Abstract
Background/Objectives:
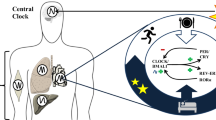
Humans carrying the genetic risk variant C at the circadian CLOCK (Circadian Locomotor Output Cycles Kaput) 3111T/C have been shown to have more difficulties to achieve desired weight loss than TT carriers. We tested the hypothesis that the daily rhythm of autonomic nervous function differs in CLOCK 3111C carriers, leading to reduced effectiveness in weight control.
Subjects/Methods:
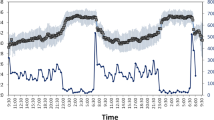
We recruited 40 overweight/obese Caucasian women (body mass index>25), 20 carrying CLOCK 3111C (CC and TC) and 20 non-carriers with matched age and body mass index who participated in a dietary obesity treatment program of up to 30 weeks. Following the treatment, ambulatory electrocardiography was continuously monitored for up to 3.5 days when subjects underwent their normal daily activities. To assess autonomic function, heart rate variability analysis (HRV) was performed hourly to obtain mean inter-beat interval between two consecutive R waves (mean RR) and s.d. of normal-to-normal heartbeat intervals (SDNN), and two parasympathetic measures, namely, proportion of differences between adjacent NN intervals that are >50 ms (pNN50), and high-frequency (HF: 0.15–0.4 Hz) power.
Results:
In the TT carriers, all tested HRV indices showed significant daily rhythms (all P-values <0.0001) with lower mean RR, SDNN, pNN50, and HF during the daytime as compared with the nighttime. The amplitudes of these rhythms except for SDNN were reduced significantly in the C carriers (mean RR: ~19.7%, P=0.001; pNN50: 58.1%, P=0.001; and HF: 41.1%, P=0.001). In addition, subjects with less weight loss during the treatment program had smaller amplitudes in the rhythms of mean RR (P<0.0001), pNN50 (P=0.007) and HF (P=0.003). Furthermore, the rhythmicity-weight loss associations were much stronger in the C carriers as compared to the TT carriers (mean RR: P=0.028, pNN50: P=0.0002; HF: P=0.015).
Conclusions:
The daily rhythm of parasympathetic modulation may play a role in the influence of the CLOCK variation on body weight control.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Garaulet M, Lee Y-C, Shen J, Parnell LD, Arnett DK, Tsai MY et al. CLOCK genetic variation and metabolic syndrome risk: modulation by monounsaturated fatty acids. Am J Clin Nutr 2009; 90: 1466–1475.
Garaulet M, Corbalán MD, Madrid JA, Morales E, Baraza JC, Lee YC et al. CLOCK gene is implicated in weight reduction in obese patients participating in a dietary programme based on the Mediterranean diet. Int J Obes 2005 2010; 34: 516–523.
Giovaninni NP, Fuly JT, Moraes LI, Coutinho TN, Trarbach EB, Jorge AA et al. Study of the association between 3111T/C polymorphism of the CLOCK gene and the presence of overweight in schoolchildren. J Pediatr (Rio J) 2014; 90: 500–505.
Bandín C, Martinez-Nicolas A, Ordovás JM, Ros Lucas JA, Castell P, Silvente T et al. Differences in circadian rhythmicity in CLOCK 3111T/C genetic variants in moderate obese women as assessed by thermometry, actimetry and body position. Int J Obes 2005 2013; 37: 1044–1050.
Galbete C, Contreras R, Martínez JA, Martínez-González MÁ, Guillén-Grima F, Marti A . Physical activity and sex modulate obesity risk linked to 3111T/C gene variant of the CLOCK gene in an elderly population: the SUN Project. Chronobiol Int 2012; 29: 1397–1404.
Garaulet M, Esteban Tardido A, Lee Y-C, Smith CE, Parnell LD, Ordovás JM . SIRT1 and CLOCK 3111T> C combined genotype is associated with evening preference and weight loss resistance in a behavioral therapy treatment for obesity. Int J Obes 2005 2012; 36: 1436–1441.
Garaulet M, Sánchez-Moreno C, Smith CE, Lee Y-C, Nicolás F, Ordovás JM . Ghrelin, sleep reduction and evening preference: relationships to CLOCK 3111 T/C SNP and weight loss. PloS One 2011; 6: e17435.
Monteleone P, Tortorella A, Docimo L, Maldonato MN, Canestrelli B, De Luca L et al. Investigation of 3111T/C polymorphism of the CLOCK gene in obese individuals with or without binge eating disorder: association with higher body mass index. Neurosci Lett 2008; 435: 30–33.
Ruiz-Lozano T, Vidal J, de Hollanda A, Canteras M, Garaulet M, Izquierdo-Pulido M . Evening-chronotype associates with obesity in severe obese subjects: interaction with CLOCK 3111T/C. Int J Obes 2005 2016; 40: 1550–1557.
Tsuzaki K, Kotani K, Sano Y, Fujiwara S, Takahashi K, Sakane N . The association of the Clock 3111 T/C SNP with lipids and lipoproteins including small dense low-density lipoprotein: results from the Mima study. BMC Med Genet 2010; 11: 150.
Reppert SM, Weaver DR . Coordination of circadian timing in mammals. Nature 2002; 418: 935–941.
Mohawk JA, Takahashi JS . Cell autonomy and synchrony of suprachiasmatic nucleus circadian oscillators. Trends Neurosci 2011; 34: 349–358.
Ozburn AR, Purohit K, Parekh PK, Kaplan GN, Falcon E, Mukherjee S et al. Functional Implications of the CLOCK 3111T/C Single-Nucleotide Polymorphism. Front Psychiatry 2016; 7: 67.
Eckel-Mahan K, Sassone-Corsi P . Metabolism and the circadian clock converge. Physiol Rev 2013; 93: 107–135.
Kalsbeek A, la Fleur S, Fliers E . Circadian control of glucose metabolism. Mol Metab 2014; 3: 372–383.
Garaulet M, Madrid JA . Chronobiology, genetics and metabolic syndrome. Curr Opin Lipidol 2009; 20: 127–134.
Scheer FAJL, Hilton MF, Mantzoros CS, Shea SA . Adverse metabolic and cardiovascular consequences of circadian misalignment. Proc Natl Acad Sci USA 2009; 106: 4453–4458.
Buxton OM, Cain SW, O’Connor SP, Porter JH, Duffy JF, Wang W et al. Adverse metabolic consequences in humans of prolonged sleep restriction combined with circadian disruption. Sci Transl Med 2012; 4: 129ra43.
Messina G, De Luca V, Viggiano A, Ascione A, Iannaccone T, Chieffi S et al. Autonomic nervous system in the control of energy balance and body weight: personal contributions. Neurol Res Int 2013; 2013: 639280.
Bray GA, York DA . The MONA LISA hypothesis in the time of leptin. Recent Prog Horm Res 1998; 53: 95–117-118.
Romijn JA, Fliers E . Sympathetic and parasympathetic innervation of adipose tissue: metabolic implications. Curr Opin Clin Nutr Metab Care 2005; 8: 440–444.
Kalsbeek A, Palm IF, La Fleur SE, Scheer F a. JL, Perreau-Lenz S, Ruiter M et al. SCN outputs and the hypothalamic balance of life. J Biol Rhythms 2006; 21: 458–469.
Shea SA, Hilton MF, Hu K, Scheer FAJL . Existence of an endogenous circadian blood pressure rhythm in humans that peaks in the evening. Circ Res 2011; 108: 980–U207.
Scheer FAJL, Michelson AD, Frelinger AL, Evoniuk H, Kelly EE, McCarthy M et al. The human endogenous circadian system causes greatest platelet activation during the biological morning independent of behaviors. PloS One 2011; 6: e24549.
Hu K, FAJL Scheer, Laker M, Smales C, Shea SA . Endogenous circadian rhythm in vasovagal response to head-up tilt. Circulation 2011; 123: 961–U85.
Scheer FAJL, Hu K, Evoniuk H, Kelly EE, Malhotra A, Hilton MF et al. Impact of the human circadian system, exercise, and their interaction on cardiovascular function. Proc Natl Acad Sci USA 2010; 107: 20541–20546.
Heart rate variability: standards of measurement, physiological interpretation and clinical use. Task Force of the European Society of Cardiology and the North American Society of Pacing and Electrophysiology. Circulation 1996; 93: 1043–1065.
Serra-Majem L, Aranceta J., SENC Working Group on Nutritional Objectives for the Spanish Population. Spanish Society of Community Nutrition. Nutritional objectives for the Spanish population. Consensus from the Spanish Society of Community Nutrition. Public Health Nutr 2001; 4: 1409–1413.
Friesen GM, Jannett TC, Jadallah MA, Yates SL, Quint SR, Nagle HT . A comparison of the noise sensitivity of nine QRS detection algorithms. IEEE Trans Biomed Eng 1990; 37: 85–98.
Chapleau MW, Sabharwal R . Methods of assessing vagus nerve activity and reflexes. Heart Fail Rev 2011; 16: 109–127.
Mietus JE, Peng C-K, Henry I, Goldsmith RL, Goldberger AL . The pNNx files: re-examining a widely used heart rate variability measure. Heart Br Card Soc 2002; 88: 378–380.
Licht CMM, Vreeburg SA, van Reedt Dortland AKB, Giltay EJ, Hoogendijk WJG, DeRijk RH et al. Increased sympathetic and decreased parasympathetic activity rather than changes in hypothalamic-pituitary-adrenal axis activity is associated with metabolic abnormalities. J Clin Endocrinol Metab 2010; 95: 2458–2466.
Chang C-J, Yang Y-C, Lu F-H, Lin T-S, Chen J-J, Yeh T-L et al. Altered cardiac autonomic function may precede insulin resistance in metabolic syndrome. Am J Med 2010; 123: 432–438.
Bandín C, Martinez-Nicolas A, Ordovás JM, Madrid JA, Garaulet M . Circadian rhythmicity as a predictor of weight-loss effectiveness. Int J Obes 2005 2014; 38: 1083–1088.
Delahanty LM, Pan Q, Jablonski KA, Watson KE, McCaffery JM, Shuldiner A et al. Genetic predictors of weight loss and weight regain after intensive lifestyle modification, metformin treatment, or standard care in the Diabetes Prevention Program. Diabetes Care 2012; 35: 363–366.
Locke AE, Kahali B, Berndt SI, Justice AE, Pers TH, Day FR et al. Genetic studies of body mass index yield new insights for obesity biology. Nature 2015; 518: 197–206.
Shungin D, Winkler TW, Croteau-Chonka DC, Ferreira T, Locke AE, Mägi R et al. New genetic loci link adipose and insulin biology to body fat distribution. Nature 2015; 518: 187–196.
Furlan R, Guzzetti S, Crivellaro W, Dassi S, Tinelli M, Baselli G et al. Continuous 24-hour assessment of the neural regulation of systemic arterial pressure and RR variabilities in ambulant subjects. Circulation 1990; 81: 537–547.
Kreier F, Yilmaz A, Kalsbeek A, Romijn JA, Sauerwein HP, Fliers E et al. Hypothesis: shifting the equilibrium from activity to food leads to autonomic unbalance and the metabolic syndrome. Diabetes 2003; 52: 2652–2656.
Buijs RM, Escobar C, Swaab DF . The circadian system and the balance of the autonomic nervous system. Handb Clin Neurol 2013; 117: 173–191.
Acknowledgements
We thank the research volunteers for their participation. We thank Yu-Hsin Tzeng for the help of ECG preprocessing, and Tommy To for the help with the figures. This study was supported by Spanish Ministry of Economy and Competitiveness (SAF2014-52480) (to MG), by National Science Council (NCU) of Taiwan grants 104-3115-E-008-001-, 103-2321-B-008-003-, 103-2221-E-008-006-MY3 (to MTL), and by joint foundation of CGH and NCU grants CNJRF-101CGH-NCU-A4 and VGHUST103-G1-3-3 (to MTL). KH was supported by NIH grants R00-HL102241, P01AG009975 and R01AG048108. FAJLS was supported in part by NIH grants R01 DK099512, R01 HL118601 and R01 DK102696.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on International Journal of Obesity website
Supplementary information
Rights and permissions
About this article
Cite this article
Lo, MT., Bandin, C., Yang, HW. et al. CLOCK 3111T/C genetic variant influences the daily rhythm of autonomic nervous function: relevance to body weight control. Int J Obes 42, 190–197 (2018). https://doi.org/10.1038/ijo.2017.168
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2017.168