression levels but increased Ldha and Pdk1expression levels compared with saline-treated RIPcre+vhlh+/+ mice. n=6 per treatment group. *, Comparison with saline-treated RIPcre+vhlh+/+ mice. #, Comparison with rHuEPO-treated RIPcre+vhlhfl/f mice. *,#P<0.05, **,##P<0.01, ***,###P<0.001. Mice were killed after 1 week on their respective treatments. Results represent means±s.e. +, +/+: RIPcre+vhlh+/+; +, −/−: RIPcre+vhlhfl/fl.
DISCUSSION
The VHL tumor suppressor has an essential role in the cellular response to hypoxia.38 To gain insight into the role of VHL in β-cell homeostasis, we have specifically deleted VHL in β-cells using the Cre-loxP recombination system. The resultant RIPcre+vhlhfl/fl mice exhibited proportionate small body size that was also found by another group, which was not related to adiposity.27
VHL gene was first described as a tumor suppressor from inactivation mutations in familial studies, which showed a variety of malignant and benign tumors of the eye, brain, spinal cord, kidney, pancreas, and adrenal glands, known as VHL.39 In our studies, the β-cell-specific VHL-knockout mice, in which cre-mediated VHL deletion was under the control of the RIP, did not demonstrate any evidence of tumor development, similar to a study by Cantley et al.27 In addition, in this study, mice in which VHL was deleted under the control of the Pdx-1 promoter (Tg (Pdx1-cre) 1Herr) was used, which also demonstrated a glucose intolerant phenotype due to impaired insulin secretion. However, another study by Shen et al40 showed that mice with Pdx-1-driven VHL deletion in a different genetic background (Tg (Pdx1-cre) 89.1Dam) resulted in tumor development and early mortality.
Contrary to our initial hypothesis, in which the increased HIF-mediated gene transcription by VHL deletion in the β-cells would increase β-cell mass and function, our study demonstrates a defect in β-cell function resulting in glucose intolerance. This is in keeping with other studies, which all show a defect in β-cell function as a result of VHL loss.27, 41, 42 Importantly, however, these studies did not show a defect in β-cell mass. In contrast to these studies, we are the first to show that VHL has a critical role in β-cell growth that occurs with aging. As such, the older RIPcre+vhlhfl/fl mice (7–9 months) exhibit a compromised age-dependent growth in β-cell mass, resulting in reduced β-cell mass compared with control littermates in the older cohort. This is likely due to enhanced apoptosis as evidenced by increased cleaved caspase-3 immunostaining in the islets in the older mutant mice. These results show the importance of the HIF pathway in modulating apoptosis, which allow for the increase in the β-cell mass that occurs with aging.43, 44
Recent published studies indicate that both of oxygen tension and hypoxia are critical regulators for β-cell development.45, 46 At E11.5–E13.5 of embryogenesis, hypoxia-induced HIF1-α/Notch signaling is important for the proliferation of endocrine progenitor cells. Later, at approximately E13.5, angiogenesis mediated by HIF1-α-encoding VEGF is necessary for increased oxygenation in endocrine tissue, which then destabilizes HIF1-α through degradation. Suppression of HIF1-α at this stage is essential for the normal differentiation of pancreatic islets. As such, persistent expression of HIF1-α during embryogenesis would suppress the transcription factor neurogenin 3,46 which is critical for endocrine differentiation.47 Collectively, the imbalance of HIF can affect the development of pancreas, and contribute to glucose intolerance and β-cell dysfunction.
Loss of VHL has profound effects on β-cell function, demonstrating a role for VHL in regulating glucose homeostasis by alterations in expression of HIF-regulated genes. The mutant mice had decreased GLUT2 expression in their β-cells, which is in keeping with previously published studies.27, 41 Decreased expression of GLUT2 has been reported in several animal models of diabetes.48, 49 In addition, mice with GLUT2-deficient β-cells also have impaired insulin secretion, leading to diabetes development.50 Therefore, defective glucose-sensing likely has a significant role in the impairment of β-cell function that is present in the VHL mutant mice. In addition to the changes observed within the VHL-deficient β-cells, there were significant alterations in islet architecture caused by increased vasculature likely due to Vegf upregulation, which may have influenced β-cell function. Indeed, an abnormal islet vascular bed and a twofold increase in vascular density were also reported in both pre-diabetic and diabetic male Zucker diabetic fatty rat compared with control mice.51
Deletion of VHL leads to the enhanced transcription of canonical HIF-target genes that promote cell and tissue survival, including Epo, Vegf, and Glut1, which led us to initially hypothesize that VHL deletion in the β-cells would provide cytoprotective effects. On the contrary, loss of VHL impaired insulin secretion in the β-cells, which may arise from stabilized HIF expression during pancreas development.45, 46 Indeed, previous studies have shown that hypoxia impairs insulin secretion.52, 53 Thus, these studies along with our findings and others,27, 41, 42 which all show impaired β-cell function with VHL loss, are in contrast to the results by Gunton et al,12 which suggests that a reduction in HIF expression in the islets contributes to the β-cell dysfunction in type 2 diabetes.12 In the same study, it was reported that knockdown of HIF-1α in Min6 cells leads to impaired glucose-stimulated insulin secretion.12 In the Gunton et al study, β-cell-specific ARNT deletion led to impaired insulin signaling in the β-cells,14 whereas VHL deletion in the β-cells led to defects in glucose uptake and metabolism,27, 41, 42 both of which are critical processes for efficient insulin secretion.54 These results exemplify the complex nature of both HIF-1a and HIF-1β in regulating β-cell function and mass, and further work is required for a more clear understanding of HIF biology in β-cells.
The Epo gene is one of the major hypoxia-inducible genes,31 and although it is expressed in many tissues (reviewed in Wijesekara et al23), it appears not to be present in pancreatic islets. Interestingly, overexpression of Epo in pancreatic islets was shown to have cytoprotective effects.55 We, therefore, assessed whether providing exogenous EPO by systemic rHuEPO administration could improve the glucose tolerance in the RIPcre+vhlhfl/fl mice. Indeed, rHuEPO treatment for 4 weeks improved glycemic control in the RIPcre+vhlh+/+ and RIPcre+vhlhfl/fl mice compared with their saline-treated counterparts. rHuEPO treatment was shown to activate the PI3K signaling pathway in isolated islets (D Choi, M Woo, unpublished data). In the VHL mutant mice, rHuEPO was able to rescue the defects in β-cell function perhaps by enhancing the glucose-sensing abilities through restoring GLUT2 and Gsk expression. rHuEPO treatment also reversed expression of the anaerobic glycolytic enzymes, Pdk1 and Ldha, which may rescue glucose metabolism and insulin secretion. Previous studies demonstrated that HIF degradation could also be mediated by VHL-independent mechanisms, such as Hsp90,56 RACK1,57 Gsk3,58 and Hsp70.59 Interestingly, EPO has been shown to activate Gsk360 and Hsp70,61 which can in turn alter HIF-regulated genes in islets of RIPcre+vhlhfl/fl mice.
rHuEPO treatment also reversed expression of the anaerobic glycolytic enzymes, Pdk1 and Ldha, which may rescue glucose metabolism and insulin secretion.
Together, we have shown that loss of VHL in β-cells leads to defects in glucose homeostasis due to impaired β-cell function as well as a defect in age-dependent increase in β-cell mass. We have also shown that exogenous EPO can overcome the glucose intolerance in the RIPcre+vhlhfl/fl mice, in part by restoring glucose-sensing mechanisms in β-cells to enhance insulin secretion. Our results indicate a critical role for HIF signaling in regulating β-cell mass and function, and as a potential therapeutic target for diabetes treatment.
References
Shaw JE, Sicree RA, Zimmet PZ . Global estimates of the prevalence of diabetes for 2010 and 2030. Diabetes Res Clin Pract 2009;87:4–14.
Wild S, Roglic G, Green A, et al. Global prevalence of diabetes: estimates for the year 2000 and projections for 2030. Diabetes Care 2004;27:1047–1053.
Kahn SE . The relative contributions of insulin resistance and beta-cell dysfunction to the pathophysiology of type 2 diabetes. Diabetologia 2003;46:3–19.
Rhodes CJ . Type 2 diabetes-a matter of β-cell life and death? Science 2005;307:380–384.
Jetton TL, Lausier J, LaRock K, et al. Mechanisms of compensatory beta-cell growth in insulin resistant rats: roles of Akt kinase. Diabetes 2005;54:2294–2304.
Liu YQ, Jetton TL, Leahy JL . Beta-cell adaptation to insulin resistance: increased pyruvate carboxylase activity and anaplerosis in islets of Zucker fatty rats. J Biol Chem 2002;277:39163–39168.
Steil GM, Trivedi N, Jonas JC, et al. Adaptation of beta-cell mass to substrate oversupply: enhanced function with normal gene expression. Am J Physiol Endocrinol Metab 2001;280:E788–E796.
Butler AE, Janson J, Bonner-Weir S, et al. Beta-cell deficit and increased apoptosis in humans with type-2 diabetes. Diabetes 2003;52:102–110.
Matthews DR, Cull CA, Stratton IM, et al. UKPDS 26: Sulphonylurea failure in non-insulin-dependent diabetic patients over six years. UK Prospective Diabetes Study (UKPDS) Group. Diabetic Med 1998;15:297–303.
Poitout V, Roberston RP . Secondary beta-cell failure in type 2 diabetes. A convergence of glucotoxicity and lipotoxicity. Endocrinology 2002;143:339–342.
Prentki M, Nolan CJ . Islet β-cell failure in type 2 diabetes. J Clin Invest 2006;116:1802–1812.
Gunton JE, Kulkarni RN, Yim S, et al. Loss of ARNT/HIF1beta mediates altered gene expression and pancreatic-islet dysfunction in human type 2 diabetes. Cell 2005;122:337–349.
Bunn HF, Poyton RO . Oxygen sensing and molecular adaptation to hypoxia. Physiol Rev 1996;76:839–885.
O’Rourke JF, Dachs GU, Gleadle JM, et al. Hypoxia response elements. Oncol Res 1997;9:327–332.
Semenza GL . HIF-1 and human disease: one highly involved factor. Genes Dev 2000;14:1983–1991.
Wenger RH, Gassman M . Oxygen(es) and the hypoxia-inducible factor-1. Biol Chem 1997;378:609–616.
Bonventre JB . Kidney ischemic preconditioning. Curr Opin Nephrol Hypertens 2002;11:43–48.
Gidday JM . Cerebral preconditioning and ischaemic tolerance. Nat Rev Neurosci 2006;7:437–448.
Yellon DM, Hausenloy DJ . Realizing the clinical potential of ischemic preconditioning and postconditioning. Nat Clin Pract Cardiovasc Med 2005;2:568–575.
Postic C, Shiota M, Niswender KD, et al. Dual roles for glucokinase in glucose homeostasis as determined by liver and pancreatic beta cell-specific gene knock-outs using cre recombinase. J Biol Chem 1999;274:305–315.
Gnarra JR, Ward JM, Porter FD, et al. Defective placental vasculogenesis causes embryonic lethality in VHL-deficient mice. Proc Natl Acad Sci USA 2005;94:9102–9107.
Liadis N, Murakami K, Eweida M, et al. Caspase-3-dependent beta-cell apoptosis in the initiation of autoimmune diabetes mellitus. Mol Cell Biol 2005;25:3620–3629.
Wijesekara N, Konrad D, Eweida M, et al. Muscle-specific PTEN deletion protects against insulin resistance and diabetes. Mol Cell Biol 2005;25:1135–1145.
Savino C, Pedotti R, Baggi F, et al. Delayed administration of erythropoietin and its non-erythropoietic derivatives ameliorates chronic autoimmune encephalomyelitis. J Neuroimmunol 2006;172:27–37.
Choudhury AI, Heffron H, Smith MA, et al. The role of the insulin receptor substrate 2 in hypothalamic and beta cell function. J Clin Invest 2005;115:940–950.
Nguyen KT, Tajmir P, Lin CH, et al. Essential role of Pten in body size determination and pancreatic β-cell homeostasis in vivo. Mol Cell Biol 2006;26:4511–4518.
Cantley J, Selman C, Shukla D, et al. Deletion of the von Hippel-Lindau gene in pancreatic beta cells impairs glucose homeostasis in mice. J Clin Invest 2009;119:125–135.
Beleslin-Cokic BB, Cokic VP, Yu X, et al. Erythropoietin and hypoxia stimulate erythropoietin receptor and nitric oxide production by endothelial cells. Blood 2004;104:2073–2080.
Semenza GL . Oxygen-dependent regulation of mitochondrial respiration by hypoxia-inducible factor 1. Biochem J 2007;405:1–9.
Beatens D, Malaisse-Lagae F, Perrelet A, et al. Endocrine pancreas: three-dimensional reconstruction shows two types of islets of Langerhans. Science 1979;206:1323–1325.
Wenger RH . Cellular adaptation to hypoxia: O2-sensing protein hydroxylases, hypoxia-inducible transcription factors, and O2-regulated gene expression. FASEB J 2002;16:1151–1162.
Pugh CW, Ratcliffe PJ . Regulation of angiogenesis by hypoxia: role of the HIF system. Nat Med 2003;9:677–684.
Lacombe C, Da Silva JL, Bruneval P, et al. Peritubular cells are the site of erythropoietin synthesis in the murine hypoxic kidney. J Clin Invest 1998;81:620–623.
Zanjani ED, Ascensao JL, McGlave PB, et al. Studies on the liver to kidney switch of erythropoietin production. J Clin Invest 1981;67:1183–1188.
Maiese K, Li F, Chong ZZ . New avenues of exploration for erythropoietin. JAMA 2005;293:90–95.
Spivak JL, Pham T, Isaacs M, et al. Erythropoietin is both a mitogen and a survival factor. Blood 1991;77:1228–1233.
Fenjves ES, Ochoa MS, Cabrera O, et al. Human, nonhuman primate, and rat pancreatic islets express erythropoietin receptors. Transplantation 2003;75:1356–1360.
Maxwell PH, Wiesener MS, Chang GW, et al. The tumour suppressor protein VHL targets hypoxia-inducible factors for oxygen-dependent proteolysis. Nature 1999;399:271–275.
Lonser RR, Glen GM, Walther M, et al. Von Hippel-Lindau disease. Lancet 2003;361:2059–2067.
Shen HC, Adem A, Ylaya K, et al. Deciphering von Hippel-Lindau (VHL/Vhl)-associated pancreatic manifestations by inactivating Vhl in specific pancreatic cell populations. PLoS One 2009;4:e4897.
Puri S, Cano DA, Hebrok M . A role for von Hippel-Lindau protein in pancreatic beta-cell function. Diabetes 2009;58:433–441.
Zehetner J, Danzer C, Collins S, et al. PVHL is a regulator of glucose metabolism and insulin secretion in pancreatic beta cells. Genes Dev 2008;22:3135–3146.
Bossenmeyer-Pourle C, Koziel V, Daval JL . CPP32/Caspase-3-like proteases in hypoxia-induced apoptosis in developing brain neurons. Mol Brain Res 1999;71:225–237.
Todor A, Sharov VG, Tanhehco EJ, et al. Hypoxia-induced cleavage of caspase-3 and DFF45/ICAD in human failed cardiomyocytes. Am J Physiol Heart Circ Physiol 2002;283:H990–H995.
Heinis M, Simon MT, Duvillié B . New insights into endocrine pancreatic development: the role of environmental factors. Horm Res Paediatr 2010;74:77–82.
Heinis M, Simon MT, Ilc K, et al. Oxygen tension regulates pancreatic β-cell differentiation through HIF1. Diabetes 2010;59:662–669.
Gradwohl G, Dierich A, LeMeur M, et al. Neurogenin-3 is required for the development of the four endocrine cell lineages of the pancreas. Proc Natl Acad Sci USA 2000;97:1607–1611.
Bonny C, Roduit R, Gremlich S, et al. The loss of GLUT2 expression in the pancreatic beta-cells of diabetic db/db mice is associated with an impaired DNA-binding activity of islet-specific transacting factors. Mol Cell Endocrinol 1997;135:59–65.
Ohneda M, Johnson JH, Inman LR, et al. GLUT2 expression and function in beta-cells of GK rats with NIDDM: dissociation between reductions in glucose transport and glucose-stimulated insulin secretion. Diabetes 1993;42:1065–1072.
Li X, Zhang L, Meshinchi S, et al. Islet microvasculature in islet hyperplasia and failure in a model of type 2 diabetes. Diabetes 2006;55:2965–2973.
Dionne KE, Colton CK, Yarmush ML . Effect of hypoxia on insulin secretion by isolated rat and canine islets of Langerhans. Diabetes 1993;42:12–21.
Narimiya M, Yamada H, Matsuba I, et al. The effect of hypoxia on insulin and glucagon secretion in the perfused pancreas of the rat. Endocrinology 1982;111:1010–1014.
Saltiel AR, Kahn CR . Insulin signaling and the regulation of glucose and lipid metabolism. Nature 2001;414:799–806.
Fenjves ES, Ochoa MS, Gay-Ravinstein C, et al. Adenoviral gene transfer of erythropoietin confers cytoprotection to isolated pancreatic islets. Transplantation 2004;77:13–18.
Isaacs JS, Jung YJ, Mimnaugh EG, et al. Hsp90 regulates a von Hippel Lindau-independent hypoxia-inducible factor-1α-degradative pathway. J Biol chem 2002;277:29936–29944.
Liu YV, Baek JH, Zhang H, et al. RACK1 Competes with HSP90 for binding to HIF-1α and is required for O2-independent and HSP90 inhibitor-induced degradation of HIF-1α. Mol Cell 2007;25:207–217.
Flügel D, Görlach A, Michiels C, et al. Glycogen synthase kinase 3 phosphorylates hypoxia-inducible factor 1 and mediates its destabilization in a VHL-independent manner. Mol Cell Biol 2007;27:3253–3265.
Kong X, Lin Z, Liang D, et al. Histone deacetylase inhibitors induce VHL and ubiquitin-independent proteasomal degradation of hypoxia-inducible factor 1α. Mol Cell Biol 2006;26:2019–2028.
Ohori K, Miura T, Tanno M, et al. Ser9-phosphorylation of mitochondrial GSK-3{beta} is a primary mechanism of cardiomyocyte protection by erythropoietin against oxidant-induced apoptosis. Am J Physiol Heart Circ Physiol 2008;295:2079–2086.
Xu B, Dong GH, Liu H, et al. human erythropoietin pretreatment attenuates myocardial infarct size: a possible mechanism involves heat shock protein 70 and attenuation of nuclear factor-kappaB. Ann Clin Lab Sci 2005;35:161–168.
Guillam MT, Hümmler E, Schaerer E, et al. Early diabetes and abnormal postnatal pancreatic islet development in mice lacking Glut-2. Nat Genet 1997;17:327–330.
Acknowledgements
DC is supported by the CIHR Federick Banting and Charles Best Canada Graduate Scholarship (Doctoral). This work was supported by the Canadian Institutes of Health Research (MOP-201188).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Laboratory Investigation website
Supplementary information
Rights and permissions
About this article
Cite this article
Choi, D., Cai, E., Schroer, S. et al. Vhl is required for normal pancreatic β cell function and the maintenance of β cell mass with age in mice. Lab Invest 91, 527–538 (2011). https://doi.org/10.1038/labinvest.2010.207
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2010.207