Abstract
Gene therapy using cDNA driven by an exogenous promoter is not suited for genetic disorders that require intrinsic expression of a transgene, such as hyperimmunoglobulin (Ig)M syndrome (HIGM), which is caused by mutations in the CD40L gene. The human artificial chromosome (HAC) vector has the potential to solve this problem, because it can be used to transfer large genomic fragments containing their own regulatory elements. In this study, we examined whether introduction of a genomic fragment of CD40L via the HAC vector permits intrinsic expression of the transgene and has an effect on immunoglobulin secretion. We constructed an HAC vector carrying the mouse CD40L genomic fragment (mCD40L-HAC) in Chinese hamster ovary (CHO) cells and transferred the mCD40L-HAC vector into a human CD4-positive active T-cell line (Jurkat) and a human myeloid cell line (U937) via microcell-mediated chromosome transfer (MMCT). The mCD40L-HAC vector permits mCD40L expression in human active T cells but not in human myeloid cells. The mCD40L-HAC also functions to stimulate mouse B cells derived from CD40L(-/-) mice, inducing secretion of IgG. This study may be an initial step toward the therapeutic application of HAC vectors for intrinsic expression of genes, a potential new direction for genome-based gene therapy.
Similar content being viewed by others
Introduction
Gene therapy for treating genetic disorders has recently become a major research focus. Transgene-based gene therapy, in which a viral vector carrying cDNA is used, is a commonly used approach that has been successful in correcting X-linked severe combined immunodeficiency (SCID) (Cavazzana-Calvo et al. 2000; Hacein-Bey-Abina et al. 2002; Gaspar et al. 2003) and SCID due to a deficiency of the adenosine deaminase enzyme (Blaese et al. 1995; Onodera et al. 1998; Muul et al. 2003). However, this type of gene therapy is not suited to treating genetic disorders caused by deficiency in a gene that requires intrinsic regulation. In this case, it would be ideal to introduce a genomic fragment containing its own regulatory element. For example, hyperimmunoglobulin (Ig)M syndrome (HIGM) is a genetic disorder caused by deficiency in a gene encoding the CD40 ligand, which requires intrinsic regulation.
HIGM is a rare, X-linked, recessive immunodeficiency disease characterized by the failure of IgM to undergo immunoglobulin-isotype switching to IgG, IgA, and IgE (Kroczek et al. 1994). The CD40 ligand is a cell-surface ligand expressed by CD4+ T cells, the expression of which is regulated by cell activation (DiSanto et al. 1993; Korthauer et al. 1993; Roy et al. 1993). Functionally, CD40L gene mutation results in dysfunctional interactions between CD4+ T cells and the CD40 glycoprotein on the surfaces of B cells, macrophages, and dendritic cells (Stout et al. 1996; Grewal and Flavell 1998; Straw et al. 2003). Gene therapy for this condition was demonstrated to be feasible by Brown et al. (1998) using ex vivo retrovirus-mediated gene transfer of cDNA to bone marrow cells in a mouse CD40L-knockout model. Although this approach corrected the CD40L deficiency and stimulated humoral and cellular immune functions, the mice developed T-lymphoproliferative disease. This suggests that nonintrinsic overexpression of CD40L may cause abnormal proliferative responses, resulting in T-lymphoproliferative disease. Thus, an alternative gene-delivery system that permits regulated transgene expression is required for HIGM gene therapy (Brown et al. 1998).
As described elsewhere, we recently developed a novel human artificial chromosome (HAC) vector (Katoh et al. 2004). The HAC vector has several advantages over conventional vectors such as viruses and plasmids: there is no limitation on the size of DNA fragment that can be inserted, the HAC is independently and stably maintained, and the copy number is strictly controlled. Thus, the HAC vector containing a genomic fragment has the potential to permit intrinsic expression of transgenes (Grimes et al. 2002; Larin and Mejia 2002; Saffery and Choo 2002)
In this study, we used an HAC vector to carry a CD40L genomic fragment and examined cell-type-specific gene expression and the function of the exogenous CD40L gene.
Materials and methods
Cell culture
Chinese hamster ovary (CHO) cells were maintained in Ham’s F-12 nutrient mixture (Wako) containing 10% FBS (FBS) (Bio West). A human CD4-positive active T-cell line (Jurkat) and a human myeloid cell line (U937) were maintained in Roswell Park Memorial Institute (RPMI)-1640 (Wako) containing 10% FBS (Bio West). We used these human cell lines to analyze expression of the mCD40L gene on the HAC vector for the following reasons. First, the cells used in this study must continue to proliferate over the long term, long enough to obtain clones containing HAC by microcell-mediated chromosome transfer (MMCT) and to carry out further analyses. Second, expression of the mCD40L gene on the HAC vector must be discriminated from expression of the endogenous CD40L gene in cells. Third, since human CD40L does not bind to mouse CD40 (Bossen et al. 2006), using human cell lines as hosts meant that we could study the function of the mCD40L genomic fragment introduced via the HAC vector. A total of 3 × 105 B cells that had been obtained from the spleens of CD40L -/- mice (Jackson Laboratory) were cultured with the same number of Jurkat-mCD40L-HAC cells. These cells were treated with mitomycin C (Sigma) in RPMI-1640 containing 10% FBS (Bio West), 0.05 mM 2-ME (Sigma), 500 U/ml mrIL-4 (R&D Systems) and 150 U/ml mrIL-5 (R&D Systems).
Subcloning the mouse CD40L genome into a PAC vector
A bacterial artificial chromosome (BAC) clone, RP23-153G22 (BACPAC Resource Center, Oakland, CA, USA), was used to clone a mouse CD40L genomic fragment (21,342 bp) into a pPAC4 vector carrying a loxP-site and the cytomegalovirus (CMV) promoter. The gap repair cassette DNA was amplified by polymerase chain reaction (PCR) using the primers 5′-AAGGATCCCACTTAAACAGAAGCCACACCCAGCTCTTTCATTGCAGAGAGATGTCTTCGAGCTCTTGGGGGA-3′ and 5′-TTGGATCCGGTTGAGGTTGGAAATTTTAGCTAGTCCACTCGCATCAGCCTTCCCCCAAGAGCTCGAAGAC-3′ and cloned into the BamHI site of pPAC4 to create a subcloning vector. The subcloning vector contained 5′ and 3′ regions that were homologous to those of the mouse CD40L genome. A homologous recombination-proficient Escherichia coli strain, DY380 (kindly provided by Dr. Donald Court, National Cancer Institute, Frederick, MD, USA), was used for the BAC engineering (Lee et al. 2001).
Construction of an HAC vector carrying the mouse CD40L genomic fragment in CHO cells
Twelve micrograms of the PAC vector containing the mouse CD40L genome and 0.3 μg of the Cre expression vector pBS185 (Life Technologies) were transformed into 5 × 105 CHO cells containing the 21Δpq HAC vector (Katoh et al. 2004) using Lipofectamine 2000 (Invitrogen), and transformed cells were selected using G418 (Invitrogen). G418-resistant colonies were isolated, and insertion of the mCD40L genomic fragment into the loxP site of the HAC vector was confirmed by genomic PCR with three primer pairs, as follows (Fig. 1b): 1F, 5′-GCTCAAGATGCCCCTGTTCT-3′ and 1R, 5′-GAGGGCACCACACAAAAAGT-3′; 2F, 5′-GGCGGCTCTAGAGGATCTG-3′ and 2R, 5′-CAGGTCGGTCTTGACAAAAAG-3′; 3F, 5′-AGAGAATTACAGGGCAGGCA-3′ and 3R, 5′-TTGAGCTGAGGCTCACAGTTG-3′.
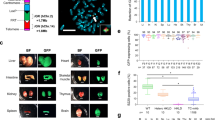
A flowchart of this study and a schematic diagram of the introduction of the mouse CD40L genomic fragment (21,342 bp) into the loxP site of the human artificial chromosome (HAC) vector. a Configuration of mouse CD40L gene. “+1” corresponds to the transcription start site. The 5′ and 3′ untranslated regions are indicated by lighter-color boxes. We referred to a previous report (Cron 2002) to draw this diagram. b A flowchart of this study: A circular targeting construct carrying the mouse CD40L genomic fragment and the nonfunctional loxP-neo target on the HAC vector is shown. The Cre recombinase-catalyzed integration regenerates a functional neogene. The resulting inserted allele is shown at the bottom. Arrowheads indicate the genomic polymerase chain reaction (PCR) primers used for detection of the site-specific insertion. c The site-specific insertion event was detected by genomic PCR. The numbers 1–3 correspond to the number of the primer pair used. The 100-bp DNA ladderTM (TaKaRa) was used as a size marker. d Fluorescent in situ hybridization (FISH) analysis of the mCD40L-HAC vector in Chinese hamster ovary (CHO) cells: a typical metaphase FISH image of CHO cells carrying the mCD40L-HAC vector. Arrowheads indicate the two-colored mCD40L-HAC vector in metaphase and interphase cells. Cells were probed with human cot-1 DNA (red) and mouse CD40L genomic DNA (green)
Fluorescence in situ hybridization (FISH) analysis
Metaphase chromosomes were prepared from exponentially growing cell cultures, and fluorescence in situ hybridization (FISH) was carried out using standard protocols. The probes used were digoxigenin-labeled human Cot1 DNA (Invitrogen; digoxigenin from Roche), biotin-labeled mouse CD40L DNA (biotin from Roche), and digoxigenin-labeled hchr21 alphoid DNA. The digoxigenin signal was detected using an anti-digoxigenin-rhodamine complex (Roche), and the biotin signal was detected using an avidin-fluorescein isothiocyanate (adivin-FITC) (Roche) complex. The chromosomes were counterstained with 4',6-diamidino-2-phenylindole (DAPI) (Sigma). Images were captured using an optical microscope (Nikon) equipped with a photometric charge-coupled device (CCD) camera and visualized using the Argus system (Hamamatsu Photonics).
Microcell-mediated chromosome transfer (MMCT)
The mCD40L-HAC vector remaining in the CHO cells was transferred to 1 × 107 Jurkat or U937 cells via microcell-mediated chromosome transfer (MMCT), as previously described (Doherty and Fisher 2003; Katoh et al. 2004; Kakeda et al. 2005).
Flow cytometry analysis
Cells carrying mCD40L-HAC were stained with FITC-labeled anti-mouse-CD154 (Santa Cruz) and PE-labeled anti-human-CD154 (BioLegend) and then analyzed by flow cytometry (Beckman Coulter).
Reverse transcriptase polymerase chain reaction (RT-PCR) analysis
Expression of the mCD40L gene on the HAC vector in human cells was detected by reverse transcriptase PCR (RT-PCR) with primer pairs: F, 5′-AGAGAATTACAGGGCAGGCA-3′ and R, 5′-TTGAGCTGAGGCTCACAGTTG-3′. These primer pairs detect a 3′UTR region of mCD40L.
Enzyme-linked immunosorbent assay (ELISA)
B cells and Jurkat-mCD40L-HAC cells were cocultured for 7 days, then the concentration of mouse IgG secreted into the culture medium was analyzed using enzyme-linked immunosorbent assay (ELISA) with horseradish-peroxidase-labeled anti-mouse-IgG (Amersham Biosciences).
Results
Construction of the mCD40L-HAC vector in CHO cells
The mouse gene encoding the CD40 ligand was obtained together with a 4-kb upstream and a 5-kb downstream DNA sequence. The gene was cloned by gap repair from BAC clone RP23-153G22 into a pPAC4 vector carrying a loxP sequence and the CMV promoter. This promoter drove expression of the promoterless neogene in the HAC vector after site-specific recombination. The integrity of the constructs was confirmed by PCR and restriction enzyme analysis (data not shown). The resulting construct and Cre-expression vector pBS185 were cotransfected into CHO carrying an HAC vector. After selection using G418, site-specific insertion of the mCD40L genomic fragment into the HAC vector was confirmed using genomic PCR (Fig. 1b, c) and FISH analysis using the Cot1 and mCD40L gene probes (Fig. 1d). The yield of CHO cells carrying mCD40L-HAC was ∼5 × 10−5. The mCD40L gene on the HAC vector was not expressed in CHO cells (data not shown). These results showed that the mCD40L gene was inserted into the loxP-site on the HAC vector by Cre-mediated recombination in the expected manner and that the construct existed as a mitotically stable extraminichromosome.
Expression of the mCD40L gene on the HAC vector in human cells
To investigate how the mCD40L gene on the HAC vector was expressed in different cells, the mCD40L-HAC vector was transferred from CHO into cells of the Jurkat (human CD4-positive active T-cell line) and U937 (human myeloid cell line) lines by MMCT. FACS analysis showed that about 10% of the Jurkat cells expressed human CD40L, whereas the U937 cells did not express human CD40L at all (data not shown). After selection using G418, the G418-resistant clones were confirmed to carry mCD40L-HAC using genomic PCR (Fig. 2a). The transfer efficiency was ∼2.4 × 10−5, and a total of 42 Jurkat clones carrying mCD40L-HAC were obtained. Twenty clones carrying the mCD40L-HAC vector were used for subsequent analyses. FISH analyses using hchr21 alphoid DNA or mouse CD40L genomic DNA as a probe showed that the mCD40L-HAC vector was maintained independently (Fig. 2b, c) and that the retention rate was more than 94% for each clone (n = 50 for each clone). Ten U937 clones carrying the mCD40L-HAC vector were also obtained, and for these, the retention rate of the mCD40L-HAC vector was more than 96% for each clone (n = 50 for each clone).
Introduction of the mCD40L-HAC vector into Jurkat cells. a Polymerase chain reaction (PCR) analysis for the genomic DNA of Jurkat cells carrying the mCD40L-HAC vector. The same primer pairs used for Chinese hamster ovary (CHO) cells were used to detect the mCD40L-HAC vector. The 100-bp DNA ladder was used as a size marker. b Fluorescent in situ hybridization (FISH) analysis of the mCD40L-HAC vector in Jurkat cells. A representative metaphase FISH image of Jurkat cells carrying the mCD40L-HAC vector. Arrowhead indicates the mCD40L-HAC vector. Cells were probed with human chromosome 21 alphoid DNA (red). Since the α-satellite probe cross-hybridizes to chromosome 13, four additional fluorescence signals, corresponding to endogenous chromosomes 21 and 13, were observed. c FISH analysis of the mCD40L-HAC vector in Jurkat cells. Cells were probed with mCD40L genomic DNA (red)
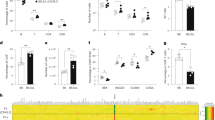
Expression of the mCD40L gene on the HAC vector in Jurkat and U937 cells was analyzed by flow cytometry (Fig. 3a, b). In the Jurkat cells carrying the mCD40L-HAC vector, an average of 8% of the cells (range 5–20%) expressed mCD40L. The percentage of cells expressing mCD40L (8%) was similar to the percentage of cells expressing hCD40L (10%). The percentage expressing both mCD40L and hCD40L was ∼1%, suggesting that CD40L is stochastically expressed in a proportion of Jurkat cells. The results of flow cytometry analysis suggested that expression levels of endogenous hCD40L and exogenous mCD40L in Jurkat cells are in a similar range, as expected. In contrast, the mCD40L gene on the HAC vector was not expressed in U937 cells (Fig. 3a). The expression of mCD40L was confirmed by RT-PCR in Jurkat cells and U937 cells (Fig. 3c). These results suggest that introduction of the mCD40L genomic fragment via an HAC vector permits cell-type-specific expression of mCD40L.
Analysis of mouse CD40L expression in Jurkat and U937 cells. a Histogram data of flow cytometry analysis. The left panel shows the expression of mCD40L in Jurkat cells. The right panel shows the expression of mCD40L in U937 cells. b Dot plot data of flow cytometry analysis. c Reverse transcriptase polymerase chain reaction (RT-PCR) in Jurkat and U937 cells
Functional analysis of the mCD40L gene introduced via the HAC vector
To investigate the function of the mCD40L gene introduced via the HAC vector, B cells obtained from CD40L knockout mice were cultured with Jurkat cells carrying the mCD40L-HAC vector or intact Jurkat cells as a control. After 7 days of coculture, the concentration of mouse IgG in the culture medium was analyzed using ELISA. In the medium in which mouse B cells and Jurkat cells with the mCD40L-HAC vector were cultured, mouse IgG was detected at a concentration of 6.8–9.8 μg/ml (Fig. 4). In contrast, no IgG was detected in the medium in which mouse B cells and intact Jurkat cells were cultured. We prepared normal mouse T cells from C57BL/6. When these cells were stimulated by phorbol myristate acetate (PMA) and ionomycin, about 34% of the cells expressed CD40L. We used the stimulated cells as a positive control for the above assay system. IgG was detected at a concentration of 9.9–12.5 μg/ml. These results show that the mCD40L genomic fragment on the HAC vector functions to stimulate mouse B cells and induce the secretion of IgG in a similar fashion to activated normal mouse T cells.
Functional analysis of the mouse CD40L gene on the human artificial chromosome (HAC) vector. B cells obtained from CD40L knockout mice were cultured with Jurkat cells carrying mCD40L-HAC (five different clones), intact Jurkat cells, or the stimulated mouse T cells for a week, and then the culture medium was harvested. The concentration of mouse immunoglobulin (Ig)G in the culture medium was measured using enzyme-linked immunosorbent assay (ELISA)
Discussion
The results of this study suggest that introduction of a CD40L genomic fragment via an HAC vector permits cell-type-specific expression and functions to stimulate mouse B cells, inducing secretion of IgG. The mCD40L-HAC was functional for mouse B cells obtained from CD40L knockout mice. Thus, mCD40L-HAC has the potential to correct the function of T cells with mutations in the gene encoding the CD40 ligand, the basis for gene therapy for HIGM. We showed that HAC vectors containing the hCD40L genomic fragment have the potential to be useful in gene-therapy-based treatment of HIGM. In this study, we tried unsuccessfully to introduce the mCD40L-HAC vector into primary T cells obtained from CD40L knockout mice. We assume that the reason for this is HAC being introduced into the cells with a very low efficiency. For ex vivo gene delivery to primary cells or somatic stem cells with a limited lifespan, improvements in efficiency are essential.
An advantage of the HAC vector for gene therapy is that it can be maintained as an independent minichromosome without integration into the host genome. In this study, the mCD40L-HAC vector was maintained independently in all 20 Jurkat clones. This suggests that introduction of the mCD40L-HAC vector into cells will not cause insertional mutagenesis and that expression of the CD40L genome would not be subject to position effects. In a previous study, we confirmed the HAC vector is mitotically stable in human primary hematopoietic cells without selection (Yamada et al. 2006). Thus, use of HAC vectors would be safe, causing no lesions in the host genome with introduction of the transgene, and would provide long-term transgene expression.
This study shows that, in combination with BAC/PAC recombineering technologies, the HAC vector enables the gene of interest to be delivered as a genomic fragment. This novel gene expression system has the potential to permit intrinsic regulation of the transgene. Also, a ready-made genomic library with a YAC or BAC backbone or multiple genomic fragments can be loaded onto the HAC vector. Furthermore, a genomic fragment several Mb long can be loaded into the HAC vector using the Cre-loxP-based chromosome engineering technology (Kuroiwa et al. 2002). In combination with these technologies, HAC vectors could be used to deliver genomic fragments more than 1 Mb in size.
We previously demonstrated that the HAC vector can be used for gene transfer into human fibroblast, hematopoietic, and mesenchymal stem cells, providing long-term expression of the therapeutic gene and conferring a growth advantage on cells carrying the vector (Kakeda et al. 2005; Ren et al. 2005; Yamada et al. 2006). In this study, we addressed the use of the HAC vector for treating a genetic disorder caused by a gene deficiency, which requires intrinsic gene expression. This study is the first to support such an approach and represents a first step toward a novel gene therapy utilizing the HAC vector and BAC engineering. Further studies using this gene expression system, which utilizes the HAC vector carrying the therapeutic gene, will make the HAC vector a novel option for ex vivo gene therapy.
References
Blaese RM, Culver KW, Miller AD, Carter CS, Fleisher T, Clerici M, Shearer G, Chang L, Chiang Y, Tolstoshev P, Greenblatt JJ, Rosenberg SA, Klein H, Berger M, Mullen CA, Ramsey WJ, Muul L, Morgan RA, Anderson WF (1995) T lymphocyte-directed gene therapy for ADA- SCID: initial trial results after 4 years. Science 270:475–480
Bossen C, Ingold K, Tardivel A, Bodmer JL, Gaide O, Hertig S, Ambrose C, Tschopp J, Schneider P (2006) Interactions of tumor necrosis factor (TNF) and TNF receptor family members in the mouse and human. J Biol Chem 281:13964–13971
Brown MP, Topham DJ, Sangster MY, Zhao J, Flynn KJ, Surman SL, Woodland DL, Doherty PC, Farr AG, Pattengale PK, Brenner MK (1998) Thymic lymphoproliferative disease after successful correction of CD40 ligand deficiency by gene transfer in mice. Nat Med 4:1253–1260
Cavazzana-Calvo M, Hacein-Bey S, de Saint Basile G, Gross F, Yvon E, Nusbaum P, Selz F, Hue C, Certain S, Casanova JL, Bousso P, Deist FL, Fischer A (2000) Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science 288:669–672
Cron RQ (2002) CD154 transcription regulation in primary human CD4 T cells. Immunol Res 27:185–202
DiSanto JP, Bonnefoy JY, Gauchat JF, Fischer A, de Saint Basile G (1993) CD40 ligand mutations in x-linked immunodeficiency with hyper-IgM. Nature 361:541–543
Doherty AM, Fisher EM (2003) Microcell-mediated chromosome transfer (MMCT): small cells with huge potential. Mamm Genome 14:583–592
Gaspar HB, Howe S, Thrasher AJ (2003) Gene therapy progress and prospects: gene therapy for severe combined immunodeficiency. Gene Ther 10:1999–2004
Grewal IS, Flavell RA (1998) CD40 and CD154 in cell-mediated immunity. Annu Rev Immunol 16:111–135
Grimes BR, Warburton PE, Farr CJ (2002) Chromosome engineering: prospects for gene therapy. Gene Ther 9:713–718
Hacein-Bey-Abina S, Le Deist F, Carlier F, Bouneaud C, Hue C, De Villartay JP, Thrasher AJ, Wulffraat N, Sorensen R, Dupuis-Girod S, Fischer A, Davies EG, Kuis W, Leiva L, Cavazzana-Calvo M (2002) Sustained correction of X-linked severe combined immunodeficiency by ex vivo gene therapy. N Engl J Med 346:1185–1193
Kakeda M, Hiratsuka M, Nagata K, Kuroiwa Y, Kakitani M, Katoh M, Oshimura M, Tomizuka K (2005) Human artificial chromosome (HAC) vector provides long-term therapeutic transgene expression in normal human primary fibroblasts. Gene Ther 12:852–856
Katoh M, Ayabe F, Norikane S, Okada T, Masumoto H, Horike S, Shirayoshi Y, Oshimura M (2004) Construction of a novel human artificial chromosome vector for gene delivery. Biochem Biophys Res Commun 321:280–290
Korthauer U, Graf D, Mages HW, Briere F, Padayachee M, Malcolm S, Ugazio AG, Notarangelo LD, Levinsky RJ, Kroczek RA (1993) Defective expression of T-cell CD40 ligand causes X-linked immunodeficiency with hyper-IgM. Nature 361:539–541
Kroczek RA, Graf D, Brugnoni D, Giliani S, Korthuer U, Ugazio A, Senger G, Mages HW, Villa A, Notarangelo LD (1994) Defective expression of CD40 ligand on T cells causes “X-linked immunodeficiency with hyper-IgM (HIGM1)”. Immunol Rev 138:39–59
Kuroiwa Y, Yoshida H, Ohshima T, Shinohara T, Ohguma A, Kazuki Y, Oshimura M, Ishida I, Tomizuka K (2002) The use of chromosome-based vectors for animal transgenesis. Gene Ther 9:708–712
Larin Z, Mejia JE (2002) Advances in human artificial chromosome technology. Trends Genet 18:313–319
Lee EC, Yu D, Martinez de Velasco J, Tessarollo L, Swing DA, Court DL, Jenkins NA, Copeland NG (2001) A highly efficient Escherichia coli-based chromosome engineering system adapted for recombinogenic targeting and subcloning of BAC DNA. Genomics 73:56–65
Muul LM, Tuschong LM, Soenen SL, Jagadeesh GJ, Ramsey WJ, Long Z, Carter CS, Garabedian EK, Alleyne M, Brown M, Bernstein W, Schurman SH, Fleisher TA, Leitman SF, Dunbar CE, Blaese RM, Candotti F (2003) Persistence and expression of the adenosine deaminase gene for 12 years and immune reaction to gene transfer components: long-term results of the first clinical gene therapy trial. Blood 101:2563–2569
Onodera M, Ariga T, Kawamura N, Kobayashi I, Ohtsu M, Yamada M, Tame A, Furuta H, Okano M, Matsumoto S, Kotani H, McGarrity GJ, Blaese RM, Sakiyama Y (1998) Successful peripheral T-lymphocyte-directed gene transfer for a patient with severe combined immune deficiency caused by adenosine deaminase deficiency. Blood 91:30–36
Ren X, Katoh M, Hoshiya H, Kurimasa A, Inoue T, Ayabe F, Shibata K, Toguchida J, Oshimura M (2005) A novel human artificial chromosome vector provides effective cell lineage-specific transgene expression in human mesenchymal stem cells. Stem Cells 23:1608–1616
Roy M, Waldschmidt T, Aruffo A, Ledbetter JA, Noelle RJ (1993) The regulation of the expression of gp39, the CD40 ligand, on normal and cloned CD4+ T cells. J Immunol 151:2497–2510
Saffery R, Choo KH (2002) Strategies for engineering human chromosomes with therapeutic potential. J Gene Med 4:5–13
Stout RD, Suttles J, Xu J, Grewal IS, Flavell RA (1996) Impaired T cell-mediated macrophage activation in CD40 ligand-deficient mice. J Immunol 156:8–11
Straw AD, MacDonald AS, Denkers EY, Pearce EJ (2003) CD154 plays a central role in regulating dendritic cell activation during infections that induce Th1 or Th2 responses. J Immunol 170:727–734
Yamada H, Kunisato A, Kawahara M, Tahimic CG, Ren X, Ueda H, Nagamune T, Katoh M, Inoue T, Nishikawa M, Oshimura M (2006) Exogenous gene expression and growth regulation of hematopoietic cells via a novel human artificial chromosome. J Hum Genet 51:147–150
Acknowledgment
This work was supported by the 21st Century COE Program: The Research Core for Chromosome Engineering Technology (HY, YL, MN, MO, and TI)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamada, H., Li, Y.C., Nishikawa, M. et al. Introduction of a CD40L genomic fragment via a human artificial chromosome vector permits cell-type-specific gene expression and induces immunoglobulin secretion. J Hum Genet 53, 447–453 (2008). https://doi.org/10.1007/s10038-008-0268-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10038-008-0268-0
Keywords
This article is cited by
-
Combinations of chromosome transfer and genome editing for the development of cell/animal models of human disease and humanized animal models
Journal of Human Genetics (2018)
-
De novo formed satellite DNA-based mammalian artificial chromosomes and their possible applications
Chromosome Research (2015)
-
A pathway from chromosome transfer to engineering resulting in human and mouse artificial chromosomes for a variety of applications to bio-medical challenges
Chromosome Research (2015)
-
A new generation of human artificial chromosomes for functional genomics and gene therapy
Cellular and Molecular Life Sciences (2013)
-
HACking the centromere chromatin code: insights from human artificial chromosomes
Chromosome Research (2012)