Abstract
Rett syndrome (RTT) is characterized by a relatively specific clinical phenotype. We screened 152 individuals with RTT phenotype. A total of 22 different known MECP2 mutations were identified in 42 subjects (27.6%). Of the 22 mutations, we identified 7 (31.8%) frameshift-causing deletions, 4 (18.2%) nonsense, 10 (45.5%) missense mutations and one insertion (4.5%). The most frequent pathologic changes were: p.Thr158Met (14.2%) and p.Arg133Cys (11.9%) missense, and p.Arg255Stop (9.5%) and p.Arg294Stop (9.5%) nonsense mutations. We also detected the c.925C>T (p.Arg309Trp) mutation in an affected patient, whose role in RTT pathogenesis is still unknown. Patients without detectable MECP2 defects were screened for mutations of cyclin-dependent kinase-like 5 (CDKL5) gene, responsible for the early-onset variant of RTT. We discovered two novel mutations: c.607G>T resulting in a termination codon at aa203, disrupting the catalytic domain, and c.1708G>T leading to a stop at aa570 of the C terminus. Both patients with CDKL5 mutation presented therapy-resistant epilepsy and a phenotype fitting with the diagnosis of early-onset variant of RTT. No FOXG1 mutation was detected in any of the remaining patients. A total of 110 (72.5%) patients remained without molecular genetic diagnosis that necessitates further search for novel gene mutations in this phenotype. Our results also suggest the need of screening for CDKL5 mutations in patients with Rett phenotype tested negative for MECP2 mutations.
Similar content being viewed by others
Introduction
Rett syndrome (RTT, OMIM 312750) is a severe neurodevelopmental disorder affecting females almost exclusively. The classic form of Rett is characterized by a period of normal development until around 6–18 months of age, followed by a rapid regression of acquired skills, loss of speech, stereotypical hand movements, microcephaly, seizures, autism, ataxia and intermittent hyperventilation.1, 2 Besides the classic form, many variants are known, such as the congenital variant that can be recognized at birth, the preserved speech variant with a partial recovery of speech, the ‘forme fruste’ characterized by a milder clinical course and late childhood regression, and the ‘early seizure type’ or Hanefeld syndrome.3, 4
Classic RTT shows an X-linked dominant pattern of inheritance and has a relatively high incidence of ∼1 in 10 000 among females.5, 6, 7 Mutations in the methyl-CpG-binding protein, MECP2 gene, were identified as disease causing,8 and according to data published, MECP2 mutations account for ∼80–96% of the classical Rett cases9, 10 and 40–50% of atypical Rett manifestations.11 The MECP2 gene is located at Xq28 and therefore it is subject to X-chromosome inactivation.12 It comprises four exons and encodes two alternatively spliced isoforms: the 498 amino-acid MeCP2E1 encompassing exons 1, 3 and 4, and MeCP2E2 of 486 residues comprising exons 2, 3 and 4.9, 13, 14 The MeCP2 protein acts as a DNA methylation-dependent transcriptional repressor.15
The lack of MECP2 mutation in a small percentage of clinically well-defined RTT patients with infantile spasms suggested the involvement of another gene locus. Indeed, mutations of the cyclin-dependent kinase-like 5 (CDKL5 or serine–threonine kinase 9) gene were identified in patients with early seizure variant of RTT.4, 5 The CDKL5 gene encodes a 1030 amino-acid protein with a highly conserved N-terminal serine–threonine kinase domain.16 Supporting the role of CDKL5 mutations in RTT, recent evidence shows that CDKL5 and MeCP2 belong to the same molecular pathway, and CDKL5 is capable of interacting with and phosphorylating MeCP2.17
In addition, FOXG1 gene mutations were identified in two unrelated girls with a congenital variant of RTT, as these girls had infantile onset of microcephaly, mental retardation and peculiar jerky movements similar to that observed in classic RTT.18 FOXG1 encodes the forkhead box protein G1, a transcriptional factor expressed exclusively in fetal and adult brain, and testis. FoxG1, via its interactions with the transcriptional repressor JARID1B and with corepressors of the Groucho family, plays an important role in early brain development. Furthermore, FoxG1 indirectly associates with the histone deacetylase 1 protein similarly to MeCP2.19
Here we report a three-stage mutation screening of 152 patients with a diagnosis of RTT.
Materials and methods
All DNA samples were from the central Biobank of the University of Pécs, registered at the National Biobank Network of Hungary (http://www.biobanks.hu). The Biobank obtained ethical approval from the National Scientific Research Ethics Committee (ETT TUKEB). During the collection and use of DNA samples and the accompanying clinical and personal data, the guidelines and regulations of the Helsinki Declaration in 1964, and the currently operative national laws and regulations were followed.
Blood DNA samples were collected from 152 patients with RTT and related individuals (parents or siblings) after informed consent. All of the subjects were originally recruited from patients who had been referred to our institute for MECP2 testing. Genomic DNA was extracted from peripheral blood leukocytes by a standard desalting method. In the first stage of the screening, we performed mutation analysis of MECP2 by direct sequencing. Exons 2–4 of MECP2 were amplified by PCR. Primers were designed by our laboratory. Primer sequences and PCR conditions are available upon request. The PCR products were then purified, followed by sequencing on both strands. Sequencing was performed with the same primers as the PCR using a BigDye Terminator v1.1 Cycle Sequencing Kit (Applied Biosystems, Foster City, CA, USA) according to the manufacturer's instructions.
In the second stage of screening, all those patients negative for a detectable MECP2 mutation were selected and screened for mutations of the CDKL5 gene by direct sequencing. The CDKL5-coding region was analyzed using the primers described by Scala et al.4 with some exceptions, as primers were redesigned if they were positioned less than 50 bp from the exon. Primer sequences are available upon request. Following PCR amplification, the fragments were submitted to direct sequencing as above.
Similarly, patients with no detected MECP2 or CDKL5 mutations were tested for FOXG1 mutations using the primer sequences published by Ariani et al.18 Specific genetic and clinical data of these patients were inserted in Rett Network Database: (http://www.rettdatabasenetwork.org). This database collects data worldwide in Rett syndrome according to OECD Guidelines on Human Biobanks and Genetic Research Databases.
Results
A total of 22 different known MECP2 alterations were identified in 42 subjects. Out of the 22 mutations, we identified that 7 (31.8%) were deletions leading to frameshift, 4 (18.2%) were nonsense mutations and 10 (45.4%) were missense mutations (Table 1). We also detected a single-nucleotide insertion. Among the pathologic mutations, the most frequent was the missense mutation p.Thr158Met (14.2%), whereas the missense mutation p.Arg133Cys (11.9%) was the second and nonsense mutations p.Arg255Stop (9.5%) and p.Arg294Stop (9.5%) were the third most frequent sequence changes, respectively.
We also detected a missense c.925C>T (p.Arg309Trp) change, originally described in a 7-year-old boy with absence of speech and psychomotor delay;20 however, the role of this alteration in Rett pathogenesis is still not clarified (database RIMV: http://www.mecp2.chw.edu.au/; database HGM: http://www.hgmd.org).20
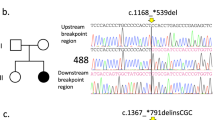
We identified an 18 bp deletion c.1162_1179del18 in a father and his daughter (R/34). As this deletion is present in the healthy father and the daughter also carried the frameshift-resulting insertion c.276_277insG, we hypothesized that this alteration is not the pathogenic mutation responsible for the Rett phenotype. However, it is important to note that data available at the RettBASE: IRSF MECP2 Variation Database indicates that it is unknown whether this deletion can be considered as a mutation or as a polymorphism. In addition, we detected a silent polymorphism, the c.753C>T transition, in a mother and her daughter (data not shown).
Following the MECP2 mutation screening, we tested all patients without any MECP2 alteration for CDKL5 mutations. During the direct sequencing, we have identified previously described polymorphism c.2372A>C (p.Gln791Pro) in eight subjects as well as both the c.3003C>T (p.His1001His) and c.3084G>C (p.Thr1028Thr) known polymorphisms in one of the patients (ref. seq.: NM_001037343.1). Furthermore, we discovered two novel nonsense mutations: in the first case, a c.607G>T change that results in a premature termination codon at amino-acid position 203, disrupting the catalytic domain of the CDKL5 protein, and c.1708G>T in a second patient that leads to a premature stop at amino-acid position 570 of the C-terminal region involved in either the catalytic activity or the subcellular localization (Table 2).
The first presented case affected by the early seizure variant of RTT is a female patient born after an uneventful pregnancy at the 39th gestation week with age-appropriate anthropometric parameters by an uncomplicated vaginal delivery to non-consanguineous parents. Postnatal adaptation was normal, and in the pedigree, there was nothing noticeable to be observed. The first epileptic fit occurred at the age of 3 weeks and since then she has experienced extremely frequent seizures, despite of the administration of several antiepileptic drug combinations and even callosotomy. Stereotypic hand-washing activities could be observed from the age of 4. At the age of 8 years, her weight, height and occipital-frontal circumference were normal for her age, but she had severe psychomotor retardation and hypotrophy, while hypotonic muscle and ataxia could also be observed. In her dysmorphic status, face asymmetry, deep-set eyes, high palate, gingival hypertrophy, smaller second incisor and small hands and feet for her age could be detected. At the age of 13 years, her dysmorphic features have been the same; poor eye contact, stereotypic hand movement and severe refractory epilepsy with very frequent seizures with microcephaly were still present. In her long-term psychomotor development, very slow progression was detected.
The second early seizure variant case is a female baby, 7 months old at the time of examination. She had been born as a second child of non-consanguineous healthy parents after an uneventful gravidity on the 39th week of gestation by cesarean section with 3650 g birth weight. The postnatal period had been without any complication. Apart from a miscarriage of the mother, the family history was negative. At the fifth week of age, the patient developed infantile spasms. She had been treated with several drug combinations in a neurology department and she had been temporarily seizure free for 4–6 weeks. At the time of examination, under levetiracetam and carbamazepine therapy, she had epileptic fits with weekly frequency, but later, the seizures became more recurrent again. Brain magnetic resonance imaging was considered normal. At that time, the patient weighed 8180 g (50–75th percentile), with length of 71 cm (90th percentile) and head circumference of 42.5 cm (10th percentile). In her dysmorphic status, occipital asymmetry, flat nasal bridge, down slanting palpebral fissures, hypertelorism, high palate, gingival hypertrophy and wide-spaced nipples were present. Neurological examination revealed gross motor hypotonia and poor eye contact.
Finally, we screened our patients for FOXG1 mutations using direct sequencing, but we did not find any alterations affecting this gene.
Discussion
Following the discovery of MECP2 gene defects in RTT patients, a wide spectrum of mutations has been identified in a high percentage of affected individuals. According to data in the literature, common point mutations of MECP2 (R106W, R133C, R168X, R255X, R270X, R294X, R306C and T158M) are present in ∼60–70% of cases with pathogenic MECP2 mutations.21, 22, 23 Interestingly, we have only detected MECP2 mutations in 27.6% of the Rett patients studied, a ratio that is likely not only because of the limitations of direct sequencing method used for mutation analysis, but possibly attributable to a high percentage of gross rearrangements of the MECP2 gene that are not detectable by sequencing. Such rearrangements may account for 37.8% of classic RTT and 7.5% of atypical RTT patients according to recent literature.24
It is important to point out that among the MECP2-negative patients only 17% showed classical Rett phenotype. In all, 8% of these patients showed the features of congenital form of RTT, 12% were male young patients and 4% have no neurological symptom. In the MECP2-positive group, slow psychomotor development could be observed in 51%, whereas in the MECP2-negative group, its incidence was 71.25%. In contrast, microcephaly was present in 74.4% of the MECP2-positive patients and only in 41% of the MECP2-negative patients. Throughout the period of the observation performed, 17.8% of these female young patients presented any dysmorphic sign. In the MECP2-positive group, 4.65% of the patients had the evidence for a mild phenotype.
Recently, a novel locus of chromosome X, CDKL5, was implicated in the atypical form of RTT4, 5 supporting the notion of genetic heterogeneity in RTT. In addition, the FOXG1 gene was recently implicated in the molecular etiology of the congenital variant of RTT. We report the molecular screening of 152 individuals with the diagnosis of RTT and 11 of their relatives. Among the subjects screened, we identified 42 RTT patients with a pathologic MECP2 mutation and 2 patients with novel CDKL5 mutation, but we did not find any alterations affecting the FOXG1 gene, that may be because of overall low frequency of FOXG1 mutations in patients with Rett phenotype.
Although the two novel CDKL5 mutations identified result in an early stop at codons 203 and 570, further studies are needed to determine the effect of these mutations resulting in truncated protein products; especially because the p.Glu203Stop mutation affects the catalytic domain of CDKL5 and it may influence its enzymatic activity.
Mutations affecting the CDKL5 gene appear to have clinical outcomes of varying severity. So far, 64 mutations (19 of which are publicly available) of the gene have been deposited to the HGMD database (http://www.hgmd.org), which include missense mutations of the catalytic domain, deletions, insertions or nonsense mutations along the open reading frame causing a premature termination, as well as large genomic rearrangements leading to the deletion of the entire gene locus. Owing to the limited number of patients with CDKL5 mutations, and the limited knowledge regarding the function and regulation of the protein, establishing genotype–phenotype correlations is rather difficult.
To date, the genotype–phenotype correlation is well known and is commonly examined in RTT caused by mutations in MECP2; however, such correlations of CDKL5 mutations in RTT, although widely recognized, are still not so well branded.
Zappella et al.25 noted that early truncating mutations of MECP2 lead to a poor prognosis (classic RTT), whereas late truncating missense mutations lead either to classic RTT or to preserved speech variant. Early truncating mutations were associated with a more severe course of the disorder.26 A large MECP2 screening project in patients diagnosed with RTT was also published,27 where composite phenotype severity scores did not correlate with mutation type, domain affected or X inactivation. Other correlations suggested that truncating mutations and mutations affecting the methyl-CpG-binding-domain tend to lead to a more severe phenotype. In a study of genotype/phenotype correlations, 85 RTT patients were analyzed for mutations in the MECP2 gene.28 They found that patients with missense mutations had lower total severity scores and better language performance than those with nonsense mutations.
A recent publication characterized the epilepsy associated with CDKL5 mutations and looked for a relationship between the genotype and the course of epilepsy,29 whereas a comprehensive survey of the key clinical features of CDKL5-positive RTT was also published.30 Artuso et al. reported a detailed clinical investigation of nine patients with CDKL5 mutations to define the clinical diagnostic criteria, and in addition, they reviewed the clinical phenotype of all patients with a CDKL5 point mutation reported so far.31 In agreement with their findings, our patients with CDKL5 mutations after a normal prenatal history had seizures with an early onset, which appears to be a key feature in the identification of patients with CDKL5 gene defects. Furthermore, these seizures of our patients proved to be therapy resistant, a common characteristic of all CDKL5-mutated cases. In our patients, hypotonia, one of the necessary diagnostic criteria as suggested by most recent literature, was also present referring to a CDKL5 mutation.31
References
Hagberg, B. Clinical manifestations and stages of Rett syndrome. Ment. Retard. Dev. Disabil. Res. Rev. 8, 61–65 (2002).
Weaving, L., Ellaway, C., Gécz, J. & Christodoulou, J. Rett syndrome: clinical review and genetic update. J. Med. Genet. 42, 1–7 (2005).
Nemos, C., Lambert, L., Giuliano, F., Doray, B., Roubertie, A., Goldenberg, A. et al. Mutational spectrum of CDKL5 in early-onset encephalopathies: a study of a large collection of French patients and review of the literature. Clin. Genet. 76, 357–371 (2009).
Scala, E., Ariani, F., Mari, F., Caselli, R., Pescucci, C., Longo, I. et al. CDKL5/STK9 is mutated in Rett syndrome variant with infantile spasms. J. Med. Genet. 42, 103–107 (2005).
Weaving, L., Christodoulou, J., Williamson, S., Friend, K. L., McKenzie, O. L., Archer, H. et al. Mutations of CDKL5 cause a severe neurodevelopmental disorder with infantile spasms and mental retardation. Am. J. Hum. Genet. 75, 1079–1093 (2004).
Terai, K., Munesue, T., Hiratani, M., Jiang, Z., Jibiki, I. & Yamaguchi, N. The prevalence of Rett syndrome in Fukui prefecture. Brain Dev. 17, 153–154 (1995).
Skjeldal, O., von Tetzchner, S., Aspelund, F., Herder, G. & Lofterłd, B. Rett syndrome: geographic variation in prevalence in Norway. Brain Dev. 19, 258–261 (1997).
Amir, R., Van den Veyver, I., Wan, M., Tran, C., Francke, U. & Zoghbi, H. Rett syndrome is caused by mutations in X-linked MECP2, encoding methyl-CpG-binding protein 2. Nat. Genet. 23, 185–188 (1999).
Moretti, P. & Zoghbi, H. MeCP2 dysfunction in Rett syndrome and related disorders. Curr. Opin. Genet. Dev. 16, 276–281 (2006).
Kriaucionis, S. & Bird, A. DNA methylation and Rett syndrome. Hum. Mol. Genet. 12 (Spec No 2), R221–R227 (2003).
Kammoun, F., de Roux, N., Boespflug-Tanguy, O., Vallée, L., Seng, R., Tardieu, M. et al. Screening of MECP2 coding sequence in patients with phenotypes of decreasing likelihood for Rett syndrome: a cohort of 171 cases. J. Med. Genet. 41, e85 (2004).
Adler, D., Quaderi, N., Brown, S., Chapman, V. M., Moore, J., Tate, P. et al. The X-linked methylated DNA binding protein, Mecp2, is subject to X inactivation in the mouse. Mamm. Genome 6, 491–492 (1995).
Kriaucionis, S. & Bird, A. The major form of MeCP2 has a novel N-terminus generated by alternative splicing. Nucleic Acids Res. 32, 1818–1823 (2004).
Mnatzakanian, G., Lohi, H., Munteanu, I., Alfred, S. E., Yamada, T., MacLeod, P. J. et al. A previously unidentified MECP2 open reading frame defines a new protein isoform relevant to Rett syndrome. Nat. Genet. 36, 339–341 (2004).
Kaludov, N. & Wolffe, A. MeCP2 driven transcriptional repression in vitro: selectivity for methylated DNA, action at a distance and contacts with the basal transcription machinery. Nucleic Acids Res. 28, 1921–1928 (2000).
Montini, E., Andolfi, G., Caruso, A., Buchner, G., Walpole, S. M., Mariani, M. et al. Identification and characterization of a novel serine-threonine kinase gene from the Xp22 region. Genomics 51, 427–433 (1998).
Mari, F., Azimonti, S., Bertani, I., Bolognese, F., Colombo, E., Caselli, R. et al. CDKL5 belongs to the same molecular pathway of MeCP2 and it is responsible for the early-onset seizure variant of Rett syndrome. Hum. Mol. Genet. 14, 1935–1946 (2005).
Ariani, F., Hayek, G., Rondinella, D., Artuso, R., Mencarelli, M. A., Spanhol-Rosseto, A. et al. FOXG1 is responsible for the congenital variant of Rett syndrome. Am. J. Hum. Genet. 83, 89–93 (2008).
Tan, K., Shaw, A., Madsen, B., Jensen, K., Taylor-Papadimitriou, J. & Freemont, P. Human PLU-1 has transcriptional repression properties and interacts with the developmental transcription factors BF-1 and PAX9. J. Biol. Chem. 278, 20507–20513 (2003).
Campos, M. J., Abdalla, C., Santos-Rebouças, C., dos Santos, A. V., Pestana, C. P., Domingues, M. L. et al. Low significance of MECP2 mutations as a cause of mental retardation in Brazilian males. Brain Dev. 29, 293–297 (2007).
Bebbington, A., Percy, A., Christodoulou, J., Ravine, D., Ho, G., Jacoby, P. et al. Updating the profile of C-terminal MECP2 deletions in Rett syndrome. J. Med. Genet. 47, 242–248 (2010).
Colvin, L., Leonard, H., de Klerk, N., Davis, M., Weaving, L., Williamson, S. et al. Refining the phenotype of common mutations in Rett syndrome. J. Med. Genet. 41, 25–30 (2004).
Percy, A., Lane, J., Childers, J., Skinner, S., Annese, F., Barrish, J. et al. Rett syndrome: North American database. J. Child Neurol. 22, 1338–1341 (2007).
Archer, H., Whatley, S., Evans, J., Ravine, D., Huppke, P., Kerr, A. et al. Gross rearrangements of the MECP2 gene are found in both classical and atypical Rett syndrome patients. J. Med. Genet. 43, 451–456 (2006).
Zappella, M., Meloni, I., Longo, I., Hayek, G. & Renieri, A. Preserved speech variants of the Rett syndrome: molecular and clinical analysis. Am. J. Med. Genet. 104, 14–22 (2001).
Smeets, E., Schollen, E., Moog, U., Matthijs, G., Herbergs, J., Smeets, H. et al. Rett syndrome in adolescent and adult females: clinical and molecular genetic findings. Am. J. Med. Genet. A 122A, 227–233 (2003).
Weaving, L., Williamson, S., Bennetts, B., Davis, M., Ellaway, C. J., Leonard, H. et al. Effects of MECP2 mutation type, location and X-inactivation in modulating Rett syndrome phenotype. Am. J. Med. Genet. A 118A, 103–114 (2003).
Schanen, C., Houwink, E., Dorrani, N., Lane, J., Everett, R., Feng, A. et al. Phenotypic manifestations of MECP2 mutations in classical and atypical Rett syndrome. Am. J. Med. Genet. A 126A, 129–140 (2004).
Bahi-Buisson, N., Kaminska, A., Boddaert, N., Rio, M., Afenjar, A., Gérard, M. et al. The three stages of epilepsy in patients with CDKL5 mutations. Epilepsia 49, 1027–1037 (2008).
Bahi-Buisson, N., Nectoux, J., Rosas-Vargas, H., Milh, M., Boddaert, N., Girard, B. et al. Key clinical features to identify girls with CDKL5 mutations. Brain 131, 2647–2661 (2008).
Artuso, R., Mencarelli, M., Polli, R., Sartori, S., Ariani, F., Pollazzon, M. et al. Early-onset seizure variant of Rett syndrome: definition of the clinical diagnostic criteria. Brain Dev. 32, 17–24 (2010).
Buyse, I. M., Fang, P., Hoon, K. T., Amir, R. E., Zoghbi, H. Y. & Roa, B. B. Diagnostic testing for Rett syndrome by DHPLC and direct sequencing analysis of the MECP2 gene: identification of several novel mutations and polymorphisms. Am. J. Hum. Genet. 67, 1428–1436 (2000).
Amano, K., Nomura, Y., Segawa, M. & Yamakawa, K. Mutational analysis of the MECP2 gene in Japanese patients with Rett syndrome. J. Hum. Genet. 45, 231–236 (2000).
Kárteszi, J., Hollódy, K., Bene, J., Morava, E., Hadzsiev, K., Czakó, M. et al. Mutation analysis of MECP2 and determination of the X-inactivation pattern in Hungarian Rett syndrome patients. Am. J. Med. Genet. A. 131, 106 (2004).
Bienvenu, T., Villard, L., De Roux, N., Bourdon, V., Fontes, M., Beldjord, C. et al. Spectrum of MECP2 mutations in Rett syndrome. Genet. Test 6, 1–6 (2002).
De Bona, C, Zappella, M, Hayek, G, Meloni, I, Vitelli, F, Bruttini, M et al. Preserved speech variant is allelic of classic Rett syndrome. Eur. J. Hum. Genet. 8, 325–330 (2000).
Philippe, C., Villard, L., De Roux, N., Raynaud, M., Bonnefond, J. P., Pasquier, L. et al. Spectrum and distribution of MECP2 mutations in 424 Rett syndrome patients: a molecular update. Eur. J. Med. Genet. 49, 9–18 (2006).
Christodoulou, J., Grimm, A., Maher, T. & Bennetts, B. RettBASE: The IRSA MECP2 variation database-a new mutation database in evolution. Hum. Mutat. 21, 466–472 (2003).
Hoffbuhr, K., Devaney, J. M., LaFleur, B., Sirianni, N., Scacheri, C., Giron, J. et al. MeCP2 mutations in children with and without the phenotype of Rett syndrome. Neurology 56, 1486–1495 (2001).
Orrico, A., Lam, C., Galli, L., Dotti, M. T., Hayek, G., Tong, S. F. et al. MECP2 mutation in male patients with non-specific X-linked mental retardation. FEBS Lett. 481, 285–288 (2000).
Fukuda, T., Yamashita, Y., Nagamitsu, S., Miyamoto, K., Jin, J. J., Ohmori, I. et al. Methyl-CpG binding protein 2 gene (MECP2) variations in Japanese patients with Rett syndrome: pathological mutations and polymorphisms. Brain Dev. 27, 211–217 (2005).
Budden, S. S., Dorsey, H. C. & Steiner, R. D. Clinical profile of a male with Rett syndrome. Brain Dev. 27 (Suppl 1), S69–S71 (2005).
Cheadle, J. P., Gill, H., Fleming, N., Maynard, J., Kerr, A., Leonard, H. et al. Long-read sequence analysis of the MECP2 gene in Rett syndrome patients: correlation of disease severity with mutation type and location. Hum. Mol. Genet. 9, 1119–1129 (2000).
Chae, J. H., Hwang, H., Hwang, Y. S., Cheong, H. J. & Kim, K. J. Influence of MECP2 gene mutation and X-chromosome inactivation on the Rett syndrome phenotype. J. Child Neurol. 19, 503–508 (2004).
Huppke, P., Laccone, F., Krämer, N., Engel, W. & Hanefeld, F. Rett syndrome: analysis of MECP2 and clinical characterization of 31 patients. Hum. Mol. Genet. 9, 1369–1375 (2000).
Zahorakova, D., Rosipal, R., Hadac, J., Zumrova, A., Bzduch, V., Misovicova, N. et al. Mutation analysis of the MECP2 gene in patients of Slavic origin with Rett syndrome: novel mutations and polymorphisms. J. Hum. Genet. 52, 342–348 (2007).
Hampson, K., Woods, C. G., Latif, F. & Webb, T. Mutations in the MECP2 gene in a cohort of girls with Rett syndrome. J. Med. Genet. 37, 610–612 (2000).
Donzel-Javouhey, A., Thauvin-Robinet, C., Cusin, V., Madinier, N., Manceau, E., Dipanda, D. et al. A new cohort of MECP2 mutation screening in unexplained mental retardation: careful re-evaluation is the best indicator for molecular diagnosis. Am. J. Med. Genet. A. 140, 1603–1607 (2006).
Tao, J., Van Esch, H., Hagedorn-Greiwe, M., Hoffmann, K., Moser, B., Raynaud, M. et al. Mutations in the X-linked cyclin-dependent kinase-like 5 (CDKL5/STK9) gene are associated with severe neurodevelopmental retardation. Am. J. Hum. Genet. 75, 1149–1154 (2004).
Acknowledgements
This work was supported by the grant of Hungarian Science Foundation OTKA T 73430. Noemi Polgar was supported by the Bolyai Foundation of the Hungarian Academy of Sciences.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Hadzsiev, K., Polgar, N., Bene, J. et al. Analysis of Hungarian patients with Rett syndrome phenotype for MECP2, CDKL5 and FOXG1 gene mutations. J Hum Genet 56, 183–187 (2011). https://doi.org/10.1038/jhg.2010.156
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jhg.2010.156
Keywords
This article is cited by
-
Genetics, Molecular Biology, and Phenotypes of X-Linked Epilepsy
Molecular Neurobiology (2014)
-
The CDKL5 disorder is an independent clinical entity associated with early-onset encephalopathy
European Journal of Human Genetics (2013)
-
Novel Mutations in Cyclin-Dependent Kinase-Like 5 (CDKL5) Gene in Indian Cases of Rett Syndrome
NeuroMolecular Medicine (2013)