Abstract
We have correlated the binding affinities of ME1036, a carbapenem, to the penicillin-binding proteins (PBPs) from Streptococcus pneumoniae strains, with its bactericidal potency against those same strains. Certain mutations in the PBPs from S. pneumonaie strains decrease the binding affinities of β-lactams for PBPs, which gives rise to clinical resistance to those β-lactams. ME1036 has been shown to be strongly active against genotypic penicillin-intermediate S. pneumoniae (gPISP) strains and genotypic penicillin-resistant S. pneumoniae (gPRSP) strains that contain more than one mutation in their PBPs, owing to its strong affinity for those PBPs.
Similar content being viewed by others
Introduction
The emergence of penicillin-intermediate and penicillin-resistant Streptococcus pneumoniae strains (PISP and PRSP strains, respectively) in Europe was first reported in the 1970s. The strains then spread throughout South America and Asia in the 1980s. In general, between 30 and 50% of clinically isolated S. pneumoniae strains are PISP or PRSP strains. In Japan, the prevalence of PISP and PRSP strains is ∼60%.1 PRSP strains are also resistant to tetracycline and macrolides. Furthermore, fluoroquinolone-resistant strains with mutations in DNA gyrase are being found.2 The increase in these S. pneumoniae strains that resist the activities of penicillins, oral cephems, tetracyclines, macrolides and quinolones is a major concern because of decrease of antibiotics effective against these strains.
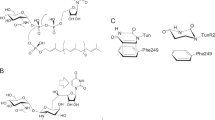
ME1036 (Figure 1) is a new broad-spectrum parenteral carbapenem that is active against multi-drug resistant Gram-positive and Gram-negative bacteria, including methicillin-resistant Staphylococcus aureus (MRSA) and extended-spectrum β-lactamase-producing Enterobacteriaceae.3 The study reported herein was designed to evaluate the relationship between the binding affinities of ME1036 for wild-type and mutant penicillin-binding proteins (PBPs) and its efficacy against S. pneumoniae strains containing those PBPs.
Results
ME1036 antibacterial activity against S. pneumoniae
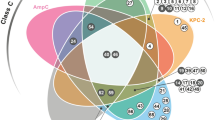
The number of strains that carried a given mutation(s) was correlated with their associated MIC values (Figure 2). The antibacterial activities against the S. pneumoniae strains for ME1036, panipenem and ceftriaxone were affected by mutations in pbp1a, pbp2x and pbp2b. The MIC100 value found for ME1036 was 0.03 μg ml−1, which is substantially smaller than the largest values found for the other antibiotics (Table 1). The activity of ceftriaxone was also influenced by mutations in pbp2x, and its MIC100 was 4 μg ml−1.
Affinity for PBPs
The fluorograms used to determine the IC50 values of ME1036 and the other antibiotics for the PBPs from S. pneumoniae R6 and 197 are shown in Figure 3 and 4, and the corresponding IC50 values are shown in Tables 2 and 3, respectively. The binding affinities of ME1036, imipenem and panipenem for PBP2A/2X and PBP2B from penicillin-susceptible S. pneumoniae (PSSP) strains are stronger than those of ceftriaxone. The strong affinities of the carbapenems for PBP2A/2X from the PRSP strain correlate with their low MICs. The IC50 values of the carbapenems against PBP1A, PBP1B and PBP2B from the PRSP strain were smaller than were those of ceftriaxone.
Time-kill study
The bactericidal activities of ME1036 and the other carbapenems against S. pneumoniae 197 over a 6-h period are shown in Figure 5. ME1036 killed >99.9% of the bacteria within 4 h when the dosage was its MIC value or greater. Conversely, imipenem and panipenem needed four times their MIC value to have the same effect within 4 h, and ceftriaxone was not bactericidal even at four times its MIC value at 6 h.
Discussion
We previously found ME1036 to be a broad spectrum antibiotic that is active against MRSA PRSP strains and extended-spectrum β-lactamase-producing Enterobacteriaceae.3 For the study reported herein, ME1036 was found to have potent anitbacterial activity against genotypic PRSPs (gPRSPs) that are resistant to β-lactams due to mutated pbps. In particular, the strong binding affinities of antibiotics for PBP1A, PBP2X and PBP2B from gPRSP is thought to be basis for its strong antibacterial activities.4 Compared with other antimicrobial drugs in terms of binding affinity for each PBPs of genotypic PSSP (gPSSP) and gPRSP, the affinity for the PBPs of ME1036 was correlated with its MIC. Therefore, the potent antibacterial activity of ME1036 is likely the result of its strong affinity for the PBPs.
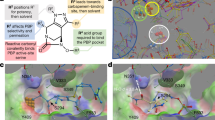
The interactions between the C-2 side chains of carbapenems and the conserved Trp and Thr residues in the PBPs have important roles in binding of carbapenems to the PBPs.5 Crystallographic structures revealed that the carbapenem C-2 side chains form non-polar interactions with the side chains of Trp374 and Thr526 of PBP2X and with those of Trp411 and Thr543 of PBP1A. Because ME1036 possesses an imidazo[5,1-b]thiazole moiety at the C-2 position, it may also interact with the respective tryptophan and threonine residues of PBP1A and PBP2X in a similar manner, which would result in strong binding affinities. Considering that the corresponding positions in PBP2B also contain a Trp and Thr,6 the strong binding affinities of ME1036 for all three PBPs may be a consequence of identical or similar non-polar interactions involving the C-2 side chains of ME1036. The binding affinities of antibiotics for PBP1A, PBP2X and PBP2B is thought to be important for antibacterial activity against S. pneumoniae.7 Although the relationship between the binding affinities of antibiotics for PBPs and their bactericidal activities has yet to be definitively proven, it is possible that the potent bactericidal activity of ME1036 against the S. pneumoniae strains, including the gPRSPs, is caused by its strong affinities for PBPs.8
Drug-resistant S. pneumoniae strains are usually isolated from the sputum, pharynx, nasal cavity or otorrhea of patients, and from the otitis media and tissues affected by adenoiditis in children. The major serotypes of gPRSPs are 6, 9, 14, 19 and 23, and these may cause meningitis in young and children, although the evidence is not definitive. Although, a vaccine against the major PRSP serotypes is widely available, PRSP infection is still of concern and requires medical intervention.9, 10, 11
In conclusion, we have shown that ME1036 is potent against MRSA and also S. pneumoniae, including PRSP, and that it has a strong affinity for PBPs. The medical need for a parenteral carbapenem with broad-spectrum and potent bactericidal activity is still great, such that further development of antimicrobial agents is expected to be necessary.
Methods
Bacterial strains
A total of 129 strains of S. pneumoniae clinically isolated in Japan in 2001 were identified as gPSSP strains (with no mutations in any pbps), genotypic PISP (gPISP) (with mutations in pbp2x or pbp2b, in pbp1a and pbp2x, in pbp2x and pbp2b) and gPRSP (with mutations in pbp1a, pbp2x and pbp2b),12, 13 and provided by Dr Kimiko Ubukata (Laboratory of Molecular Epidemiology for Infectious Agents, Kitasato Institute for Life Sciences, Kitasato University). S. pnenumoniae R6 (BAA-255) was purchased from the American Type Culture Collection and S. pneumoniae 197 had been clinically isolated. These strains were stored at Meiji Seika Pharma.
Antibacterial agents
ME1036 was synthesized at the Pharmaceutical Research Center of Meiji Seika Kaisha. Other antibiotics were purchased commercially: meropenem (Dainippon Sumitomo Pharma, Osaka, Japan), panipenem (Daiichi Sankyo, Tokyo, Japan), cefotaxime, (Sanofi-aventis KK, Tokyo, Japan), ceftriaxone (Chugai Pharmaceutical, Tokyo, Japan), penicillin G and amoxicillin (Sigma-Aldrich Chemical, St Louis, MO, USA), vancomycin (Shionogi, Osaka, Japan), levofloxacin (Daiichi Sankyo) and imipenem (MSD KK, Tokyo, Japan).
Susceptibility testing
The MICs for the S. pneumoniae were determined by the two-fold agar dilution method that used Mueller-Hinton agar (Difco, Becton Dickinson, Sparks, MD, USA), supplemented with 5% defibrinated sheep blood.14 Each inoculum was prepared by making a direct Mueller-Hinton broth (Difco) suspension of isolated colonies selected from an agar plate. Each inoculum (104 CFU) was spotted onto a separate agar plate with a multiple inoculator (Microplanter, Sakuma Seisakusho, Tokyo, Japan). MIC values were defined as the lowest concentrations of the antibiotics that prevented visible bacterial growth after incubation at 35 °C for 20 h.
PCR for the identification of PBP genes
Fragments of PBP genes and lytA, which encodes the autolysin15 specific to S. pneumoniae were PCR amplified as reported.13 After PCR amplification, if the DNA for these gene fragments was not detected or was found to be a size different from that expected, the strains from which the gene fragments were acquired were considered to possess mutant PBPs, and the S. pneumoniae strains were classified as gPSSP, gPISP or gPRSP.
Antibiotic affinities for PBPs
The affinities of ME1036 and other β-lactams for PBPs were assessed using a [3H]-benzylpenicillin-competition assay as reported.16 Membrane proteins were isolated from S. pneumoniae strains R6 (PSSP) and 197 (PRSP). Antibiotics were individually incubated with the membrane proteins at 30 °C for 10 min, and then [3H]-benzylpenicillin was added. After a 10-min incubation, excess unlabeled benzylpenicillin was added. Sarkosyl-solubilized membrane fractions were subjected to SDS-polyacrylamide gel electrophoresis and fluorography. Because PBP2X and PBP2A were not completely separable, their binding to the antibiotics were assessed together as PBP2A/2X. The binding affinities of antibiotics to PBPs were quantified by scanning densitometry from the fluorographs. The fluorograph images were analyzed with NIH image software, and IC50 was estimated from the logistic curve of the dose–response relationship.
Time-kill study
Overnight cultures of S. pneumoniae grown on Mueller-Hinton agar supplemented with 5% horse blood were individually inoculated into 5 ml of cation-adjusted Mueller-Hinton broth supplemented with 5% lysed horse blood. After a 2-h incubation at 35 °C, the antibiotics were each added at concentrations equivalent to 1/4, 1/2, 1, 2 or 4 times its MIC to a medium sample. Surviving bacteria were counted after 0, 2, 4 and 6 h of incubation at 35 °C by subculturing 50 μl of serial 10-fold dilutions on Mueller-Hinton agar supplemented with 5% horse blood. The number of colonies for each sample was counted after 24 h of incubation at 35 °C.
References
Goto, H., Shimada, K., Ikemoto, H., Oguri, T. & Study Group on Antimicrobial Susceptibility of Pathogens Isolated from Respiratory Infections. Antimicrobial susceptibility of pathogens isolated from more than 10 000 patients with infectious respiratory diseases: a 25-year longitudinal study. J. Infect. Chemother. 15, 347–360 (2009).
Doern, G. V. et al. Antimicrobial resistance among Streptococcus pneumoniae in the United States: have we begun to turn the corner on resistance to certain antimicrobial classes? Clin. Infect. Dis. 41, 139–148 (2005).
Kurazono, M. et al. In vitro activities of ME1036 (CP5609), a novel parenteral carbapenem, against methicillin-resistant staphylococci. Antimicrob. Agents Chemother. 48, 2831–2837 (2004).
Kosowska-Shick, K., McGhee, P. L. & Appelbaum, P. C. Affinity of ceftaroline and other beta-lactams for penicillin-binding proteins from Staphylococcus aureus and Streptococcus pneumoniae. Antimicrob. Agents Chemother. 54, 1670–1677 (2010).
Yamada, M. et al. Crystal structures of biapenem and tebipenem complexed with penicillin-binding proteins 2X and 1A from Streptococcus pneumoniae. Antimicrob. Agents Chemother. 52, 2053–2060 (2008).
Contreras-Martel, C., Dahout-Gonzalez, C., Martins Ados, S., Kotnik, M. & Dessen, A. PBP active site flexibility as the key mechanism for beta-lactam resistance in pneumococci. J. Mol. Biol. 387, 899–909 (2009).
Cafini, F. et al. Alterations of the penicillin-binding proteins and murM alleles of clinical Streptococcus pneumoniae isolates with high-level resistance to amoxicillin in Spain. J. Antimicrob. Chemother. 57, 224–229 (2006).
Dalhoff, A., Nasu, T. & Okamoto, K. Target affinities of faropenem to and its impact on the morphology of gram-positive and gram-negative bacteria. Chemotherapy 49, 172–183 (2003).
Ho, P. L., Chiu, S. S., Ang, I. & Lau, Y. L. Serotypes and antimicrobial susceptibilities of invasive Streptococcus pneumoniae before and after introduction of 7-valent pneumococcal conjugate vaccine, Hong Kong, 1995–2009. Vaccine 29, 3270–3275 (2011).
Rozenbaum, M. H., Boersma, C., Postma, M. J. & Hak, E. Observed differences in invasive pneumococcal disease epidemiology after routine infant vaccination. Expert Rev. Vaccines 10, 187–199 (2011).
Yao, K. H. et al. Pneumococcal serotype distribution and antimicrobial resistance in Chinese children hospitalized for pneumonia. Vaccine 29, 2296–2301 (2011).
Kobayashi, R. et al. In vitro activity of tebipenem, a new oral carbapenem antibiotic, against penicillin-nonsusceptible Streptococcus pneumoniae. Antimicrob. Agents Chemother. 49, 889–894 (2005).
Ubukata, K. et al. Antibiotic susceptibility in relation to penicillin-binding protein genes and serotype distribution of Streptococcus pneumoniae strains responsible for meningitis in Japan, 1999 to 2002. Antimicrob. Agents Chemother. 48, 1488–1494 (2004).
Clinical and Laboratory Standards Institute.. Methods for dilution antimicrobial susceptibility tests for bacteria that grow aerobically: approved standard M7-A7 (Clinical and Laboratory Standards Institute: Wayne, PA, 2006).
Garcia, P., Garcia, J. L., Garcia, E. & Lopez, R. Nucleotide sequence and expression of the pneumococcal autolysin gene from its own promotor in Escherichia coli. Gene 43, 265–272 (1986).
Utsui, Y. & Yokota, T. Role of an altered penicillin-binding protein in methicillin- and cephem-resistant Staphylococcus aureus. Antimicrob. Agents Chemother. 28, 397–403 (1985).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hirai, Y., Takahata, S., Yamada, K. et al. Correlation of the antimicrobial activity of ME1036 with its binding affinities to the penicillin-binding proteins from Streptococcus pneumoniae strains. J Antibiot 64, 741–746 (2011). https://doi.org/10.1038/ja.2011.76
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2011.76