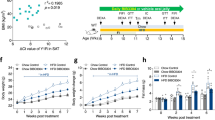
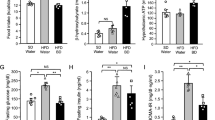
Abstract
Objective:
Fatty acid oxidation has been implicated in amelioration of obesity by burning off excessive accumulated lipid. BPR697, a peripheral cannabinoid receptor 1 (CB1) antagonist, elevated fat oxidation without added energy expenditure. Its impact on food intake, body weight changes and metabolic alterations were examined in rats fed standard chow and in diet-induced obesity (DIO) mice.
Materials and methods:
CB1 agonist-induced hypothermia and analgesia responses were measured to examine the brain activity of BPR697. The acute effects of BPR697 on food intake, body weight change and post-absorptive metabolic profiles were investigated in rats. Energy utilization with BPR697 was examined by indirect calorimetry. Chronic treatment of DIO mice was used to evaluate the long-term effects of BPR697.
Results:
Distribution of BPR697 was significantly biased in favor of the periphery instead of the brain, as shown by its low brain/plasma concentration ratio and confirmed by the negative response of BPR697 in CB1 agonist-induced hypothermia and analgesia. When administered to rats at 20 mg kg−1, BPR697 showed a unique spectrum of effects with significant weight loss without altered food intake. Furthermore, BPR697 increased serum levels of free fatty acids and ketone bodies and reduced hepatic lipid accumulation with preservation of liver glycogen in postprandial rats. Indirect calorimetric profiling of BPR697 revealed a similar trend, shifting whole-body energy catabolism toward fat oxidation, but without elevated energy expenditure. In DIO mice with chronic treatment, animals treated with BPR697 at 20 mg kg−1 resisted weight gain and showed a reduction of high-fat-induced cardiometabolic abnormalities such as hyperglycemia, abdominal fat and liver steatosis.
Conclusion:
The induction of fatty acid oxidation without concomitant elevation of energy expenditure by the peripheral CB1 antagonist BPR697 is sufficient to cause substantial weight loss in chow-fed rats. In the presence of high-dietary fat intake, BPR697 resists weight gain and alleviates obesity-related cardiometabolic risk factors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Flier JS . Obesity wars: molecular progress confronts an expanding epidemic. Cell 2004; 116: 337–350.
Rosen ED, Spiegelman BM . Adipocytes as regulators of energy balance and glucose homeostasis. Nature 2006; 444: 847–853.
Murphy KG, Bloom SR . Gut hormones and the regulation of energy homeostasis. Nature 2006; 444: 854–859.
Di Marzo V, Goparaju SK, Wang L, Liu J, Batkai S, Jarai Z et al. Leptin-regulated endocannabinoids are involved in maintaining food intake. Nature 2001; 410: 822–825.
Cota D, Marsicano G, Tschop M, Grubler Y, Flachskamm C, Schubert M et al. The endogenous cannabinoid system affects energy balance via central orexigenic drive and peripheral lipogenesis. J Clin Invest 2003; 112: 423–431.
Verty AN, Allen AM, Oldfield BJ . The effects of rimonabant on brown adipose tissue in rat: implications for energy expenditure. Obesity (Silver Spring) 2009; 17: 254–261.
Quarta C, Bellocchio L, Mancini G, Mazza R, Cervino C, Braulke LJ et al. CB(1) signaling in forebrain and sympathetic neurons is a key determinant of endocannabinoid actions on energy balance. Cell Metab 2010; 11: 273–285.
Osei-Hyiaman D, DePetrillo M, Pacher P, Liu J, Radaeva S, Batkai S et al. Endocannabinoid activation at hepatic CB1 receptors stimulates fatty acid synthesis and contributes to diet-induced obesity. J Clin Invest 2005; 115: 1298–1305.
Liu YL, Connoley IP, Wilson CA, Stock MJ . Effects of the cannabinoid CB1 receptor antagonist SR141716 on oxygen consumption and soleus muscle glucose uptake in Lep(ob)/Lep(ob) mice. Int J Obes Relat Metab Disord 2005; 29: 183–187.
Matias I, Gonthier MP, Orlando P, Martiadis V, De Petrocellis L, Cervino C et al. Regulation, function, and dysregulation of endocannabinoids in models of adipose and beta-pancreatic cells and in obesity and hyperglycemia. J Clin Endocrinol Metab 2006; 91: 3171–3180.
Pagotto U, Marsicano G, Cota D, Lutz B, Pasquali R . The emerging role of the endocannabinoid system in endocrine regulation and energy balance. Endocr Rev 2006; 27: 73–100.
Starowicz KM, Cristino L, Matias I, Capasso R, Racioppi A, Izzo AA et al. Endocannabinoid dysregulation in the pancreas and adipose tissue of mice fed with a high-fat diet. Obesity (Silver Spring) 2008; 16: 553–565.
Di Marzo V . The endocannabinoid system in obesity and type 2 diabetes. Diabetologia 2008; 51: 1356–1367.
Ravinet Trillou C, Arnone M, Delgorge C, Gonalons N, Keane P, Maffrand JP et al. Anti-obesity effect of SR141716, a CB1 receptor antagonist, in diet-induced obese mice. Am J Physiol Regul Integr Comp Physiol 2003; 284: R345–R353.
Addy C, Wright H, Van Laere K, Gantz I, Erondu N, Musser BJ et al. The acyclic CB1R inverse agonist taranabant mediates weight loss by increasing energy expenditure and decreasing caloric intake. Cell Metab 2008; 7: 68–78.
Herling AW, Kilp S, Elvert R, Haschke G, Kramer W . Increased energy expenditure contributes more to the body weight-reducing effect of rimonabant than reduced food intake in candy-fed Wistar rats. Endocrinology 2008; 149: 2557–2566.
Scheen AJ . CB1 receptor blockade and its impact on cardiometabolic risk factors: overview of the RIO programme with rimonabant. J Neuroendocrinol 2008; 20 (Suppl 1): 139–146.
Schreurs M, Kuipers F, van der Leij FR . Regulatory enzymes of mitochondrial beta-oxidation as targets for treatment of the metabolic syndrome. Obes Rev 2010; 11: 380–388.
Di Marzo V, Despres JP . CB1 antagonists for obesity—what lessons have we learned from rimonabant? Nat Rev Endocrinol 2009; 5: 633–638.
Kunos G, Osei-Hyiaman D, Bátkai S, Sharkey KM, Makriyannis A . Should peripheral CB1 cannabinoid receptors be selectively targeted for therapeutic gain? Trends Pharmacol Sci 2009; 30: 1–7.
Hung MS, Chang CP, Li TC, Yeh TK, Song JS, Lin Y et al. Discovery of 1-(2,4-dichlorophenyl)-4-ethyl-5-(5-(2-(4-(trifluoromethyl)phenyl)ethynyl)thiophen-2-yl)-N-(piperidin-1-yl)-1H-pyrazole-3-carboxamide as a potential peripheral cannabinoid-1 receptor inverse agonist. ChemMedChem 2010; 5: 1439–1443.
Lin Y, Shia KS, Hsiao WC, Hsieh WP, Yeh TK, Tseng SL et al. Biphasic suppression of appetite by cannabinoid CB1 receptor antagonists with distinct functional activities. Pharmacol Res 2010; 62: 337–343.
Folch J, Lees M, Sloane Stanley GH . A simple method for the isolation and purification of total lipids from animal tissues. J Biol Chem 1957; 226: 497–509.
Chan TM, Exton JH . A rapid method for the determination of glycogen content and radioactivity in small quantities of tissue or isolated hepatocytes. Anal Biochem 1976; 71: 96–105.
Howlett AC, Barth F, Bonner TI, Cabral G, Casellas P, Devane WA et al. International Union of Pharmacology. XXVII. Classification of cannabinoid receptors. Pharmacol Rev 2002; 54: 161–202.
Savage DB, Petersen KF, Shulman GI . Disordered lipid metabolism and the pathogenesis of insulin resistance. Physiol Rev 2007; 87: 507–520.
Gomez R, Navarro M, Ferrer B, Trigo JM, Bilbao A, Del Arco I et al. A peripheral mechanism for CB1 cannabinoid receptor-dependent modulation of feeding. J Neurosci 2002; 22: 9612–9617.
Nogueiras R, Veyrat-Durebex C, Suchanek PM, Klein M, Tschop J, Caldwell C et al. Peripheral, but not central, CB1 antagonism provides food intake-independent metabolic benefits in diet-induced obese rats. Diabetes 2008; 57: 2977–2991.
Tam J, Vemuri VK, Liu J, Batkai S, Mukhopadhyay B, Godlewski G et al. Peripheral CB1 cannabinoid receptor blockade improves cardiometabolic risk in mouse models of obesity. J Clin Invest 2010; 120: 2953–2966.
Randall PA, Vemuri VK, Segovia KN, Torres EF, Hosmer S, Nunes EJ et al. The novel cannabinoid CB1 antagonist AM6545 suppresses food intake and food-reinforced behavior. Pharmacol Biochem Behav 2010; 97: 179–184.
Herling AW, Gossel M, Haschke G, Stengelin S, Kuhlmann J, Muller G et al. CB1 receptor antagonist AVE1625 affects primarily metabolic parameters independently of reduced food intake in Wistar rats. Am J Physiol Endocrinol Metab 2007; 293: E826–E832.
Osei-Hyiaman D, Liu J, Zhou L, Godlewski G, Harvey-White J, Jeong WI et al. Hepatic CB1 receptor is required for development of diet-induced steatosis, dyslipidemia, and insulin and leptin resistance in mice. J Clin Invest 2008; 118: 3160–3169.
Ishac EJ, Jiang L, Lake KD, Varga K, Abood ME, Kunos G . Inhibition of exocytotic noradrenaline release by presynaptic cannabinoid CB1 receptors on peripheral sympathetic nerves. Br J Pharmacol 1996; 118: 2023–2028.
Savage DB, Choi CS, Samuel VT, Liu ZX, Zhang D, Wang A et al. Reversal of diet-induced hepatic steatosis and hepatic insulin resistance by antisense oligonucleotide inhibitors of acetyl-CoA carboxylases 1 and 2. J Clin Invest 2006; 116: 817–824.
Ravinet Trillou C, Delgorge C, Menet C, Arnone M, Soubrie P . CB1 cannabinoid receptor knockout in mice leads to leanness, resistance to diet-induced obesity and enhanced leptin sensitivity. Int J Obes Relat Metab Disord 2004; 28: 640–648.
Fox CS, Massaro JM, Hoffmann U, Pou KM, Maurovich-Horvat P, Liu CY et al. Abdominal visceral and subcutaneous adipose tissue compartments: association with metabolic risk factors in the Framingham Heart Study. Circulation 2007; 116: 39–48.
Liu J, Fox CS, Hickson DA, May WD, Hairston KG, Carr JJ et al. Impact of abdominal visceral and subcutaneous adipose tissue on cardiometabolic risk factors: the Jackson Heart Study. J Clin Endocrinol Metab 2010; 95: 5419–5426.
Despres JP, Ross R, Boka G, Almeras N, Lemieux I . Effect of rimonabant on the high-triglyceride/ low-HDL-cholesterol dyslipidemia, intraabdominal adiposity, and liver fat: the ADAGIO-Lipids trial. Arterioscler Thromb Vasc Biol 2009; 29: 416–423.
Yamamoto T, Yamaguchi H, Miki H, Kitamura S, Nakada Y, Aicher TD et al. A novel coenzyme A:diacylglycerol acyltransferase 1 inhibitor stimulates lipid metabolism in muscle and lowers weight in animal models of obesity. Eur J Pharmacol 2011; 650: 663–672.
Acknowledgements
We thank Dr Nan-Shih Liao at the Institute of Molecular Biology, Academia Sinica, Taiwan, for sharing the indirect calorimetry system, and Ms Wan-Ling Chung for providing technical support. This work was supported by grants from the National Health Research Institutes and National Science Council of the Republic of China, Taiwan (Grant NSC 98-2323-B-400-001 and NSC 98-2320-B-400-002-MY3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Vijayakumar, R., Lin, Y., Shia, KS. et al. Induction of fatty acid oxidation resists weight gain, ameliorates hepatic steatosis and reduces cardiometabolic risk factors. Int J Obes 36, 999–1006 (2012). https://doi.org/10.1038/ijo.2011.171
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2011.171
Keywords
This article is cited by
-
Anti-obesity therapy with peripheral CB1 blockers: from promise to safe(?) practice
International Journal of Obesity (2020)