Abstract
Hepcidin is downregulated during progressive cholestasis in biliary atresia, but the mechanism is unknown. To verify whether downregulation of hepcidin is specific to cholestasis irrespective of the patient's age, we first analyzed liver hepcidin mRNA and protein expression in adults with primary biliary cirrhosis (PBC) (n=4), non-cholestatic cirrhosis (n=19) and in controls (n=9). We evaluated the tyrosine phosphorylation of signal transducer and activator of transcription 3 (pSTAT3) expressions in the liver sections. A rat model of cholestasis by ligation of the extrahepatic bile duct (BDL) was created, and lipopolysaccharide (LPS)-induced cholangitis in cholestatic rats 2 weeks after BDL was also established to study the modulation of hepcidin by interleukin-6 (IL-6) and STAT3 signaling pathway in these models, using real-time quantitative reverse transcription–PCR, immunohistochemistry, western blotting and enzyme-linked immunosorbent assay (ELISA). An in vitro study of the effect of bile acids on hepcidin expression was carried out to re-confirm the in vivo findings. There was significantly lower hepcidin mRNA and pSTAT3 protein expression in cholestatic cirrhosis compared with non-cholestatic cirrhosis in adults. BDL caused significant decrease in hepcidin and gp130 mRNA expression compared with sham-operated group and normal control. Furthermore, there was significantly lower pSTAT3 protein expression and nuclear translocation in the cholestatic liver from the patients and the BDL rats, which was comparable to lower liver hepcidin mRNA and plasma hepcidin expression. Furthermore, BDL for 2 weeks attenuated the upregulation of hepcidin expression induced by LPS. Hydrophobic bile acid glycochenodeoxycholate inhibited IL-6-induced pSTAT3 expression in primary hepatocytes and resulted in the downregulation of hepcidin mRNA expression. In conclusion, the study shows that cholestasis or its important component—hydrophobic bile acids—can downregulate hepcidin expression through inhibiting IL-6-induced STAT3 phosphorylation and pSTAT3 protein nuclear translocation.
Similar content being viewed by others
Main
Biliary atresia (BA) is characterized by progressive destruction and complete obliteration of the extrahepatic bile ducts of unknown etiology within months of birth. BA still accounts for a great proportion of patients receiving liver transplantation in many transplant centers.1 Many factors are responsible for deterioration of the liver in BA even after a successful Kasai's procedure (KP), which may include impaired reticuloendothelial system to clear the translocated bacteria and endotoxin from the intestine.2 Even in patients of benign biliary obstruction3 and in animals with bile duct ligation (BDL),4, 5 there are still more bacterial overgrowth in the liver and the bile, but the underlying mechanism is still unknown.
Human hepcidin is first isolated and characterized as a highly disulfide-bonded peptide with antimicrobial activity, expressed mainly in the liver and is also named liver-expressed antimicrobial peptide-1, or LEAP-1.6 Hepcidin, as a negative regulator of iron absorption from the duodenum and released from macrophages, is upregulated in the liver by inflammation and iron overload.7, 8 Hepcidin has significant antimicrobial property for Escherichia coli, 9 which is the most common bacterium involved in post-operative cholangitis in BA.10 The latter may exacerbate preexisting liver fibrosis leading to end-stage liver cirrhosis.
In our previous study, hepatic expression of hepcidin decreased remarkably in the cirrhotic liver due to BA11 However, in the study of Aoki et al12 liver hepcidin mRNA correlates only with iron stores, but not inflammation and viral genotype in patients with chronic hepatitis C. In addition, Bergmann et al13 also showed that levels of hepcidin mRNA did not differ between the controls and cirrhotic livers. On the contrary, a significant decrease in serum prohepcidin was observed in patients with liver cirrhosis compared with healthy individuals, which was most prominent in patients with hepatitis C virus and alcohol-related liver cirrhosis.14 HCV-induced oxidative stress suppresses hepcidin expression through increased histone deacetylase activity.15 Hepcidin is also downregulated in ethanol-loaded mice liver.16 There are many conflicting observations about hepcidin level in cirrhosis. It is likely that the decreased hepcidin expression in the late stage of BA may be a disease-specific phenomenon reflecting cholestatic liver injury characteristic of BA.
The control of hepcidin expression is very complex, which includes the interaction of inflammatory cytokines. Pietrangelo et al17 first show that interleukin-6 (IL-6) increases hepcidin expression through a complex of the IL-6 receptor and gp130. It involves promoter binding of signal transducer and activator of transcription 3 (STAT3) or iron metabolism through bone morphogenetic protein (BMP)/Smad4 pathway and reactive oxygen species through CCAAT/enhancer-binding protein alpha.18, 19, 20 In our previous study, we have shown the higher hepatic IL-6 expression in the late stage of BA than in early stage,21 which did not correlate with lower hepcidin expression in the late stage of BA. Graf et al22 found that bile acids inhibit IL-6 signaling through decreased gp130 protein expression and inhibition of STAT3 phosphorylation in rat liver, which implies that cholestasis may be a major determinant of hepcidin expression during the progression of liver cirrhosis. To prove the hypothesis, we studied the hepcidin expression in patients with primary biliary cirrhosis (PBC), which is a chronic cholestatic liver disease leading to biliary cirrhosis in adult,23 very similar to children with BA. We also used a rat model of cholestasis and cholangitis, as well as in vitro bile acid modulation of hepcidin expression to unveil the mechanism of inhibition of hepcidin expression in cholestatic liver injury by blocking the IL-6–gp130–phoshpoSTAT3 signaling pathway.
MATERIALS AND METHODS
Patients and Samples
Fresh liver tissues were obtained from six patients at the early stage of BA when they received KP and six at the late stage of BA when they received liver transplantation. Six patients of choledochal cyst when they underwent surgical intervention, and five patients of neonatal hepatitis when they underwent exploratory laparotomy and liver biopsy, served as disease control. The livers removed at liver transplantation from four patients with PBC served as adult cholestatic cirrhosis (PBC); nine with hepatitis B virus-related cirrhosis (HBV), six with hepatitis C virus-related cirrhosis (HCV) and four with alcoholic cirrhosis (alcohol) served as non-cholestatic cirrhosis. Eight non-cirrhotic liver specimens from patients receiving liver resection for HBV-associated hepatoma were used as disease controls (HBV non-cirrhosis). Nine liver samples from five children and four adults undergoing liver resection for benign lesion of various causes in which the livers were devoid of fibrosis and cholestasis were served as normal control (normal control). Detailed history of the patients was recorded, including age when the patient received operation, sex and serum aspartate aminotransferase, alanine aminotransferase and total bilirubin (Table 1). After surgical removal, all the liver samples were immediately divided into four pieces, put into cryogenic vials (CORNINGs), immersed in liquid nitrogen and then stored at −80°C until use. The study was approved by the Ethics and Clinical Research Committee of the Chang Gung Memorial Hospital, and the informed consent had been obtained from the patients or their legal guardians.
Animal Model and Experimental Protocol
Male Sprague–Dawley rats, purchased from the National Animal Center of Academia Sinica, Taipei. Taiwan, were maintained on standard laboratory rat chow under a 12-h light–dark cycle. Care and use of rats strictly followed a protocol that was approved by the Animal Ethics Committee of the Chang Gung Memorial Hospital. The rats received laparotomy and were randomly allocated into two groups, either with (BDL) or without (sham) common bile duct ligation. The detailed procedures were described elsewhere.24 The rats were killed at different time points, including 6 h, 24 h, 3 days, 1 week and 2 weeks, and the number of animals at each time point is six.
To create bacterial cholangitis, a silicone catheter with an inner diameter of 0.508 mm and outer diameter of 0.930 mm (Silicone Elastomer, Helix Medical, Carpinteria, CA, USA) was inserted into the proximal and distal bile ducts, with the middle segment tunneled and located in the subcutaneous space. Rats undergoing ligation of the subcutaneous segment of the indwelling catheter, which caused complete biliary obstruction, were defined as bile duct-ligated rats. Rats that did not undergo ligation of the catheter were served as sham rats. Two weeks later, a small incision was made over the previous laparotomy wound and 0.5 μg of lipopolysaccharide (LPS) (from E. coli O111:B4, Sigma, St Louis, MO, USA) dissolved in 0.25 ml of normal saline, or equivalent amount of normal saline, was injected to the bile duct through the subcutaneous segment of the indwelling catheter with a Fr. 30 short needle (ULTRA-FINE, Becton Dickinson, Franklin Lakes, NJ, USA). Three hours later, all the animals were killed with an overdose of thiopentone sodium (Pentothal). Liver tissues were snap-frozen for mRNA and protein determinations and the remaining were fixed in 4% paraformaldehyde and embedded in paraffin for histology and immunohistochemical analysis.
Cell Culture and Isolation of Primary Rat Hepatocytes
The liver of a group of normal rats was perfused in situ through the portal vein with 30 ml Ca2+- and Mg2+-free Hank's balanced salt solution (5 ml/minute) (Mediatech Inc, Herndon, VA, USA), and followed by perfusion with 0.1% pronase and 0.04% collagenase (Sigma). The liver was then removed, meshed, and incubated with 0.08% pronase E, 0.08% collagenase and 20 μg/ml of DNase. The suspension was filtered through a 150-μm steel mesh and purified through Percoll (Sigma) gradient centrifugation, which resulted in a hepatocyte cell-enriched fraction layer. Cells (1 × 106) were plated on collagen-coated culture dishes and were cultured for 24 h in 1.5 ml William's medium E (Sigma Chemical Co., Munich, Germany), supplemented with 2 mmol/l glutamine, 100 U/ml penicillin, 0.1 mg/ml streptomycin, 10–7 mol/l insulin, 10–7 mol/l dexamethasone and 10% fetal calf serum. Two hours before starting the experiments, cells were washed again and incubated with the same medium. Hepatocytes were treated with sodium glycochenodeoxycholic acid (GCDC) (Sigma) for 3 h followed by 10 ng/ml IL-6. Cell were harvested 30 min later for protein study and 6 h later for mRNA study.
RNA Isolation and Real-Time QRT-PCR
To quantitate the tissue amount of mRNA, we used real-time quantitative reverse transcription–PCR (QRT-PCR) with ABI 7700 Sequence Detection System (TaqMan, Perkin-Elmer Applied Biosystems). In all, 2 μg of total RNA was used to generate cDNA using an oligodeoxynucleotide primer (oligo dT15) following the protocol for transcription (Promega, Madison, WI, USA). PCR was carried out in SYBR Green PCR Master Mix (Applied Biosystems) containing 10 μM specific forward and reverse primers. Amplification and detection were carried out with an ABI7700 system with the following profile: 1 cycle at 95°C for 10 min, and 40 cycles at 95, 60 and 72°C for 15, 30 and 15 s, respectively. The mRNA of the β-actin was chosen as internal control in each sample. Relative quantification of gene expression was based on comparative CT method, in which the amount of the target was given by  or
or  . PCR products were electrophoresed on a 2% agarose gel to confirm the sizes of the products. The forward and reverse primer sequences were designed as follows: human hepcidin, forward 5′-TCCCACAACAGACGGGACA-3′, reverse 5′-GAACATGGGCATCCAGCTG-3′; human β-actin, forward 5′-TCACCCACACTGTGCCCATCTACGA-3′, reverse 5′-CAGCGGAACCGCTCATTGCCAATGG-3′; rat hepcidin, forward 5′-TGTCTCCTGCTTCTCCTCCT-3′, reverse 5′-CTCTGTAGTCTGTCTCATCTGTTG-3′; rat gp130, forward 5′-CCGTCAGTGCAAGTGTTCTCA-3′, reverse 5′-CACTATCCACCAGCTGCAGGT-3′; rat IL-6, forward 5′-GTGTGAAAGCAGCAAAGAGGC-3′, reverse 5′-TGCAGGAACTGGATCAGGACT-3′; rat β-actin, forward 5′-TCACCCACAATGTGCCCATCTTCGA-3′, reverse 5′-CAGCGGAACCGCTCATTG CCAATGG-3′.
. PCR products were electrophoresed on a 2% agarose gel to confirm the sizes of the products. The forward and reverse primer sequences were designed as follows: human hepcidin, forward 5′-TCCCACAACAGACGGGACA-3′, reverse 5′-GAACATGGGCATCCAGCTG-3′; human β-actin, forward 5′-TCACCCACACTGTGCCCATCTACGA-3′, reverse 5′-CAGCGGAACCGCTCATTGCCAATGG-3′; rat hepcidin, forward 5′-TGTCTCCTGCTTCTCCTCCT-3′, reverse 5′-CTCTGTAGTCTGTCTCATCTGTTG-3′; rat gp130, forward 5′-CCGTCAGTGCAAGTGTTCTCA-3′, reverse 5′-CACTATCCACCAGCTGCAGGT-3′; rat IL-6, forward 5′-GTGTGAAAGCAGCAAAGAGGC-3′, reverse 5′-TGCAGGAACTGGATCAGGACT-3′; rat β-actin, forward 5′-TCACCCACAATGTGCCCATCTTCGA-3′, reverse 5′-CAGCGGAACCGCTCATTG CCAATGG-3′.
The validation experiments were carried out in duplicate and amplification efficiencies were validated.
Immunohistochemistry
Immunohistochemical staining of hepcidin, STAT3 and pSTAT3 (Alpha Diagnostic International, San Antonio, TX, USA; Cat. # HEPC 12-A, Santa Cruz Biotechnology, CA, USA, sc-482 and Cell Signaling Technology, Inc, No. 9145, respectively) was carried out on the paraffin-embedded, formalin-fixed, archival liver tissues obtained from the Department of Pathology and rats. Sections of 2-μm thickness were deparaffinized, treated with 3% hydrogen peroxide to inactivate the endogenous peroxidase activity and microwaved for 10 min in 10 mM citrate buffer to retrieve antigen. The sections were then incubated with a first antibody at room temperature for 2 h and detected by a second antibody with SuperPicTure Polymer detection kit (Cat. # 87–8963; Zymed Laboratories, South San Francisco, CA, USA) with diaminobenzidine (DAB) chromogen (Cat # K3467; DAKO, Carpinteria, CA, USA). As hepcidin is expressed in normal liver, a liver sample from polycystic liver disease served as normal positive control.6, 8 Omitting the primary antibody in one sample served as negative control. The intensity of immunoreactivity for hepcidin was graded according to the following criteria: grade 0, no visible immunoreactive dot; grade 1, immunoreactive dots visible less than positive control; grade 2, immunoreactive dots visible equal to positive control; and grade 3, immunoreactive dots visible more than positive control. The intensity of immunoreactivity for STAT3 and pSTAT3 was graded as grades 1–3 according to the following criteria: the areas of positive cells in hepatic lobules, hepatic sinusoid, portal areas and fibrous plate <10%, 10–50% and >50% of total liver tissues, respectively.
Western Blot Analysis
Thirty micrograms of crude proteins were treated with sample buffer, then boiled for 10 min, separated in 12% SDS-PAGE gels and transferred to nitrocellulose. Blots were incubated for the respective first antibodies (hepcidin, STAT3 and pSTAT3). After washing with TBST and incubation with horseradish peroxidase-coupled anti-rabbit immunoglobulin G antibody (dilution 1:10,000) at room temperature for 2 h, the blots were developed using enhanced chemiluminescent detection (Amersham Pharmacia Biotech, Uppsala, Sweden) and were exposed to film. Signals were quantified by densitometric analysis.
Perls' Acid Ferrocyanide Stain
Paraffin-embedded, formalin-fixed sections were deparaffinized and hydrated with distilled water. We then mixed equal parts of hydrochloric acid and potassium ferrocyanide prepared immediately before use. Slides were immersed in this solution for 20 min and washed in distilled water for three changes. The slides were counterstained with nuclear fast red for 5 min, rinsed twice in distilled water, and then dehydrated through 95% and two changes of 100% alcohol. The slides were cleared in xylene, two changes, for 3 min each and then covered with resinous mounting medium. In this stain, iron (ferric form) was stained bright blue, nuclei were stained red and cytoplasm was stained pink.
Hepcidin ELISA
Plasma samples were obtained from experimental animals. Determinations were performed by using 96-well microtiter plates coated with a polyclonal antibody against hepcidin (Cat # E1979r; USCNLIFE, China). Assay procedures were according to the manufacturer's instructions and the absorbance of each well was determined at 450 nm wavelength.
Statistical Analysis
All values in the figures and tables were expressed as mean±s.e. of the man. Student's t-test (unpaired, two-tailed) was used for comparison between experimental groups with continuous variables. A P-value <0.05 was considered as statistically significant.
RESULTS
Hepcidin Expression is Significantly Downregulated in Cholestatic Cirrhosis
The liver mean hepcidin mRNA expression was >20-fold less in PBC than in normal control (P=0.01) (Figure 1), which was consistent with our previous report11 that hepcidin mRNA expression decreased significantly in cholestatic cirrhosis of BA than that in normal child control. Meanwhile, the liver hepcidin mRNA expressions of HBV, HCV and alcohol-induced non-cholestatic cirrhosis showed no significant difference from the normal control (P=0.12, 0.13 and 0.29 respectively). There was also no significant difference in liver hepcidin mRNA expression between cirrhotic and non-cirrhotic stages of HBV-associated liver diseases (P=0.33). Taken together, hepcidin mRNA expression was significantly downregulated only in cholestatic cirrhosis, but not in non-cholestatic cirrhosis, regardless of the age of the patients.
To further characterize hepcidin protein expression in the liver, we carried out immunohistochemical analysis on liver sections from four adults with PBC, hepatitis B, C and alcohol-induced cirrhosis. In histological normal and non-cholestatic cirrhotic livers, immunohistochemistry (brown color) revealed the expression of hepcidin in the cytoplasm of the hepatocytes and bile ductular epithelial cells (Figure 2). Immunoreactive hepcidin staining of representative liver tissues showed a grade 2 staining in a polycystic liver disease without cirrhosis serving as a positive control (Figure 2a), a grade 3 staining in the alcoholic cirrhosis (Figure 2b), a grade 2 staining in the hepatitis B-induced cirrhosis (Figure 2c) and a grade 0 staining in PBC (Figure 2d). Immunoreactive hepcidin staining in the liver samples from hepatitis B, C and alcoholic cirrhosis were in grades 2–3, which was either comparable to or a little higher than the control liver. On the contrary, no visible immunoreactive hepcidin staining in both hepatocytes and bile ductular epithelial cells was found in the liver sample of PBC. The trend of hepcidin immunoreactivity of bile ductular epithelial cells (arrow) was the same as that of the hepatocytes.
Immunoreactive hepcidin staining and hepatic stainable iron between cirrhotic and non-cirrhotic liver in adults. Hepcidin (brown color) are localized in the cytoplasm of the hepatocytes and the bile ductular epithelial cells. The representative liver tissues show a grade 2 staining in a polycystic liver disease without cirrhosis serving as a positive control (a), a grade 3 staining in the alcoholic cirrhosis (b), a grade 2 staining in the hepatocytes and cholangiocytes (arrow) of the hepatitis B-induced cirrhosis (c), and a grade 0 staining in the hepatocytes and cholangiocytes (arrow) of the primary biliary cirrhosis (PBC) (d). There are more coarse granules of stainable iron in hepatocytes in the specimen from hepatitis B-induced cirrhosis (e) than in PBC (f) (original magnification, × 400).
We compared the hepatic stainable iron in PBC and other non-cholestatic cirrhosis. There were more coarse granules of stainable iron in hepatocytes in the specimen from hepatitis B-induced cirrhosis (Figure 2e) than that in PBC (Figure 2f); the latter was essentially devoid of stainable iron.
Phospho-STAT3 Expression is Significantly Downregulated in Cholestatic Cirrhosis in BA
As a transcription factor for hepcidin expression, phospho-STAT3 (pSTAT3) protein expression was characterized in the liver by immunohistochemical staining. In the liver tissues of the early stage of BA (Figure 3a), pSTAT3 immunoreactivity was constantly expressed in both the cytoplasm and the nuclei of the hepatocytes (arrow), bile ductular epithelial cells and some nonparenchymal cells morphologically identical to Kupffer cells (arrowhead). The pSTAT3 staining was weak in the late stage of BA and there was a significant decrease in the hepatic nuclear pSTAT3 immunoreactivity (Figure 3b) (arrow). Using western blotting, significant downregulation of hepatic STAT3 protein expression was found in the late stage of BA, comparing with the early stage and to neonatal hepatitis and choledochal cyst (Figure 3c and d). The trend of pSTAT3 nuclear protein expression was the same as hepcidin mRNA expression in the early and the late stage of BA.11
Phospho-STAT3 (pSTAT3) expression in biliary atresia (BA) and other liver disorders. (a) In the early stage of BA, increased pSTAT3 immunoreactivity is constantly expressed in both the cytoplasm and the nuclei of the hepatocytes (arrow), bile ductular epithelial cells and some nonparenchymal cells morphologically identical to Kupffer cells (arrowhead). (b) In the late stage of BA, pSTAT3 immunoreactivity is very faintly present in the hepatocytes and no nuclear immunoreactivity is found. (c) Comparison of pSTAT3 protein expression by western blotting between neonatal hepatitis, choledochal cyst, and the early and late stages of BA. (d) Statistical comparison of pSTAT3 protein between groups. N indicates the sample size. * indicates P<0.05 between groups (original magnification, × 200; insert, × 400).
Both Hepatic Hepcidin and pSTAT3 Expressions Decrease Significantly in the Rats with Cholestasis
IL-6 family uses the receptor gp130 as a signal transducer to activate the nuclear translocation of pSTAT3, which can transactivate hepcidin in response to inflammation. In our previous study, we have shown higher hepatic IL-6 expression in the late stage of BA than in early stage and in choledochal cyst, which was not consistent with lower hepcidin expression in the late stage of BA.21 Therefore, we measured IL-6, gp 130 and hepcidin mRNA expressions in an animal model of the BDL by QRT-PCR. We found higher IL-6 mRNA expression in the BDL group than in the sham group and the non-operative control group (Figure 4a), but lower hepcidin (Figure 4b) and gp 130 (Figure 4c) mRNA expressions in the BDL group than in the sham group. Excluding the factor of cholestasis, there was only marginal positive correlation between IL-6 and hepcidin mRNA expression (R2=0.072, P=0.049). To show the effect of cholestasis, we divided the rat into cholestatic group and non-cholestatic group for comparison. Interestingly, there was a significant correlation between IL-6 and hepcidin mRNA expression in both groups (R2=0.403, P<0.001; R2=0.362, P<0.001, respectively), but cholestasis could attenuate the upregulation of hepcidin expression induced by IL-6 (Figure 4d). By using multiple linear regression analysis in the BDL animal model, we could create a mathematic equation to predict the hepatic expression of hepcidin mRNA with the level of IL-6 mRNA (Log10Hepcidin=0.617 × log10IL6–0.946 × cholestasis (0,1)–0.339; R2=0.469, P<0.001) and concluded that cholestasis is as important as IL-6 in the regulation of hepcidin expression. By using hepcidin enzyme-linked immunosorbent assay (ELISA) , the levels of plasma hepcidin were higher in sham group than in the BDL group (Figure 5). The plasma hepcidin also showed the same trend as liver hepcidin mRNA expression (P=0.028). Furthermore, we found that the immunoreactive hepcidin staining in the sham-operated rat (Figure 6a) was higher than that in the BDL rat (Figure 6b). The immunoreactive STAT3 staining did not show any difference between the sham-operated rat (Figure 6c) and the BDL rat (Figure 6d). In the sham-operated group (Figure 6e), higher pSTAT3 immunoreactivity is constantly expressed in both the cytoplasm and the nuclei of the hepatocytes (arrow), and in some nonparenchymal cells morphologically identical to Kupffer cells (arrowhead). There is no pSTAT3 immunoreactivity in the liver of the BDL rat (Figure 6f). The trend of pSTAT3 immunoreactivity is the same as hepcidin expression. Together, these data suggest that cholestasis may block IL-6–gp130–phoshpoSTAT3 pathway, resulting in decreased hepcidin expression in spite of increased IL-6 expression. Hepcidin served as a negative regulator of iron absorption from the duodenum and released from macrophages. We studied the stainable iron in the jaundiced rats. Perls' stain showed remarkably higher stainable iron (blue color) in BDL rat. Stainable iron was sequestrated and localized mainly in Kupffer cells (Figure 6h, arrowheads). The increased hepatic stainable iron in jaundiced rat compared with the sham-operated group (Figure 6g and h) was compatible with the downregulation of hepcidin in cholestasis.
IL-6, gp130 and hepcidin mRNA expression in the bile duct-ligated (BDL) rat. (a) Higher IL-6 mRNA expression in the BDL group compared with that in the sham and control groups. (b) Lower hepcidin and (c) gp 130 mRNA expressions in the BDL group compared with that in the sham group. (d) To show the effect of cholestasis, we divide the rat into cholestasis group and non-cholestasis group for comparison. There is a significant correlation in the both groups, but cholestasis could attenuate the upregulation of hepcidin expression induced by IL-6 (line A, non-cholestasis group, R2=0.403, P<0.001; line B, cholestasis group, R2=0.362, P<0.001). N=6 in each group. Data are expressed as mean±s.e. of the mean. * indicates P<0.05 between the groups.
Hepcidin, STAT3, Phospho-STAT3 (pSTAT3) and hepatic stainable iron expression of representative liver tissues of the sham-operated and bile duct-ligated rats (BDL). Immunoreactive hepcidin staining (brown color) shows a grade 3 staining in the sham-operated rat (a), a weak grade 1 staining in BDL rat (b). Immunoreactive STAT3 staining (brown color) shows a grade 3 staining in the sham-operated rat (c) and the same grade of staining in BDL rat (d). In the sham-operated group (e), increased pSTAT3 immunoreactivity is constantly expressed in both the cytoplasm and the nuclei of the hepatocytes (arrow), and in some nonparenchymal cells morphologically identical to Kupffer cells (arrowhead) There is no pSTAT3 immunoreactivity in the liver of BDL rat (f). Perls' stain shows no stainable iron in the liver of sham-operated rat (g), but remarkably higher stainable iron (blue color) in BDL rat (h), which is localized mainly in Kupffer cells (arrowheads) (original magnification, × 400).
Cholestasis Attenuates the Endotoxin-Induced Hepcidin Expression
Acute suppurative cholangitis is associated with significant morbidity and mortality, particularly in patients with obstructive jaundice.25 In our previous study, LPS-induced cholangitis in the jaundiced rats is significantly associated with higher grade of liver necrosis and mortality rate.24 Endotoxin (LPS) is a potent inducer of inflammatory cytokines, including IL-6, and is known to induce hepcidin expression. We suspected that hepcidin response in the jaundiced rats is impaired even when challenged with endotoxin. Compared with the BDL animals receiving normal saline infusion, hepatic hepcidin mRNA was elevated for less than twofolds after infusion with LPS. However, LPS infusion in the sham group resulted in more than sixfolds of hepatic hepcidin mRNA increase, which was also significantly higher than that in the BDL rats (Figure 7). Our findings indicated that hepcidin, as a natural antimicrobial peptide, was significantly suppressed in the jaundiced rats when challenged with LPS infusion in the biliary system.
Hepcidin mRNA expression by real-time QRT-PCR shows significant upregulation for more than sixfolds in the sham rats receiving LPS infusion, comparing with the jaundiced rats and sham control. There is only marginally significant upregulation of hepcidin mRNA expression in the jaundiced rats receiving LPS than in those receiving normal saline infusion. NS indicates normal saline. Data are expressed as mean±s.e. of the mean of six samples in each group. * indicates P<0.05 between the groups.
Bile Acid can Decrease Hepatocyte Hepcidin Expression in Response to IL-6 Through Inhibition of IL-6-Induced STAT3 Phosphorylation
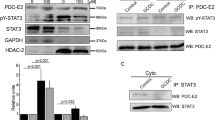
To investigate the mechanism why cholestasis may suppress hepatic hepcidin expression, we treated primary hepatocytes with one of the hydrophobic bile acids—GCDC—with concentration of 50 and 100 μmole before IL-6 (10 ng/ml) stimulation. We found that GCDC could decrease the upregulation of hepatocyte hepcidin mRNA expression in response to IL-6 stimulation (Figure 8a). At the same time, the upregulation of pSTAT3 induced by IL-6 was significantly inhibited (Figure 8b). Collectively, hydrophobic bile acids could downregulate hepcidin expression by blocking IL-6–gp130–phoshpoSTAT3 pathway.
Effects of GCDC on IL-6-induced hepcidin expression and STAT3 phosphorylation (Tyr705) in cultured rat hepatocytes. Rat hepatocytes are preincubated without (control) or with GCDC (100 μmol/l) for 6 h. When indicated, IL-6 (10 ng/ml) is added for another 6 h and 30 min for mRNA and protein study respectively after the GCDC preincubation period. (a) IL-6 could increase hepatocyte hepcidin mRNA expression but hydrophobic bile acid decreases the upregulation of hepcidin expression in response to IL-6 stimulation. (b) IL-6 could increase phospho-STAT3 expression, but pretreatment with GCDC results in significant inhibition of upregulation of phospho-STAT3 induced by IL-6. Data are expressed as mean±s.e. of the mean of six samples in each group. * indicates P<0.05 between the groups.
DISCUSSION
We show for the first time that cholestasis can decrease hepcidin expression through inhibiting IL-6-induced STAT3 tyrosine705 phosphorylation in human, jaundiced rats and rat hepatocytes. Significant downregulation of hepatic hepcidin expression occurs only in cholestatic cirrhosis of BA and PBC, but not in virus-related or alcoholic cirrhosis. Cholestasis can also attenuate the endotoxin-induced hepcidin upregulation in the rats, which may explain our previous study that LPS-induced cholangitis in the jaundiced rats is associated with higher grade of liver necrosis and mortality rate.24
Cholestasis causes profound stress in humans and animals, which is associated with the induction of inflammatory cytokines, including IL-6. In our time-course study in the jaundiced rat model, there is a good temporal association of IL-6 with hepcidin mRNA expressions in both sham-operated and BDL groups. Excluding the factor of cholestasis, there was only marginal positive correlation between IL-6 and hepcidin mRNA (R2=0.072, P=0.049). However, when taking into account the factor of cholestasis, it has an important role in the regulation of hepcidin expression (P<0.001).
In the study by Graf et al22, hydrophobic bile acids compromise IL-6 signaling through both a caspase-mediated downregulation of gp130 and a p38MAPK-dependent inhibition of STAT3 phosphorylation. In this study, we unveil significant decrease of hepcidin, gp130 mRNA, pSTAT3 protein expression and pSTAT3 protein nuclear translocation in the liver of cholestatic patients and jaundiced rats compared with that of non-cholestatic patients and sham-operated rats. We further show that hydrophobic bile acids can decrease hepcidin expression in response to IL-6 stimulation through inhibiting STAT3 phosphorylation in hepatocytes. The findings are consistent with Graf et al's study and can explain our previous findings why there are higher IL-6 levels but lower hepcidin levels in the late stage of BA.11, 21 Furthermore, Huang et al26 has shown that erythropoiesis drive, even in the presence of LPS or iron-loading, downregulates hepcidin through inhibiting both inflammatory and iron-sensing pathways by the suppression of STAT3 and SMAD4 signaling in vivo. It still need more evidence to answer whether cholestasis can regulate BMP/SMAD4 or CCAAT/enhancer-binding protein pathway to modulate hepcidin expression.
Hepcidin has meaningful antimicrobial property, especially for E. coli.9 In a study of fishes' resistance to infection, Huang et al27 found that these fishes had high expression of hepcidin. Hepcidin is a cysteine-rich molecule that contains four disulfide bonds and can also inhibit the growth of Salmonella typhimurium and Mycobacterium tuberculosis.28, 29 A decrease of hepcidin in the late stage of BA indicates a loss of protection, which may account for the progressive deterioration of the patients. On the contrary, those patients with higher hepcidin levels enjoy a better outcome.11 Our clinical observation echoes Giacometti et al's30 study, in which administration of protegrin peptide IB-367—a synthetic antimicrobial peptide with cysteine-rich molecules and two disulfide bonds—can prevent endotoxin-induced mortality in the bile duct-ligated rats.
Endotoxin can stimulate expression of inflammatory cytokines, including tumor necrosis factor alpha (TNF-α), IL-1-β and IL-6.31 Nemeth et al32 have shown that hepcidin expression was induced only by IL-6, but not by IL-1 or TNF-α in human. On the contrary, Lee et al33 have shown that hepcidin transcription was stimulated in murine hepatocytes not only by IL-6 but also by IL-1-β, which reflects that there was difference between species in the regulation of hepcidin expression by inflammatory cytokines. IL-6 has been identified to be essential for the homeostasis during inflammation, because IL-6-deficient animals are more susceptible to hepatotoxic challenges, such as endotoxin.34, 35 IL-6 is also a positive inducer of hepatocyte proliferation.36 Signal transduction through gp130 is shared among the receptors for the IL-6-related cytokine subfamily. Interestingly, chronic cholestatic mice without gp130 expression results in more bacterial infection and higher mortality, which confirmed the importance of gp130 in innate immunity and IL-6 signaling.37 Intrahepatic STAT-3 activation predicted the outcome,38 and STAT3 knockout mice showed significantly higher mortality than the wide type after cecal ligation and puncture sepsis.39 Kano et al40 have shown that STAT3 signaling within endothelia has as a critical anti-inflammatory mediator against endotoxin-induced inflammation. Together, these findings suggest that maintaining the intact IL-6–gp130–phoshpoSTAT3 pathway is important for the survival of the animals and human beings. Impairment of the pathway in cholestasis may not only be associated with decreased hepatic hepcidin expression, but also with significantly higher mortality.
It has been shown that the increased deposition of iron in liver often triggers oxidative stress,41 inflammation and induces liver cell damage and cirrhosis.42 The increased hepatic stainable iron in BDL rats compared with the sham-operated rats was compatible with the downregulation of hepcidin in cholestasis. Reduction of iron load by phlebotomy or iron-deficient diet can attenuate liver injury and prolong survival in BDL rats.43, 44 Our findings of increased stainable iron in hepatitis B-induced cirrhosis compared with that in PBC are consistent with the deposition of iron, which occurs frequently in viral hepatitis, alcoholic liver injury and nonalcoholic steatohepatitis, but rarely in biliary cirrhosis.45 Lack of hepatic stainable iron in PBC and late stage of BA11 in the presence of downregulation of hepcidin expression might be a disease-specific phenomenon.
In summary, the results of this study suggest that factors such as cholestasis and hydrophobic bile acid can downregulate hepcidin expression through inhibiting IL-6 signaling and blocking pSTAT3 signal transduction. A strategy to augment hepcidin expression in the course of cholestasis may be helpful in alleviating cholestasis liver injury caused by BA and PBC, as hepcidin might be one of the final effecter molecular in defending microorganisms in both humans and animals.
References
Sokol RJ, Shepherd RW, Superina R, et al. Screening and outcomes in biliary atresia: summary of a National Institutes of Health workshop. Hepatology 2007;46:566–581.
Shih HH, Lin TM, Chuang JH, et al. Promoter polymorphism of the CD14 endotoxin receptor gene is associated with biliary atresia and idiopathic neonatal cholestasis. Pediatrics 2005;116:437–441.
Sakrak O, Akpinar M, Bedirli A, et al. Short and long-term effects of bacterial translocation due to obstructive jaundice on liver damage. Hepatogastroenterology 2003;50:1542–1546.
Chuang JH, Lee SY, Chen WJ, et al. Changes in bacterial concentration in the liver correlate with that in the hepaticojejunostomy after bile duct reconstruction: implication in the pathogenesis of postoperative cholangitis. World J Surg 2001;25:1512–1518.
Clements WD, Erwin P, McCaigue MD, et al. Conclusive evidence of endotoxaemia in biliary obstruction. Gut 1998;42:293–299.
Krause A, Neitz S, Magert HJ, et al. LEAP-1, a novel highly disulfide-bonded human peptide, exhibits antimicrobial activity. FEBS Lett 2000;480:147–150.
Ganz T . Hepcidin, a key regulator of iron metabolism and mediator of anemia of inflammation. Blood 2003;102:783–788.
Pigeon C, Ilyin G, Courselaud B, et al. A new mouse liver-specific gene, encoding a protein homologous to human antimicrobial peptide hepcidin, is overexpressed during iron overload. J Biol Chem 2001;276:7811–7819.
Park CH, Valore EV, Waring AJ, et al. Hepcidin, a urinary antimicrobial peptide synthesized in the liver. J Biol Chem 2001;276:7806–7810.
Wu ET, Chen HL, Ni YH, et al. Bacterial cholangitis in patients with biliary atresia: impact on short-term outcome. Pediatr Surg Int 2001;17:390–395.
Huang YH, Yu-Hsieh H, Huang CC, et al. Liver hepcidin and stainable iron expression in biliary atresia. Pediatr Res 2006;59:662–666.
Aoki CA, Rossaro L, Ramsamooj R, et al. Liver hepcidin mRNA correlates with iron stores, but not inflammation, in patients with chronic hepatitis C. J Clin Gastroenterol 2005;39:71–74.
Bergmann OM, Mathahs MM, Broadhurst KA, et al. Altered expression of iron regulatory genes in cirrhotic human livers: clues to the cause of hemosiderosis? Lab Invest 2008;88:1349–1357.
Jaroszewicz J, Rogalska M, Flisiak R . Serum prohepcidin reflects the degree of liver function impairment in liver cirrhosis. Biomarkers 2008;13:478–485.
Miura K, Taura K, Kodama Y, et al. Hepatitis C virus-induced oxidative stress suppresses hepcidin expression through increased histone deacetylase activity. Hepatology 2008;48:1420–1429.
Kohgo Y, Ohtake T, Ikuta K, et al. Dysregulation of systemic iron metabolism in alcoholic liver diseases. J Gastroenterol Hepatol 2008;23:S78–S81.
Pietrangelo A, Dierssen U, Valli L, et al. STAT3 is required for IL-6-gp130-dependent activation of hepcidin in vivo. Gastroenterology 2007;132:294–300.
Choi SO, Cho YS, Kim HL, et al. ROS mediate the hypoxic repression of the hepcidin gene by inhibiting C/EBPalpha and STAT-3. Biochem Biophys Res Commun 2007;356:312–317.
Milward E, Johnstone D, Trinder D, et al. The nexus of iron and inflammation in hepcidin regulation: SMADs, STATs, and ECSIT. Hepatology 2007;45:253–256.
Wang RH, Li C, Xu X, et al. A role of SMAD4 in iron metabolism through the positive regulation of hepcidin expression. Cell Metab 2005;2:399–409.
Huang YH, Chou MH, Du YY, et al. Expression of toll-like receptors and type 1 interferon specific protein MxA in biliary atresia. Lab Invest 2007;87:66–74.
Graf D, Kohlmann C, Haselow K, et al. Bile acids inhibit interleukin-6 signaling via gp130 receptor-dependent and -independent pathways in rat liver. Hepatology 2006;44:1206–1217.
Kaplan MM, Gershwin ME . Primary biliary cirrhosis. N Engl J Med 2005;353:1261–1273.
Lee CW, Chuang JH, Wang PW, et al. Effect of glucocorticoid pretreatment on oxidative liver injury and survival in jaundiced rats with endotoxin cholangitis. World J Surg 2006;30:2217–2226.
Chijiiwa K, Kozaki N, Naito T, et al. Treatment of choice for choledocholithiasis in patients with acute obstructive suppurative cholangitis and liver cirrhosis. Am J Surg 1995;170:356–360.
Huang H, Constante M, Layoun A, Santos MM . Contribution of STAT3 and SMAD4 pathways to the regulation of hepcidin by opposing stimuli. Blood 2009;9:113:3593–113:3599.
Huang PH, Chen JY, Kuo CM . Three different hepcidins from tilapia, Oreochromis mossambicus: analysis of their expressions and biological functions. Mol Immunol 2007;44:1922–1934.
Nairz M, Fritsche G, Brunner P, et al. Interferon-gamma limits the availability of iron for intramacrophage Salmonella typhimurium. Eur J Immunol 2008;38:1923–1936.
Sow FB, Florence WC, Satoskar AR, et al. Expression and localization of hepcidin in macrophages: a role in host defense against tuberculosis. J Leukoc Biol 2007;82:934–945.
Giacometti A, Cirioni O, Ghiselli R, et al. Administration of protegrin peptide IB-367 to prevent endotoxin induced mortality in bile duct ligated rats. Gut 2003;52:874–878.
Izeboud CA, Hoebe KH, Grootendorst AF, et al. Endotoxin-induced liver damage in rats is minimized by beta 2-adrenoceptor stimulation. Inflamm Res 2004;53:93–99.
Nemeth E, Valore EV, Territo M, et al. Hepcidin, a putative mediator of anemia of inflammation, is a type II acute-phase protein. Blood 2003;101:2461–2463.
Lee P, Peng H, Gelbart T, et al. Regulation of hepcidin transcription by interleukin-1 and interleukin-6. Proc Natl Acad Sci USA 2005;102:1906–1910.
Kovalovich K, DeAngelis RA, Li W, et al. Increased toxin-induced liver injury and fibrosis in interleukin-6-deficient mice. Hepatology 2000;31:149–159.
Sewnath ME, van der Poll T, van Noorden CJ, et al. Cholestatic interleukin-6-deficient mice succumb to endotoxin-induced liver injury and pulmonary inflammation. Am J Respir Crit Care Med 2004;169:413–420.
Cressman DE, Greenbaum LE, DeAngelis RA, et al. Liver failure and defective hepatocyte regeneration in interleukin-6-deficient mice. Science 1996;274:1379–1383.
Wuestefeld T, Klein C, Streetz KL, et al. Lack of gp130 expression results in more bacterial infection and higher mortality during chronic cholestasis in mice. Hepatology 2005;42:1082–1090.
Andrejko KM, Chen J, Deutschman CS . Intrahepatic STAT-3 activation and acute phase gene expression predict outcome after CLP sepsis in the rat. Am J Physiol 1998;275:G1423–G1429.
Sakamori R, Takehara T, Ohnishi C, et al. Signal transducer and activator of transcription 3 signaling within hepatocytes attenuates systemic inflammatory response and lethality in septic mice. Hepatology 2007;46:1564–1573.
Kano A, Wolfgang MJ, Gao Q, et al. Endothelial cells require STAT3 for protection against endotoxin-induced inflammation. J Exp Med 2003;198:1517–1525.
Okada S . Iron-induced tissue damage and cancer: the role of reactive oxygen species-free radicals. Pathol Int 1996;46:311–332.
Pietrangelo A . Hereditary hemochromatosis—a new look at an old disease. N Engl J Med 2004;350:2383–2397.
Otogawa K, Ogawa T, Shiga R, et al. Attenuation of acute ∣and chronic liver injury in rats by iron-deficient diet. Am J Physiol Regul Integr Comp Physiol 2008;294:311–320.
Peretz G, Link G, Pappo O, et al. Effect of hepatic iron concentration reduction on hepatic fibrosis and damage in rats with cholestatic liver disease. World J Gastroenterol 2006;12:240–245.
Ko C, Siddaiah N, Berger J, et al. Prevalence of hepatic iron overload and association with hepatocellular cancer in end-stage liver disease: results from the National Hemochromatosis Transplant Registry. Liver Int 2007;27:1394–1401.
Acknowledgements
The authors thank Ms Ming-Huei Chou for her excellent technical support and the liver transplant team of this hospital, led by Prof Chao-Long Chen. This study was supported by Chang Gung Memorial Hospital with the Grant #CMRPG860412, CMRPG850161 & CMRPG850162.
Author information
Authors and Affiliations
Corresponding author
Additional information
DISCLOSURE/CONFLICT OF INTEREST
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Huang, YH., Chuang, JH., Yang, YL. et al. Cholestasis downregulate hepcidin expression through inhibiting IL-6-induced phosphorylation of signal transducer and activator of transcription 3 signaling. Lab Invest 89, 1128–1139 (2009). https://doi.org/10.1038/labinvest.2009.82
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2009.82
Keywords
This article is cited by
-
A novel model based on immune-related genes for differentiating biliary atresia from other cholestatic diseases
Pediatric Surgery International (2022)
-
Low hepcidin in liver fibrosis and cirrhosis; a tale of progressive disorder and a case for a new biochemical marker
Molecular Medicine (2018)
-
Orphan nuclear receptor SHP regulates iron metabolism through inhibition of BMP6-mediated hepcidin expression
Scientific Reports (2016)