Abstract
Multi-drug efflux pumps contribute to the resistance of Escherichia coli to many antibiotics and biocides. In this study, we report that the AraC–XylS family regulator YdeO increases the multi-drug resistance of E. coli through activation of the MdtEF efflux pump. Screening of random fragments of genomic DNA for their ability to increase β-lactam resistance led to the isolation of a plasmid containing ydeO, which codes for the regulator of acid resistance. When overexpressed, ydeO significantly increased the resistance of the E. coli strain to oxacillin, cloxacillin, nafcillin, erythromycin, rhodamine 6G and sodium dodecyl sulfate. The increase in drug resistance caused by ydeO overexpression was completely suppressed by deleting the multifunctional outer membrane channel gene tolC. TolC interacts with different drug efflux pumps. Quantitative real-time PCR showed that YdeO activated only mdtEF expression and none of the other drug efflux pumps in E. coli. Deletion of mdtEF completely suppressed the YdeO-mediated multi-drug resistance. YdeO enhances the MdtEF-dependent drug efflux activity in E. coli. Our results indicate that the YdeO regulator, in addition to its role in acid resistance, increases the multi-drug resistance of E. coli by activating the MdtEF multi-drug efflux pump.
Similar content being viewed by others
Introduction
Multi-drug efflux pumps cause serious problems in cancer chemotherapy and in the treatment of bacterial infections. Bacterial drug resistance is often associated with multi-drug efflux pumps that decrease drug accumulation in the cell.1, 2 Bacterial multi-drug efflux pumps are classified into five families on the basis of sequence similarity: major facilitator, resistance-nodulation-cell division (RND), small multi-drug resistance, multidrug and toxic compound extrusion, and ATP-binding cassette.3, 4, 5 Of these, RND family efflux pumps play major roles in both intrinsic and elevated resistance of Gram-negative bacteria to a wide range of compounds, including β-lactams.1, 6, 7, 8, 9, 10, 11, 12 RND efflux pumps require two other proteins to function: a membrane fusion protein and an outer membrane protein. Many drug efflux pumps in Escherichia coli need TolC to function.12, 13, 14, 15 TolC is responsible for resistance to various antibiotics, including β-lactams,12 quinolones16 and macrolides.17 Bacterial genome sequencing enables us to trace drug-resistance genes.18, 19, 20 There are many putative and proven drug efflux pumps in the E. coli genome, and we have identified earlier 20 functional drug efflux pumps.9, 20 As many such efflux pumps have overlapping substrate spectra,9 it is intriguing that bacteria, with their economically organized genomes, harbor such large sets of multi-drug efflux genes.
The key to understanding how bacteria utilize these multiple efflux pumps lies in the regulation of pump expression. The currently available data show that multi-drug efflux pumps are often expressed under precise and elaborate transcriptional control.21, 22, 23, 24 Expression of acrAB, which encodes the major AcrAB efflux pump, is subject to multiple levels of regulation. In E. coli, it is modulated locally by the repressor AcrR25 and AcrS.26 At a more global level, it is modulated by stress conditions and by global regulators, such as MarA, SoxS and Rob.27, 28 These examples illustrate the complexity and diversity of the mechanisms regulating bacterial multi-drug efflux pumps.
Stomach acid (pH⩽2) kills most bacteria;29 however, E. coli survives this acidity.30, 31 The acid resistance appears to contribute to the low infectious dose of pathogenic E. coli, and aids in the gastric passage of commensal strains.32, 33 One of the most efficient acid resistance systems in E. coli, the Gad system, is based on the coordinated action of two isoforms of glutamate decarboxylase (GadA and GadB) and of a specific glutamate/γ-aminobutyrate (GABA) antiporter (GadC).34, 35 The gadA/BC genes, activated in response to acid stress, are subject to complex circuits of regulation involving the AraC–XylS family regulator YdeO.36 In this study, we demonstrate that YdeO contributes to the multi-drug resistance as well as the acid resistance of E. coli. The results suggest a role of YdeO in the multi-drug resistance of E. coli.
Materials and methods
Bacterial strains, plasmids and growth conditions
The bacterial strains and plasmids used in this study are listed in Table 1. The E. coli strains were derived from the wild-type strain MG1655.37 Phage P1-mediated transductions were performed as described earlier.38 Bacterial strains were grown at 37 °C in Luria–Bertani (LB) broth.39 Cells were collected for total RNA extraction when the cultures reached an optical density of 0.6 at 600 nm.
Screening for positive regulators of multi-drug resistance
DNA manipulation generally followed standard practice.39 A genomic library was developed by partial Sau3AI digestion of the chromosomal DNA as follows. Chromosomal DNA prepared from an overnight culture of the wild-type strain MG165537 was digested with Sau3AI (1 U μl−1) for 15, 20, 30 and 40 min. The digested DNA was separated on a 0.8% agarose gel, and fragments approximately 0.5–3 kb in size were purified and ligated into the BamHI site of vector pHSG398 (Takara Bio Inc., Otsu, Japan). The ligation products were transformed into E. coli DH5α39 to select chloramphenicol-resistant transformants. Plasmid DNA was prepared from a pool of 16,000 transformants and used to transform the acrB deletion strain NKE96. Cells were plated on LB agar medium39 containing 15 μg ml−1 chloramphenicol and inhibitory concentrations of various drugs.
Plasmid construction
The ydeO gene was amplified from MG1655 genomic DNA using the primers ydeO-F_BamHI and ydeO-R_SalI listed in Table 2, which introduced BamHI and SalI sites at the ends of the amplified fragment. This PCR product was cloned between the BamHI and SalI sites of the vector pHSG398 (Takara Bio Inc.) to produce the plasmid pydeO.
Construction of gene deletion mutants
Gene deletion was performed according to the method of Datsenko and Wanner, with recombination between short homologous DNA regions catalyzed by phage λ Red recombinase.40 A curable expression plasmid encoding Red recombinase (pKD46) was introduced into the MG1655 strain. The chloramphenicol resistance gene cat or the kanamycin resistance gene kan, flanked by Flp recognition target sites, was amplified by PCR using the primers listed in Table 2. The plasmid pKD3 or pKD4 was used as a template. This PCR product was used to transform the MG1655 strain expressing Red recombinase, and recombinant clones were isolated as chloramphenicol- or kanamycin-resistant colonies. The pKD46 vector was eliminated by incubating at a non-permissive temperature (37 °C), as confirmed by the loss of ampicillin resistance. The chromosomal structure of the mutated loci was verified by PCR, as described earlier,40 and by Southern hybridization using probes specific for (i) the antibiotic resistance genes used during the construction of chromosomal deletions and (ii) sequences flanking the inactivated loci. The deletions were then transferred to the wild-type MG1655 strain by P1 transduction. The cat and kan genes were eliminated using the plasmid pCP20, as described earlier.40
Determination of the MICs for toxic compounds
The antibacterial activities of different agents were determined on Luria agar (1% tryptone, 0.5% yeast extract and 0.5% NaCl) plates containing various concentrations of the compounds (Sigma-Aldrich, St Louis, MO, USA) listed in Table 3. The agar plates were prepared by the two-fold agar dilution technique, as described earlier.41 The MIC was defined as the lowest concentration of a compound that inhibited cell growth. To determine the MICs, bacteria were grown in LB broth at 37 °C overnight and diluted in the same medium. Then, the organisms were tested at a final inoculum size of 104 cfu μl−1 using a multipoint inoculator (Sakuma Seisakusyo, Tokyo, Japan), and were incubated at 37 °C for 20 h.
RNA extraction
Total RNA was isolated from bacterial cultures using the RNeasy Protect Bacteria Mini Kit (Qiagen, Hilden, Germany) and RNase-Free DNase (Qiagen), as described earlier.42 The total RNA was isolated from exponential-phase cultures of ΔacrB/vector (NKE154) and ΔacrB/pydeO (NKE169). The absence of genomic DNA from the DNase-treated RNA samples was confirmed by both non-denaturing agarose electrophoresis gels and PCR with primers against target genomic DNA. The RNA concentration was then determined spectrophotometrically.39
Determination of specific transcript levels by quantitative real-time PCR following reverse transcription (RT)
Bulk cDNA samples were synthesized from total RNA using TaqMan Reverse Transcription Reagents (Applied Biosystems, Carlsbad, CA, USA) and random hexamers, as described earlier.43, 44 The specific primer pairs listed in Table 2 were designed using ABI PRISM Primer Express software (Applied Biosystems). rrsA of 16S rRNA was chosen as the normalizing gene.45 Real-time PCR was performed with each specific primer pair using SYBR Green PCR Master Mix (Applied Biosystems). The reactions were run on an ABI PRISM 7000 Sequence Detection System (Applied Biosystems); the fluorescence signal due to SYBR Green intercalation was monitored to quantify the double-stranded DNA product formed in each PCR cycle. The expression levels of drug efflux pump genes and tolC in ΔacrB/pydeO (NKE169) were compared with those in ΔacrB/vector (NKE154).
Acid resistance assay
A single colony of an E. coli strain harboring plasmid was inoculated into 1 ml of LB broth containing chloramphenicol and grown overnight at 37 °C. The LB broth (20 ml) was inoculated with 0.1 ml of the overnight culture and grown at 37 °C. When the cultures reached a cell density of 2 × 108 cfu ml−1, 50 μl of the culture was transferred to 2 ml of phosphate-buffered saline (PBS; pH 7.2) and to 2 ml of warmed LB broth (pH 2.5, adjusted with HCl). The cfu ml−1 in PBS was determined by plating serial dilutions in PBS buffer (pH 7.2) on LB agar and using these as initial cell populations. The LB broth (pH 2.5) inoculated with E. coli was incubated at 37 °C for 1 h, and the cfu ml−1 in the LB broth (pH 2.5) was determined as described above and used as the final cell population. The percentage acid survival was then calculated as the number of cfu ml−1 remaining after acid treatment divided by the initial cfu ml−1 at time zero. Each experiment was performed in triplicate. The percentage survival values were converted to logarithmic values (log10 x, where x equals the percentage survival) for the calculation of geometric means and standard errors (s.e.).
Drug efflux assay
The drug efflux activities of E. coli cells were measured using cells preloaded with rhodamine 6G. The exponential cultures of E. coli cells were harvested and washed twice with 100 mM potassium phosphate buffer (pH 7.5) containing 5 mM MgSO4. For maximal accumulation of the fluorophore, the cells (optical density of 1.0 at 600 nm) were incubated with 1 μM rhodamine 6G and 40 μM carbonyl cyanide m-chlorophenylhydrazone (CCCP) at 37 °C for 1 h. The cells were then centrifuged, resuspended in the same medium with the addition of 25 mM glucose as an energy source, and subjected to fluorescence measurement. The fluorescence of the compound was continuously monitored using a Hitachi model F-2000 fluorescence spectrophotometer (Hitachi High-Technologies Corp., Tokyo, Japan). Rhodamine 6G transport was measured with excitation at 529 nm and emission at 553 nm.
Results
Overexpression of ydeO increases resistance to oxacillin
The expression of multi-drug efflux genes is often regulated in a complex manner, as described in the Introduction section. We therefore screened the genomic library of the E. coli for genes that increased multi-drug resistance levels in this organism. We screened a host strain lacking a functional acrB gene in order to identify regulatory elements involved in the expression of other multi-drug resistance systems. The library was developed from the chromosomal DNA of the MG1655 strain, and then, the recombinant plasmids were transformed into the ΔacrB strain NKE96 as described in the Materials and methods section. In one experiment, we found a 32-fold increase in oxacillin MIC against the transformant (data not shown). Introduction of the plasmid isolated from this strain into fresh ΔacrB cells resulted in the same oxacillin resistance phenotype: the MIC increased 32-fold over the recipient strain (data not shown).
Sequencing of the plasmid revealed an insertion containing the complete coding sequence of ydeO and a partial sequence of yneN. YdeO is the AraC–XylS family regulator that controls genes involved in acid resistance, such as glutamate decarboxylase genes (gadA and gadB) and a specific glutamate/GABA antiporter (gadC).34, 36 It seemed likely that overexpressed YdeO caused the transcriptional activation of genes involved in oxacillin resistance in the cells carrying this plasmid.
Full-length wild-type ydeO was cloned into the pHSG398 vector to obtain pydeO (Table 1). Oxacillin MICs for NKE96 cells harboring pydeO were 32 times higher (16 versus 0.5 μg ml−1) than that for cells harboring the pHSG398 vector (Table 3), suggesting that the YdeO regulator produced by this plasmid conferred oxacillin resistance on E. coli. Further experiments were therefore carried out with pydeO.
Overexpression of ydeO increases resistance to β-lactams, erythromycin, rhodamine 6G and sodium dodecyl sulfate (SDS)
Our results showed that overexpression of ydeO increased E. coli resistance to oxacillin, a β-lactam antibiotic. We therefore investigated the effect of ydeO overexpression on the susceptibility of E. coli to other β-lactams. pydeO also increased the resistance of ΔacrB cells to cloxacillin and nafcillin (Table 3). Various other drugs were tested, including common substrates of multi-drug efflux pumps, and we found that pydeO increased the resistance of the ΔacrB strain to erythromycin, rhodamine 6G and SDS (Table 3). These results indicate that the overproduced YdeO regulator induces the multi-drug resistance of E. coli.
Effect of tolC deletion on multi-drug resistance modulated by the YdeO regulator
The results described above indicate that the expression of a multi-drug efflux pump may be induced by overexpression of ydeO. In order to determine whether YdeO-mediated multi-drug resistance is attributable to TolC-dependent drug efflux pump(s), we investigated the effect of tolC deletion on drug resistance in cells overexpressing ydeO. Deletion of tolC from the ΔacrB strain increased susceptibility to many antimicrobial agents and chemical compounds, including cloxacillin, nafcillin, erythromycin and SDS (Table 3), which is in good agreement with earlier reports.12 The tolC deletion completely inhibited YdeO-mediated multi-drug resistance (Table 3). This result indicates that YdeO-mediated multi-drug resistance is attributable to increased expression of a TolC-dependent drug efflux pump.
Determination of the amounts of drug exporter transcripts by quantitative real-time RT-PCR (qRT-PCR)
In order to determine which drug efflux pump shows increased expression when ydeO is overexpressed, we used qRT-PCR to investigate changes in the amounts of drug exporter gene mRNAs. The results are shown in Figure 1. The expression levels of mdtE and mdtF were significantly increased (more than 10-fold in comparison with basal levels) by ydeO overexpression: 87- and 48-fold increases were observed for mdtE and mdtF, respectively. Overexpression of ydeO did not increase the expression of other drug exporter gene transcripts (Figure 1).
YdeO activates the expression of mdtEF multi-drug efflux genes. The amount of transcript was determined by quantitative real-time PCR as described in the Materials and methods section. The fold increase ratio was calculated by dividing the expression level of the gene in the ΔacrB/pydeO strain by that in the ΔacrB/vector strain. Experiments were performed in triplicate and the data are represented as mean±s.d.
Effects of deletion of the MdtEF drug efflux pump on YdeO-mediated multi-drug resistance
To determine whether the multi-drug resistance mediated by ydeO overexpression is because of increased expression of mdtEF, we investigated the effects of deleting these genes on drug resistance levels in ΔacrB/vector and ΔacrB/pydeO (Table 3). When mdtEF was deleted from the ΔacrB strain, there was no change in drug resistance in the resulting strains. In the ΔmdtEF acrB strain, overexpression of ydeO conferred no drug resistance (Table 3). Together, these data indicate that the multi-drug resistance conferred by the YdeO regulator is because of the increased expression of mdtEF multi-drug efflux genes.
Effects of YdeO and MdtEF on the acid resistance of E. coli
It was reported that YdeO activates the expression of genes involved in acid resistance.36 In this study, we found that YdeO induces the expression of mdtEF multi-drug efflux genes. To test whether MdtEF contributes to acid resistance modulated by YdeO, we measured survival percentage of the wild-type strain and the ΔmdtEF mutant harboring vector (pHSG398) or pydeO at pH 2.5 (Figure 2). Overexpression of ydeO enhances the acid resistance of both the wild-type strain and the ΔmdtEF mutant (Figure 2), indicating that the MdtEF multi-drug efflux pump is not essential for YdeO-induced acid resistance.
Effect of YdeO and MdtEF on acid resistance of E. coli. Acid resistance of the wild-type strain (WT) and the ΔmdtEF mutant harboring vector (pHSG398) or pydeO were grown to mid-log phase in LB broth (pH 7.0). Cells were diluted 40-fold using LB broth (pH 2.5) and incubated for 1 h at 37 °C. Error bars represent standard errors of the mean values.
YdeO enhances the MdtEF-dependent drug efflux activity of E. coli
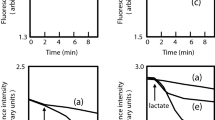
In this study, we found that YdeO activates the expression of mdtEF multi-drug efflux genes and drug resistance to E. coli cells. E. coli cells with a plasmid carrying the ydeO gene became resistant to rhodamine 6G, a toxic dye. This compound can be detected by its fluorescence. To determine whether the ydeO-induced drug resistance is because of efflux of this compound from the cells, we measured the efflux activity of rhodamine 6G from the cells. As shown in Figure 3, rapid efflux of rhodamine 6G from E. coli ΔacrB/pydeO cells was observed as an increase in fluorescence. However, no significant efflux was observed from ΔacrB and ΔacrB mdtEF/pydeO cells (Figure 3). These results indicate that the YdeO-induced multi-drug resistance is because of the enhanced drug efflux activity of E. coli cells, which is caused by the increased expression of mdtEF.
Discussion
In this study, we performed a genome-wide search for a regulator of multi-drug resistance of E. coli by random shotgun cloning and discovered YdeO, which up-regulates mdtEF expression, thereby increasing the resistance to β-lactams, erythromycin, rhodamine 6G and SDS. We initially found that the plasmid carrying ydeO conferred oxacillin resistance on the ΔacrB strain. Then, we investigated the susceptibility of the ydeO-overexpressing ΔacrB strain to various drugs, including the common substrates of multi-drug efflux pumps, and found that YdeO stimulates E. coli resistance to oxacillin, cloxacillin, nafcillin, erythromycin, rhodamine 6G and SDS (Table 3). We discovered the importance of YdeO as a drug resistance factor through induction of the multi-drug efflux gene. Earlier, we reported that the MdtEF multi-drug efflux pump requires TolC to function.12 YdeO-mediated multi-drug resistance was completely suppressed by deleting the tolC or mdtEF gene (Table 3). Although nothing is known of the potential YdeO-binding consensus sequence, it was reported that YdeO has an ability to bind the intergenic region between hdeD and gadE, located upstream of mdtEF.36 The gadE and mdtEF genes are located in tandem and they might be co-transcribed. These facts suggest that the up-regulation of mdtEF expression might have occurred by the YdeO-binding to the upstream region of gadE.
An important feature of E. coli pathogenesis is the organism's ability to withstand extremely acidic environments (pH 2 or lower). This acid resistance contributes to the low infectious dose of pathogenic E. coli species. One very efficient E. coli acid resistance system encompasses two isoforms of glutamate decarboxylase (gadA and gadB) and a putative glutamate/GABA antiporter (gadC).34, 35 It is subject to complex controls that vary with growth medium, growth phase and growth pH. Earlier work has revealed that this system is also controlled by YdeO (Figure 4).36 The YdeO protein is involved in transcriptional activation of the gadE gene coding for a protein that regulates gadABC (Figure 4).36, 46 YdeO belongs to the AraC–XylS family of bacterial transcriptional regulators known to activate acid resistance36, 47, 48 and repress virulence.49 We investigated the contribution of the MdtEF multi-drug efflux pump in YdeO-induced acid resistance, but MdtEF is not essential for acid resistance of E. coli.
Model for YdeO control of multi-drug resistance and acid resistance. YdeO controls expression of gadABCE acid resistance genes and mdtEF multi-drug efflux pump genes. The results of this study show that overexpression of ydeO activates expression of mdtEF and confers multi-drug resistance on E. coli.
In addition to the roles of YdeO in acid resistance, we found that it contributes to the multi-drug resistance of E. coli by activating the MdtEF multi-drug efflux pump (Figure 4). The present evidence suggests that it may enhance the resistance of E. coli to low pH and multiple drugs in hostile environments. Further investigation of the regulation of multi-drug efflux systems in several natural environments, such as inside hosts, is required to elucidate the biological significance of their regulatory networks. Such investigations may provide further insights into the role of multi-drug efflux systems in the physiology of the cell.
References
Nikaido, H. Multidrug efflux pumps of gram-negative bacteria. J. Bacteriol. 178, 5853–5859 (1996).
Zgurskaya, H. I. & Nikaido, H. Multidrug resistance mechanisms: drug efflux across two membranes. Mol. Microbiol. 37, 219–225 (2000).
Putman, M., van Veen, H. W. & Konings, W. N. Molecular properties of bacterial multidrug transporters. Microbiol. Mol. Biol. Rev. 64, 672–693 (2000).
Brown, M. H., Paulsen, I. T. & Skurray, R. A. The multidrug efflux protein NorM is a prototype of a new family of transporters. Mol. Microbiol. 31, 394–395 (1999).
Paulsen, I. T., Chen, J., Nelson, K. E. & Saier, M. H. Comparative genomics of microbial drug efflux systems. J. Mol. Microbiol. Biotechnol. 3, 145–150 (2001).
Murakami, S., Nakashima, R., Yamashita, E. & Yamaguchi, A. Crystal structure of bacterial multidrug efflux transporter AcrB. Nature 419, 587–593 (2002).
Yu, E. W., Aires, J. R. & Nikaido, H. AcrB multidrug efflux pump of Escherichia coli: composite substrate-binding cavity of exceptional flexibility generates its extremely wide substrate specificity. J. Bacteriol. 185, 5657–5664 (2003).
Murakami, S., Nakashima, R., Yamashita, E., Matsumoto, T. & Yamaguchi, A. Crystal structures of a multidrug transporter reveal a functionally rotating mechanism. Nature. 443, 173–179 (2006).
Nishino, K. & Yamaguchi, A. Analysis of a complete library of putative drug transporter genes in Escherichia coli. J. Bacteriol. 183, 5803–5812 (2001).
Ma, D., Cook, D. N., Hearst, J. E. & Nikaido, H. Efflux pumps and drug resistance in gram-negative bacteria. Trends Microbiol. 2, 489–493 (1994).
Nikaido, H. Prevention of drug access to bacterial targets: permeability barriers and active efflux. Science. 264, 382–388 (1994).
Nishino, K., Yamada, J., Hirakawa, H., Hirata, T. & Yamaguchi, A. Roles of TolC-dependent multidrug transporters of Escherichia coli in resistance to β-lactams. Antimicrob. Agents Chemother. 47, 3030–3033 (2003).
Elkins, C. A. & Nikaido, H. Substrate specificity of the RND-type multidrug efflux pumps AcrB and AcrD of Escherichia coli is determined predominantly by two large periplasmic loops. J. Bacteriol. 184, 6490–6498 (2002).
Fralick, J. A. Evidence that TolC is required for functioning of the Mar/AcrAB efflux pump of Escherichia coli. J. Bacteriol. 178, 5803–5805 (1996).
Nishino, K. & Yamaguchi, A. EvgA of the two-component signal transduction system modulates production of the yhiUV multidrug transporter in Escherichia coli. J. Bacteriol. 184, 2319–2323 (2002).
Baucheron, S. et al. AcrAB-TolC directs efflux-mediated multidrug resistance in Salmonella enterica serovar Typhimurium DT104. Antimicrob. Agents Chemother. 48, 3729–3735 (2004).
Moore, S. D. & Sauer, R. T. Revisiting the mechanism of macrolide-antibiotic resistance mediated by ribosomal protein L22. Proc. Natl Acad. Sci. USA 105, 18261–18266 (2008).
Paulsen, I. T., Sliwinski, M. K. & Saier, M. H. Microbial genome analyses: global comparisons of transport capabilities based on phylogenies, bioenergetics and substrate specificities. J. Mol. Biol. 277, 573–592 (1998).
Paulsen, I. T., Nguyen, L., Sliwinski, M. K., Rabus, R. & Saier, M. H. Microbial genome analyses: comparative transport capabilities in eighteen prokaryotes. J. Mol. Biol. 301, 75–100 (2000).
Nishino, K. Bacterial multidrug exporters: insights into acquisition of multidrug resistance. Science (online): [http://www.sciencemag.org/feature/data/prizes/ge/2004/nishino.dtl] (2005).
Ahmed, M., Borsch, C. M., Taylor, S. S., Vazquez-Laslop, N. & Neyfakh, A. A. A protein that activates expression of a multidrug efflux transporter upon binding the transporter substrates. J. Biol. Chem. 269, 28506–28513 (1994).
Brooun, A., Tomashek, J. J. & Lewis, K. Purification and ligand binding of EmrR, a regulator of a multidrug transporter. J. Bacteriol. 181, 5131–5133 (1999).
Lomovskaya, O., Lewis, K. & Matin, A. EmrR is a negative regulator of the Escherichia coli multidrug resistance pump EmrAB. J. Bacteriol. 177, 2328–2334 (1995).
Grkovic, S., Brown, M. H. & Skurray, R. A. Regulation of bacterial drug export systems. Microbiol. Mol. Biol. Rev. 66, 671–701 (2002).
Ma, D., Alberti, M., Lynch, C., Nikaido, H. & Hearst, J. E. The local repressor AcrR plays a modulating role in the regulation of acrAB genes of Escherichia coli by global stress signals. Mol. Microbiol. 19, 101–112 (1996).
Hirakawa, H. et al. AcrS/EnvR represses expression of the acrAB multidrug efflux genes in Escherichia coli. J. Bacteriol. 190, 6276–6279 (2008).
Randall, L. P. & Woodward, M. J. The multiple antibiotic resistance (mar) locus and its significance. Res. Vet. Sci. 72, 87–93 (2002).
Rosenberg, E. Y., Bertenthal, D., Nilles, M. L., Bertrand, K. P. & Nikaido, H. Bile salts and fatty acids induce the expression of Escherichia coli AcrAB multidrug efflux pump through their interaction with Rob regulatory protein. Mol. Microbiol. 48, 1609–1619 (2003).
Smith, J. L. The role of gastric acid in preventing foodborne disease and how bacteria overcome acid conditions. J. Food Prot. 66, 1292–1303 (2003).
Gorden, J. & Small, P. L. Acid resistance in enteric bacteria. Infect. Immun. 61, 364–367 (1993).
Lin, J. et al. Mechanisms of acid resistance in enterohemorrhagic Escherichia coli. Appl. Environ. Microbiol. 62, 3094–3100 (1996).
Giannella, R. A., Broitman, S. A. & Zamcheck, N. Influence of gastric acidity on bacterial and parasitic enteric infections. A perspective. Ann. Intern. Med. 78, 271–276 (1973).
Price, S. B., Wright, J. C., DeGraves, F. J., Castanie-Cornet, M. P. & Foster, J. W. Acid resistance systems required for survival of Escherichia coli O157:H7 in the bovine gastrointestinal tract and in apple cider are different. Appl. Environ. Microbiol. 70, 4792–4799 (2004).
Castanie-Cornet, M. P., Penfound, T. A., Smith, D., Elliott, J. F. & Foster, J. W. Control of acid resistance in Escherichia coli. J. Bacteriol. 181, 3525–3535 (1999).
De Biase, D., Tramonti, A., Bossa, F. & Visca, P. The response to stationary-phase stress conditions in Escherichia coli: role and regulation of the glutamic acid decarboxylase system. Mol. Microbiol. 32, 1198–1211 (1999).
Ma, Z., Masuda, N. & Foster, J. W. Characterization of EvgAS-YdeO-GadE branched regulatory circuit governing glutamate-dependent acid resistance in Escherichia coli. J. Bacteriol. 186, 7378–7389 (2004).
Blattner, F. R. et al. The complete genome sequence of Escherichia coli K-12. Science 277, 1453–1474 (1997).
Davis, R. W., Bolstein, D. & Roth, J. R. Advanced Bacterial Genetics, Cold Spring Harbor Laboratory Press: Cold Spring Harbor, NY, (1980).
Sambrook, J., Fritsch, E. F. & Maniatis, T. Molecular Cloning: A Laboratory Manual, 2nd edn. Cold Spring Harbor Laboratory: Cold Spring Harbor, NY, (1989).
Datsenko, K. A. & Wanner, B. L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl Acad. Sci. USA 97, 6640–6645 (2000).
Nishino, K. & Yamaguchi, A. Role of histone-like protein H-NS in multidrug resistance of Escherichia coli. J. Bacteriol. 186, 1423–1429 (2004).
Nishino, K., Honda, T. & Yamaguchi, A. Genome-wide analyses of Escherichia coli gene expression responsive to the BaeSR two-component regulatory system. J. Bacteriol. 187, 1763–1772 (2005).
Nishino, K. & Yamaguchi, A. Overexpression of the response regulator evgA of the two-component signal transduction system modulates multidrug resistance conferred by multidrug resistance transporters. J. Bacteriol. 183, 1455–1458 (2001).
Nishino, K., Inazumi, Y. & Yamaguchi, A. Global analysis of genes regulated by EvgA of the two-component regulatory system in Escherichia coli. J. Bacteriol. 185, 2667–2672 (2003).
Nishino, K., Latifi, T. & Groisman, E. A. Virulence and drug resistance roles of multidrug efflux systems of Salmonella enterica serovar Typhimurium. Mol. Microbiol. 59, 126–141 (2006).
Ma, Z. et al. GadE (YhiE) activates glutamate decarboxylase-dependent acid resistance in Escherichia coli K-12. Mol. Microbiol. 49, 1309–1320 (2003).
Gallegos, M. T., Schleif, R., Bairoch, A., Hofmann, K. & Ramos, J. L. Arac/XylS family of transcriptional regulators. Microbiol. Mol. Biol. Rev. 61, 393–410 (1997).
Martin, R. G. & Rosner, J. L. The AraC transcriptional activators. Curr. Opin. Microbiol. 4, 132–137 (2001).
Nadler, C., Shifrin, Y., Nov, S., Kobi, S. & Rosenshine, I. Characterization of enteropathogenic Escherichia coli mutants that fail to disrupt host cell spreading and attachment to substratum. Infect. Immun. 74, 839–849 (2006).
Acknowledgements
We thank Barry L Wanner for providing the strains and plasmids. This research was supported by a research aid from the Japan Antibiotics Research Association (KN); the Nakajima Foundation (KN); the Kao Foundation for Arts and Sciences (KN); the Program for Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation (AY and KN); a grant from the Ministry of Education, Culture, Sports, Science and Technology of Japan (KN); a Grant-in-Aid for Young Scientists (S) from the Japan Society for the Promotion of Science (KN); and PRESTO (KN), Japan Science and Technology Agency, Japan.
Author information
Authors and Affiliations
Corresponding author
Additional information
This study was supported by the 19th Yasushi Ueda Prize, which was awarded to K.N. in 2008. This paper is dedicated to the memory of Professor Yasushi Ueda.
Rights and permissions
About this article
Cite this article
Nishino, K., Senda, Y., Hayashi-Nishino, M. et al. Role of the AraC–XylS family regulator YdeO in multi-drug resistance of Escherichia coli. J Antibiot 62, 251–257 (2009). https://doi.org/10.1038/ja.2009.23
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ja.2009.23
Keywords
This article is cited by
-
Escherichia coli displays a conserved membrane proteomic response to a range of alcohols
Biotechnology for Biofuels and Bioproducts (2023)
-
Efficient biosynthesis of (R)-mandelic acid from styrene oxide by an adaptive evolutionary Gluconobacter oxydans STA
Biotechnology for Biofuels and Bioproducts (2023)
-
Improving heterologous polyketide production in Escherichia coli by transporter engineering
Applied Microbiology and Biotechnology (2015)
-
Enhancing isoprenoid production through systematically assembling and modulating efflux pumps in Escherichia coli
Applied Microbiology and Biotechnology (2013)