Abstract
Objective:
The immediate cause of obesity is the massive deposition of subcutaneous and visceral fat attributing to the continuous proliferation and differentiation of preadipocytes. The identification of the underlying molecular mechanisms of preadipocytes differentiation is urgent, and will have an important role in plastic and reconstructive surgical procedures.
Methods:
Two small hairpin RNA (shRNA)-mediated RNA interference plasmids have been constructed on the basis of the activity of H1 promoter-driven expression vector psiRNA-hH1neo to suppress the expression of angiotensinogen (AGT) in human preadipocytes-visceral (HPA-v). Subsequently, glycerol-3-phosphate dehydrogenase (G3PDH) activity and intracytoplasmic lipids content were detected during the process of HPA-v differentiation.
Results:
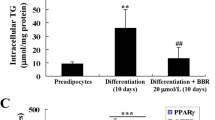
Small hairpin RNA-expressing vectors have been successfully constructed to suppress the expression of AGT significantly. Both intracytoplasmic lipids content and G3PDH activity decreased to a certain extent compared with that in the control group in the whole process of HPA-v differentiation.
Conclusions:
Two shRNA-mediated AGT-targeting plasmids inhibited the process of HPA-v differentiation to a certain extent. However, the accumulation of intracytoplasmic lipids was not exclusively determined by the expression of AGT, and it may also be regulated by other factors. In conclusion, this study provided a method to inhibit the process of preadipocytes differentiation, and it may have a role in obesity treatment and adipose tissue engineering application.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Beahm EK, Walton RL, Patrick Jr CW . Progress in adipose tissue construct development. Clin Plast Surg 2003; 30: 547–558.
Mathieu P, Pibarot P, Larose E, Poirier P, Marette A, Després JP . Visceral obesity and the heart. Int J Biochem Cell Biol 2008; 40: 821–836.
Aneja A, El-Atat F, McFarlane SI, Sowers JR . Hypertension and obesity. Recent Prog Horm Res 2004; 59: 169–205.
Van Gaal LF, Mertens IL, De Block CE . Mechanisms linking obesity with cardiovascular disease. Nature 2006; 444: 875–880.
Kahn SE, Hull RL, Utzschneider KM . Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 2006; 444: 840–846.
Langstein HN, Robb GL . Reconstructive approaches in soft tissue sarcoma. Semin Surg Oncol 1999; 17: 52–65.
Gomillion CT, Burg KJL . Stem cells and adipose tissue engineering. Biomaterials 2006; 27: 6052–6063.
Jones BH, Standridge MK, Moustaid N . Angiotensin II increases lipogenesis in 3T3-L1 and human adipose cells. Endocrinology 1997; 138: 1512–1519.
Darimont C, Vassaux G, Ailhaud G, Negrel R . Differentiation of preadipose cells: paracrine role of prostacyclin upon stimulation of adipose cells by angiotensin-II. Endocrinology 1994; 135: 2030–2036.
Saint-Marc P, Kozak LP, Ailhaud G, Darimont C, Negrel R . Angiotensin II as a trophic factor of white adipose tissue: stimulation of adipose cell formation. Endocrinology 2001; 142: 487–492.
Engeli S, Gorzelniak K, Kreutz R, Runkel N, Distler A, Sharma AM . Co-expression of renin-angiotensin system genes in human adipose tissue. J Hypertens 1999; 17: 555–560.
Jones BH, Standridge MK, Taylor JW, Moustaïd N . Angiotensinogen gene expression in adipose tissue: analysis of obese models and hormonal and nutritional control. Am J Physiol 1997; 273: R236–R242.
Paul M, Wagner J, Dzau VJ . Gene expression of the renin-angiotensin system in human tissues. Quantitative analysis by the polymerase chain reaction. J Clin Invest 1993; 91: 2058–2064.
Bader M, Peters J, Baltatu O, Müller DN, Luft FC, Ganten D . Tissue renin-angiotensin systems: new insights from experimental animal models in hypertension research. J Mol Med 2001; 79: 76–102.
Urata H, Healy B, Stewart RW, Bumpus FM, Husain A . Angiotensin II-forming pathways in normal and failing human hearts. Circ Res 1990; 66: 883–890.
Urata H, Kinoshita A, Misono KS, Bumpus FM, Husain A . Identification of a highly specific chymase as the major angiotensin II-forming enzyme in the human heart. J Biol Chem 1990; 265: 22348–22357.
Belova LA . Angiotensin II-generating enzymes. Biochemistry (Mosc) 2000; 65: 1337–1345.
Ye ZW, Wu XM, Jiang JG . Expression changes of angiotensin II pathways and bioactive mediators during human preadipocytes-visceral differentiation. Metabolism 2009; 58: 1288–1296.
Tong Q, Hotamisligil GS . Molecular mechanisms of adipocyte differentiation. Rev Endocr Metab Disord 2001; 2: 349–355.
Morrison RF, Farmer SR . Hormonal signaling and transcriptional control of adipocyte differentiation. J Nutr 2000; 130: 3116S–3121S.
Elbashir SM, Harborth J, Lendeckel W, Yalcin A, Weber K, Tuschl T . Duplexes of 21-nucleotide RNAs mediate RNA interference in cultured mammalian cells. Nature 2001; 411: 494–498.
Tuschl T . Expanding small RNA interference. Nat Biotechnol 2002; 20: 446–448.
Reynolds A, Leake D, Boese Q, Scaringe S, Marshall WS, Khvorova A . Rational siRNA design for RNA interference. Nat Biotechnol 2004; 22: 326–330.
Vandesompele J, De Preter K, Pattyn F, Poppe B, Van Roy N, De Paepe A et al. Accurate normalization of real-time quantitative RT-PCR data by geometric averaging of multiple internal control genes. Genome Biol 2002; 3, research0034.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method. Methods 2001; 25: 402–408.
Bradford MM . A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 1976; 72: 248–254.
Ramírez-Zacarías JL, Castro-Muñozledo F, Kuri-Harcuch W . Quantitation of adipose conversion and triglycerides by staining intracytoplasmic lipids with Oil red O. Histochemistry 1992; 97: 493–497.
Tijsterman M, Ketting RF, Plasterk RH . The genetics of RNA silencing. Annu Rev Genet 2002; 36: 489–519.
Mello CC, Conte Jr D . Revealing the world of RNA interference. Nature 2004; 431: 338–342.
Caplen NJ, Parrish S, Imani F, Fire A, Morgan RA . Specific inhibition of gene expression by small double-stranded RNAs in invertebrate and vertebrate systems. Proc Natl Acad Sci USA 2001; 98: 9742–9747.
Yang S, Tutton S, Pierce E, Yoon K . Specific double-stranded RNA interference in undifferentiated mouse embryonic stem cells. Mol Cell Biol 2001; 21: 7807–7816.
Hebert CG, Valdes JJ, Bentley WE . Beyond silencing—engineering applications of RNA interference and antisense technology for altering cellular phenotype. Curr Opin Biotechol 2008; 19: 500–505.
Fire A, Xu S, Montgomery MK, Kostas SA, Driver SE, Mello CC . Potent and specific genetic interference by double-stranded RNA in Caenorhabditis elegans. Nature 1998; 391: 806–811.
Deitel M . Overweight and obesity worldwide now estimated to involve 1.7 billion people. Obes Surg 2003; 13: 329–330.
Rodriguez AM, Elabd C, Delteil F, Astier J, Vernochet C, Saint-Marc P et al. Adipocyte differentiation of multipotent cells established from human adipose tissue. Biochem Biophys Res Commun 2004; 315: 255–263.
Cowherd RM, Lyle RE, McGehee Jr RE . Molecular regulation of adipocyte differentiation. Semin Cell Dev Biol 1999; 10: 3–10.
Lin FT, Lane MD . Antisense CCAAT/enhancer-binding protein RNA suppresses coordinate gene expression and triglyceride accumulation during differentiation of 3T3-L1 preadipocytes. Genes Dev 1992; 6: 533–544.
Freytag SO, Paielli DL, Gilbert JD . Ectopic expression of the CCAAT/enhancer-binding protein alpha promotes the adipogenic program in a variety of mouse fibroblastic cells. Genes Dev 1994; 8: 1654–1663.
Ron D, Brasier AR, Habener JF . Transcriptional regulation of hepatic angiotensinogen gene expression by the acute-phase response. Mol Cell Endocrinol 1990; 74: C97–C104.
Ailhaud G, Fukamizu A, Massiera F, Negrel R, Saint-Marc P, Teboul M . Angiotensinogen, angiotensin II and adipose tissue development. Int J Obes 2000; 24: S33–S35.
Schling P, Löffler G . Effects of angiotensin II on adipose conversion and expression of genes of the renin-angiotensin system in human preadipocytes. Horm Metab Res 2001; 33: 189–195.
Janke J, Engeli S, Gorzelniak K, Luft FC, Sharma AM . Mature adipocytes inhibit in vitro differentiation of human preadipocytes via angiotensin type 1 receptors. Diabetes 2002; 51: 1699–1707.
Saiki A, Koide N, Watanabe F, Murano T, Miyashita Y, Shirai K . Suppression of lipoprotein lipase expression in 3T3-L1 cells by inhibition of adipogenic differentiation through activation of the renin-angiotensin system. Metabolism 2008; 57: 1093–1100.
Cook JR, Kozak LP . Sn-glycerol-3-phosphate dehydrogenase gene expression during mouse adipocyte development in vivo. Dev Biol 1982; 92: 440–448.
Ryall RL, Goldrick RB . Glycerokinase in human adipose tissue. Lipids 1977; 12: 272–277.
Swierczynski J, Zabrocka L, Goyke E, Raczynska S, Adamonis W, Sledzinski Z . Enhanced glycerol 3-phosphate dehydrogenase activity in adipose tissue of obese humans. Mol Cell Biochem 2003; 254: 55–59.
Acknowledgements
This project was supported by the National Natural Foundation of China (Grant 30870025).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ye, ZW., Wu, XM. & Jiang, JG. Knockdown of angiotensinogen by shRNA-mediated RNA interference inhibits human visceral preadipocytes differentiation. Int J Obes 34, 157–164 (2010). https://doi.org/10.1038/ijo.2009.197
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ijo.2009.197