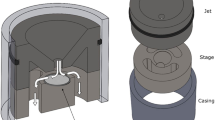
Abstract
The potential for human exposure to engineered nanoparticles due to the use of nanotechnology-based consumer sprays (categorized as such by the Nanotechnology Consumer Products Inventory) is examined along with analogous products, which are not specified as nanotechnology-based (regular products). Photon correlation spectroscopy was used to obtain particle size distributions in the initial liquid products. Transmission electron microscopy was used to determine particle size, shape, and agglomeration of the particles. Realistic application of the spray products near the human breathing zone characterized airborne particles that are released during use of the sprays. Aerosolization of sprays with standard nebulizers was used to determine their potential for inhalation exposure. Electron microscopy detected the presence of nanoparticles in some nanotechnology-based sprays as well as in several regular products, whereas the photon correlation spectroscopy indicated the presence of particles <100 nm in all investigated products. During the use of most nanotechnology-based and regular sprays, particles ranging from 13 nm to 20 μm were released, indicating that they could he inhaled and consequently deposited in all regions of the respiratory system. The results indicate that exposures to nanoparticles as well as micrometer-sized particles can be encountered owing to the use of nanotechnology-based sprays as well as regular spray products.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 6 print issues and online access
$259.00 per year
only $43.17 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Abismaïl B., Canselier J.P., Wilhelm A.M., Delmas H., and Gourdon C. Emulsification by ultrasound: drop size distribution and stability. Ultrason Sonochem 1999: 6 (1 and 2): 75–83.
Allen T. Powder Sampling and Particle Size Determination, 1 illustrated edn. Elsevier, Amsterdam, the Netherlands, 2003.
Bermudez E., Mangum J.B., Wong B.A., Asgharian B., Hext P.M., and Warheit D.B., et al. Pulmonary responses of mice, rats, and hamsters to subchronic inhalation of ultrafine titanium dioxide particles. Toxicol Sci 2004: 77: 347–357.
Bodycomb J. Questions and Answers for Driving the Brookhaven 90Plus. Brookhaven Instruments Corporation, Holtsville, NY, 2009.
Bradford A., Handy R.D., Readman J.W., Atfield A., and Mühling M. Impact of silver nanoparticle contamination on the genetic diversity of natural bacterial assemblages in estuarine sediments. Environ Sci Technol 2009: 43 (12): 4530–4536.
Brookhaven Instruments Corporation. Instruction Manual for 90Plus/BI-MAS Multi Angle Particle Sizing Option Operation Manual. Brookhaven Instruments Corporation, Holtsville, NY, 1995.
Bruce J., and Berne R.P. Dynamic Light Scattering: With Applications to Chemistry, Biology, and Physics, 2 unabridged, illustrated edn Courier Dover Publications, Mineola, NY, 2000.
Carlo S.D., El-Bez C., Alvarez-Rua C., Borge J., and Dubochet J. Cryo-negative staining reduces electron-beam sensitivity of vitrified biological particles. J Struct Biol 2002: 138: 216–226.
Drobne D. Nanotoxicology for safe and sustainable nanotechnology. Arhiv Higijenu Rada Toksikologiju 2007: 58 (4): 471–478.
Egerton R.F., Li P., and Malac M. Radiation damage in the TEM and SEM. Micron 2004: 35 (6): 399–409.
Elder A., Gelein R., Silva V., Feikert T., Opanashuk L., and Carter J., et al. Translocation of inhaled ultrafine manganese oxide particles to the central nervous system. Environ Health Perspect 2006: 114 (8): 1172–1178.
Frater L., Stokes E., Lee R., and Oriola T. An Overview of the Framework of Current Regulation affecting the Development and Marketing of Nanomaterials. Cardiff University, Cardiff, 2006 p 192.
Geiser M., Rothen-Rutishauser B., Kapp N., Schürch S., Kreyling W., and Schulz H., et al. Ultrafine particles cross cellular membranes by nonphagocytic mechanisms in lungs and in cultured cells. Environ Health Perspect 2005: 113 (11): 1555–1560.
Grassian V.H., Adamcakova-Dodd A., Pettibone J.M., O’Shaughnessy P.T., and Thorne P.S. Inflammatory response of mice to manufactured titanium dioxide nanoparticles: comparison of size effects through different exposure routes. Nanotoxicology 2007a: 1 (3): 211–226.
Grassian V.H., O’Shaughnessy P.T., Adamcakova-Dodd A., Pettibone J.M., and Thorne P.S. Inhalation exposure study of titanium dioxide nanoparticles with a primary particle size of 2 to 5 nm. Environ Health Perspect 2007b: 115 (3): 397–402.
Hagendorfer H., Lorenz C., Kaegi R., Sinnet B., Gehrig R., and Goetz N.V., et al. Size-fractionated characterization and quantification of nanoparticle release rates from a consumer spray product containing engineered nanoparticles. J Nanoparticle Res 2009: 12 (7): 2481–2494.
Han H-S., Whitbey E.R., Plate D.B., and Albertson D.P. Combining TSI Scanning Mobility Particle Sizer and Aerodynamic Particle Sizer for Wide Range Particle Size Distribution Measurement. European Aerosol Conference, Ghent, Belgium, 2005.
Han T., O’Neil D.L., and Ortiz C.A. A generic-tee-plenum mixing system for application to single point aerosol sampling in stacks and ducts. Health Phys 2007: 92 (1): 40–49.
Hand J.L., and Kreidenweis S.M. A new method for retrieving particle refractive index and effective density from aerosol size distribution data. Aerosol Sci Technol 2002: 36: 1012–1026.
Hansen S.F., Michelson E.S., Kamper A., Borling P., Stuer-Lauridsen F., and Baun A. Categorization framework to aid exposure assessment of nanomaterials in consumer products. Ecotoxicology 2008: 17: 438–447.
Hinds W.C. Aerosol Technology: Properties, Behavior, and Measurement of Airborne Particles, 2 illustrated edn. Wiley, the University of Michigan, New York, 1999.
Hobbs L.W. Electron-beam sensitivity in inorganic specimens. Ultramicroscopy 1987: 23 (3 and 4): 339–344.
Keenan C.R., Goth-Goldstein R., Lucas D., and Sedlak D.L. Oxidative Stress Induced by Zero-Valent Iron Nanoparticles and Fe(II) in Human Bronchial Epithelial Cells. Environ Sci Technol 2009: 43 (12): 4555–4560.
Khlystov A., Stanier C., and Pandis S.N. An Algorithm for Combining Electrical Mobility and Aerodynamic Size Distributions Data when Measuring Ambient Aerosol. Aerosol Sci Technol 2004: 38 (S1): 229–238.
Kim Y.D., Morr C.V., and Schenz T.W. Microencapsulation Properties of Gum Arabic and Several Food Proteins: Liquid Orange Oil Emulsion Particles. J Agric Food Chem 1996: 44 (5): 1308–1313.
Leapman R.D., and Sun S. Cryo-electron energy loss spectroscopy: observations on vitrified hydrated specimens and radiation damage. Ultramicroscopy 1995: 59: 71–79.
Lioy P.J., Han T.W., Nazarenko Y., Lioy M.J., and Mainelis G. Nanotechnology and Exposure Science—What Is Needed To Fill the Research and Data Gaps for Consumer Products. Nanotechnol Expos Assessment (Special issue) 2010: 16 (4): 376–385.
Lioy P.J., Weisel C.P., Millette J., Vallero S.E.D., Offenberg J., and Buckley B., et al. Characterization of the dust/smoke aerosol that settled east of the World Trade Center (WTC) in lower Manhattan after the collapse of the WTC September 11, 2001. Environ Health Perspect 2002: 110: 703–714.
Lioy P.J. Exposure Science: A View of the Past and Major Milestones for the Future. Environ Health Perspect 2010b: 118: 1081–1090.
Matyi R.J., Schwartz L.H., and Butt J.B. Particle Size, Particle Size Distribution, and Related Measurements of Supported Metal Catalysts. Catalysis Reviews 1987: 29 (1): 41–99.
Maynard A.D., Aitken R.J., Butz T., Colvin C., Donaldson K., and Oberdörster G., et al. Safe Handling of Nanotechnology. Nature 2006: 444: 267–269.
Maynard A.D. Nanotechnology: The Next Big Thing, or Much Ado about Nothing. Ann Occup Hyg 2007: 51 (1): 1–12.
Maynard A.D., and Aitken R.J. Assessing exposure to airborne nanomaterials: current abilities and future requirements. Nanotoxicology 2007: 1 (1): 26–41.
Michelson E.S. Globalization at the nano frontier: the future of nanotechnology policy in the United States, China, and India. Technol Soc 2008: 30: 405–410.
National Science and Technology Council. The National Nanotechnology Initiative Strategic Plan (NNISP). Executive Office of the President, National Science and Technology Council, Washington, DC, 2011.
Oberdörster G., Maynard A., Donaldson K., Castranova V., Fitzpatrick J., and Ausman K., et al. Principles for characterizing the potential human health effects from exposure to nanomaterials: elements of a screening strategy. Particle Fibre Toxicol 2005a: 2 (8): 1–35.
Oberdörster G., Oberdörster E., and Oberdörster J. Nanotoxicology: An Emerging Discipline Evolving from Studies of Ultrafine Particles. Environ Health Perspect 2005b: 113 (7): 823–839.
Oberdörster G., Sharp Z., Atudorei V., Elder A., Gelein R., and Kreyling W., et al. Translocation of Inhaled Ultrafine Particles to the Brain. Inhal Toxicol 2004: 16 (6 and 7): 437–445.
Pant V., Deshpande C.G., and Kamra A.K. The concentration and number size distribution measurements of the Marine Boundary Layer aerosols over the Indian Ocean. Atmos Res 2009: 92 (4): 381–393.
Paull J., and Lyons K. Nanotechnology: the next challenge for organics. J Organic Syst 2008: 3 (1): 3–22.
Qu G., Bai Y., Zhang Y., Jia Q., Zhang W., and Yan B. The effect of multiwalled carbon nanotube agglomeration on their accumulation in and damage to organs in mice. Carbon 2009: 47 (8): 2060–2069.
Schmid A., Goel S., Wang W., Beiu V., Carrara S., and Riediker M. Chances and risks of nanomaterials for health and environment. In: Akan O., Bellavista P., Cao J., Dressler F., Ferrari D., Gerla M., et al. (Eds.). Nano-Net. Springer, Berlin, Heidelberg, 2009 Vol. 20, pp. 128–133.
Segal S.H. Environmental Regulation of Nanotechnology: Avoiding Big Mistakes for Small Machines. Nanotechnol Law Business 2004: 1 (3): 290–304.
Shimada M., Wang W-N., Okuyama K., Myojo T., Oyabu T., and Morimoto Y., et al. Development and Evaluation of an Aerosol Generation and Supplying System for Inhalation Experiments of Manufactured Nanoparticles. Environ Sci Technol 2009: 43 (14): 5529–5534.
Shrader-Frechette K. Nanotoxicology and Ethical Conditions for Informed Consent. Nanoethics 2007: 1: 47–56.
Som C., Berges M., Chaudhry Q., Dusinska M., Fernandes T., and Olsen S., et al. The importance of life cycle concepts for the development of safe nanoproducts. Toxicology 2010: 269: 160–169.
Thomas T., and Bahadori T., et al. Moving toward exposure and risk evaluation of nanomaterials: challenges and future directions. Wiley Interdiscip Rev Nanomed Nanobiotechnol 2009: 1 (4): 426–433.
Turgis J-D., and Coqueret X. Electron beam sensitivity of butyl acrylate copolymers: effects of composition on reactivity. Macromol Chem Phys 1999: 200: 652–660.
Van Calster G. Regulating Nanotechnology in the European Union. Nanotechnol Law Business 2006: 3 (3): 359–374.
Wang J., Zhou G., Chen C., Yu H., Wang T., and Ma Y., et al. Acute toxicity and biodistribution of different sized titanium dioxide particles in mice after oral administration. Toxicol Lett 2007: 168 (2): 176–185.
Warheit D.B., Borm P.J.A., Hennes C., and Lademann J. Testing Strategies to Establish the Safety of Nanomaterials: Conclusions of an ECETOC Workshop. Inhal Toxicol 2007a: 19: 631–643.
Warheit D.B., Webb T.R., Reed K.L., Frerichs S., and Sayes C.M. Pulmonary toxicity study in rats with three forms of ultrafine-TiO2 particles: differential responses related to surface properties. Toxicol Lett 2007b: 230 (1): 90–104.
Wick P., Manser P., Limbach L.K., Dettlaff-Weglikowska U., Krumeich F., and Roth S., et al. The degree and kind of agglomeration affect carbon nanotube cytotoxicity. Toxicol Lett 2007: 168 (2): 121–131.
Wirnitzer U., Herbold B., Voetz M., and Ragot J. Studies on the in vitro genotoxicity of baytubes®, agglomerates of engineered multi-walled carbon-nanotubes (MWCNT). Toxicol Lett 2009: 186 (3): 160–165.
Woodrow Wilson International Center for Scholars. The Project on Emerging Nanotechnologies. http://www.nanotechproject.org/(accessed 10, June 2010a).
Woodrow Wilson International Center for Scholars. Nanotechnology Consumer Products Inventory. http://www.nanotechproject.org/inventories/consumer/(accessed 10, June 2010b).
Acknowledgements
This research was supported in part by the NIEHS sponsored UMDNJ Center for Environmental Exposures and Disease, Grant No.: NIEHS P30ES005022. We thank Dr. Satya Seshadri for help in building the experimental system.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Nazarenko, Y., Han, T., Lioy, P. et al. Potential for exposure to engineered nanoparticles from nanotechnology-based consumer spray products. J Expo Sci Environ Epidemiol 21, 515–528 (2011). https://doi.org/10.1038/jes.2011.10
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/jes.2011.10
Keywords
This article is cited by
-
Potential consumer exposure to respirable particles and TiO2 due to the use of eyebrow powders
Journal of Exposure Science & Environmental Epidemiology (2021)
-
Estimates of lung burden risk associated with long-term exposure to TiO2 nanoparticles as a UV-filter in sprays
Environmental Science and Pollution Research (2021)
-
Ultrafine particles in scanning sprays: a standardized examination of five powders used for dental reconstruction
Journal of Occupational Medicine and Toxicology (2020)
-
Modeling consumer exposure to spray products: an evaluation of the ConsExpo Web and ConsExpo nano models with experimental data
Journal of Exposure Science & Environmental Epidemiology (2020)
-
Assessment of information availability for environmental impact assessment of engineered nanomaterials
Journal of Nanoparticle Research (2018)