Abstract
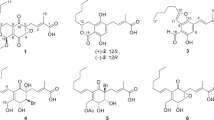
From a study on the biological activity of fungal stemphones and their derivatives, five derivatives having an O-alkyl moiety at C-11 of stemphone C were found to inhibit lipid droplet accumulation in macrophages without any cytotoxic effect. Among the derivatives, those having O-isopropyl and O-isobutyl were the most potent inhibitors by blocking the synthesis of both cholesteryl ester (CE) and triacylglycerol (TG), the main constituents of lipid droplets in macrophages.
Similar content being viewed by others
Article PDF
References
Koyama N, Nagahiro T, Yamaguchi Y, Masuma R, Tomoda H, Ōmura S . Stemphones, novel potentiators of imipenem activity against methicillin-resistant Staphylococcus aureus, produced by Aspergillus sp. FKI-2136. J Antibiot 58: 695–703 ( 2005)
Yamazaki H, Koyama N, Ōmura S . Tomoda H . Structureactivity relationships of stemphones, potentiators of imipenem activity against methicillin-resistant Staphylococcus aureus. J Antibiot 61: 426–441 ( 2008)
Namatame I, Tomoda H, Arai H, Inoue K, Ōmura S . Complete inhibition of mouse macrophage-derived foam cell formation by triacsin C. J Biochem 125: 319–327 ( 1999)
Tomoda H, Ōmura S . Potential therapeutics for obesity and atherosclerosis: Inhibitors of neutral lipid metabolism from microorganisms. Pharmacol Therapeut 115: 375–389 ( 2007)
Goldstein JL, Basu SK, Brown MS . Receptor-mediated endocytosis of low-density lipoprotein in cultured cells. Methods Enzymol 98: 241–260 ( 1983)
Berg T, Boman D, Seglen PO . Induction of tryptophan oxygenase in primary rat liver cell suspensions by glucocorticoid hormone. Exp Cell Res 72: 571–574 ( 1972)
Bligh EG, Dyer W . A rapid method of total lipid extraction and purification. Can J Biochem Physiol 37: 911–917 ( 1959)
Namatame I, Tomoda H, Ishibashi S, Ōmura S . Antiatherogenic activity of fungal beauveriolides, inhibitors of lipid droplet accumulation in macrophages. Proc Natl Acad Sci USA 101: 737–742 ( 2004)
Tomoda H, Ito M, Tabata N, Masuma R, Yamaguchi Y, Omura S . Amidepsines, inhibitors of diacylglycerol acyltransferase produced by Humicola sp. FO-2942. I. Production, isolation and biological properties. J Antibiot 48: 937–941 ( 1995)
Fujioka T, Yao K, Hamano K, Hosoya T, Kagasaki T, Furukawa Y, Haruyama H, Sato S, Koga T, Tsujita Y . Epi-cochlioquinone A, a novel acyl-CoA: cholesterol acyltransferase inhibitor produced by Stachybotrys bisbyi. J Antibiot 49: 409–413 ( 1996)
Schultz TW, Yarbrough JW, Hunter RS, Aptula AO . Verification of the structural alerts for Michael acceptors. Chem Res Toxicol 20: 1359–1363 ( 2007)
Cases S, Smith SJ, Zheng YW, Myers HM, Lear SR, Sande E, Novak S, Collins C, Welch CB, Lusis AJ, Erickson SK, Farese RV Jr . Identification of a gene encoding an acyl CoA: diacylglycerol acyltransferase, a key enzyme in triacylglycerol synthesis. Proc Natl Acad Sci USA 95: 13018–13023 ( 1998)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Koyama, N., Kobayashi, K., Yamazaki, H. et al. Inhibition of Lipid Droplet Accumulation in Mouse Macrophages by Stemphone Derivatives. J Antibiot 61, 509–514 (2008). https://doi.org/10.1038/ja.2008.68
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2008.68