Abstract
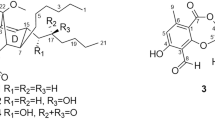
Directed bioassay guided fraction led to a new macrodiolide antimycin A17 (1), isolated from a Streptomyces sp. GAAS7310, which showed significant antimicrobial activity against eleven fungal species, including Curvularia lunata (Wakker) Boed, Rhizopus nigrtcans Ehrb and Colletotrichum nigrum EL. et Halst. The structure was unambiguously established by interpretation of 1D and 2D NMR data and comparison with the known antimycin A1a.
Similar content being viewed by others
Article PDF
References
Osada H . Actinomycetes: how fascinating microorganisms. Actinomycetologica 12: 85–88 ( 1998)
Takahashi Y, Ōmura S . Isolation of new actinomycete strains for the screening of new bioactive compounds. J Gen Appl Microbiol 49: 141–154 ( 2003)
Miyadoh S . Research on antibiotic screening in Japan over the last decade: a producing microorganism approach. Actinomycetologica 7: 100–106 ( 1993)
Shiomi K, Hatae K, Hatano H, Matsumoto A, Takahashi Y, Jiang CL, Tomoda H, Kobayashi S, Tanaka H, Ōmura S . A new antibiotic, antimycin A9, produced by Streptomyces sp. K01-0031. J Antibiot 58: 74–78 ( 2005)
Barrow CJ, Oleynek JJ, Marinelli V, Sun HH, Kaplita P, Sedlock DM, Gillum AM, Chadwick CC, Cooper R . Antimycins, inhibitors of ATP-citrate lyase, from a Streptomyces sp. J Antibiot 50: 729–733 ( 1997)
Dunshee BR, Leben C, Keitt GW, Strong FM . The isolation and properties of antimycin A. J Am Chem Soc 71: 2436 ( 1949)
Abidi SL . High-performance liquid chromatographic separation of subcomponents of antimycin A. J Chromatogr 447: 65–79 ( 1988)
Abidi SL, Adams BR . Proton and carbon-13 resonance designation of antimycin A1 by two-dimensional NMR spectroscopy. Magnetic Resonance in Chemistry 12: 1078–1080 ( 1987)
Kim H, Esser L, Hossain MB, Xia D, Yu CA, Rizo J, Helm D, Deisenhofer J . Structure of antimycin A1, a specific electron transfer inhibitor of ubiquinol-cytochrome c oxidoreductase. J Am Chem Soc 121: 4902–4903 ( 1999)
Tsunoda T, Nishii T, Yoshizuka M, Yamasaki C, Suzuki T, Ito S . A total synthesis of (−)-antimycin A3b . Tetrahedron Lett 41: 7667–7671 ( 2000)
Hosotani N, Kumagai K, Nakagawa H, Shimatani T, Saji I . Antimycins A10∼A16, seven new antimycin antibiotics produced by Streptomyces spp. SPA-10191 and SPA-8893. J Antibiot 58: 460–467 ( 2005)
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chen, G., Lin, B., Lin, Y. et al. A New Fungicide Produced by a Streptomyces sp. GAAS7310. J Antibiot 58, 519–522 (2005). https://doi.org/10.1038/ja.2005.70
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ja.2005.70
Keywords
This article is cited by
-
Marine Streptomyces sp. derived antimycin analogues suppress HeLa cells via depletion HPV E6/E7 mediated by ROS-dependent ubiquitin–proteasome system
Scientific Reports (2017)
-
A new xanthone derivative from the co-culture broth of two marine fungi (strain No. E33 and K38)
Chemistry of Natural Compounds (2011)
-
Antimycin A18 produced by an endophytic Streptomyces albidoflavus isolated from a mangrove plant
The Journal of Antibiotics (2010)
-
A Practical Total Synthesis of (+)-Antimycin A9
The Journal of Antibiotics (2007)