Abstract
Masked uncontrolled hypertension (MUCH) has been proven to be associated with increased cardiovascular risk in the general population. We performed the current analysis to determine its prevalence in dialysis patients and its association with pulse wave velocity (PWV). From 368 participants of another cohort study, we selected 145 subjects with controlled predialysis blood pressure (BP). All subjects underwent ambulatory BP monitoring and PWV measurement. MUCH was defined as controlled predialysis BP with daytime BP⩾135/85 mm Hg (definition-1); total ambulatory BP⩾130/80 mm Hg (definition-2); and either daytime BP⩾135/85 mm Hg or nighttime BP⩾120/70 mm Hg (definition-3). The prevalence of MUCH was 43.4% (definition-1), 55.9% (definition-2) and 74.5% (definition-3). Multivariable logistic regression analysis showed that the use of antihypertensive medication was the most consistent predictor of MUCH within all 3 definitions (all odds ratio (OR)⩾4.28, P<0.001). Predialysis systolic BP (both OR>1, P⩽0.04), predialysis diastolic BP (both OR>1, P⩽0.001) and hemoglobin (both OR<1, P=0.02) were all significantly associated with MUCH in two models. Interdialytic weight gain (OR=0.52, P=0.02) was associated with MUCH under definition-2, and BMI (OR=0.86, P=0.03) was associated with MUCH under definition-3. Patients with MUCH had significantly elevated PWV compared with their counterparts according to all three definitions with or without adjusting for covariates (all P⩽0.03). In conclusion, MUCH affects a large proportion of dialysis patients with controlled predialysis BP and is associated with increased PWV. Patients on antihypertensive medications and with higher predialysis BP are more likely to have MUCH.
Similar content being viewed by others
Introduction
Masked hypertension refers to those with normal clinic blood pressure (BP) but elevated ambulatory BP. It is the opposite of white-coat hypertension and has been proven to be associated with increased cardiovascular risk.1, 2 Conventionally, this term particularly applies to individuals who are untreated. For patients who are already diagnosed with hypertension, the new term masked uncontrolled hypertension (MUCH), coined by Banegas et al.,3 is currently accepted and adopted by clinical researchers.4, 5, 6, 7, 8 For patients on maintenance hemodialysis, MUCH is a more accurate diagnosis since these patients usually undergo a long course of kidney disease, and management of BP is a critical aspect of CKD management, and, therefore, the patients are unlikely to be untreated.
For dialysis patients, the BP target recommended by the K/DOQI guideline was based on dialysis-unit BP.9 However, routine BP measured in the dialysis department has been proven to be inconsistent with interdialytic ambulatory BP.10 Moreover, similar to that in the general population, ambulatory BP is more informative of a patient’s prognosis.11 Therefore, performing ambulatory BP monitoring in these patients is valuable and will reveal a group of patients with controlled dialysis-unit BP but elevated ambulatory BP, which is known as MUCH.
Research on dialysis patients with MUCH is currently insufficient. Here, we performed a cross-sectional analysis in a cohort of dialysis patients to determine the prevalence of MUCH in patients with controlled predialysis BP using various definitions and the clinical variables associated with MUCH. Additionally, we hypothesized that, in these patients, MUCH is associated with increased pulse wave velocity (PWV), which is a measure of arterial stiffness.
Methods
Study population
This is a post hoc analysis of baseline data from an ongoing cohort study that aimed to evaluate vascular dysfunction in dialysis patients. The study included 368 patients on maintenance hemodialysis aged 18–80 years. Detailed inclusion and exclusion criteria together with a full list of coordinating centers are presented in the Supplementary Information. The current analysis included only those with controlled predialysis BP. The study protocol was approved by the Institutional Ethics Committee of The Second Affiliated Hospital of Nanjing Medical University. All participants provided written informed consent.
Blood pressure
Using data from the dialysis records, predialysis BP was averaged over 2 weeks (six dialysis treatments) before inclusion. Predialysis BP measurements in the coordinating centers were performed by dialysis-unit staff members using automatic devices usually 5–10 min before a dialysis session. Patients with predialysis BP⩽140/90 mm Hg were considered to have controlled clinic BP.
Ambulatory monitoring of BP using a SpaceLabs 90217 monitor (SpaceLabs Medical, Redmond, WA, USA) began on a midweek nondialysis day after PWV measurement and was terminated before the next dialysis session. BP was measured at 20-minute intervals in the daytime (6:00 am–10:00 pm) and at 30-minute intervals in the nighttime (10:00 pm–6:00 am). When collecting the monitors, the physicians recorded the patients’ self-reported bedtime and wake time for the determination of daytime and nighttime periods for data analysis. The recordings were downloaded using the manufacturer’s software (SpaceLabs Report Manager System) and were further extracted and analyzed by SPSS 19.0 (SPSS, Chicago, IL, USA). At least 12 measurements in the daytime and 6 in the nighttime were required for a valid ABPM.
Definitions of MUCH
Currently, there is no general agreement on the standards for MUCH diagnosis. In the report where the term ‘MUCH’ was first introduced, Banegas et al. defined MUCH as 24-hour BP⩾130/80 mm Hg with controlled clinic BP (<140/90 mm Hg). However, this definition did not consider isolated nocturnal hypertension. Given that blunted BP circadian rhythm and nocturnal hypertension were highly prevalent in dialysis patients and had the ability to predict adverse events,12, 13, 14 we defined MUCH in three ways as adopted by Agarwal et al.:5
Definition-1: Predialysis BP<140/90 mm Hg with daytime BP⩾135/85 mm Hg;
Definition-2: Predialysis BP<140/90 mm Hg with total ambulatory BP⩾130/80 mm Hg; and
Definition-3: Predialysis BP<140/90 mm Hg with either daytime BP⩾135/85 mm Hg or nighttime BP⩾120/70 mm Hg.
Pulse wave velocity
Carotid-femoral PWV was determined on a midweek nondialysis day using the Complior Analyzer device (Artech Medical, France). Caffeine, meals and nitrates were not allowed within 2 h, and long-acting nitrates were not permitted for 12 h before measurement. Patients rested for over 10 min, and then, an experienced technician from the Second Affiliated Hospital of Nanjing Medical University performed the test for all participants. Any measurement with inconsistent waveform and a tolerance value >3 ms was considered invalid. Arterial stiffness, as measured by PWV, is a proven type of hypertension-associated organ damage and has been widely used in various studies, including those on masked hypertension.15, 16, 17, 18, 19
Data collection
General information, including demographic characteristics and medical information, were determined using the combination of patient interviews and medical records. Height was measured before PWV evaluation. Routine biochemical tests were performed in laboratory departments at each center using blood samples drawn from venous access just before dialysis treatment on the second or last dialysis day of the week. C-reactive protein was measured using the Milliplex Map assay (Merck Millipore, Shanghai, China).
Statistical analyses
Numerical variables were presented as the mean±s.d. or the median (interquartile range) and were transformed into logarithmic values if the distribution was skewed. Categorical variables were expressed as counts (%). Comparisons between the two groups were done using Student’s t-test, the Mann–Whitney U-test or the χ2 test, as appropriate. For adjusted comparisons, analysis of covariance was used. To compare PWV between true normotensive and MUCH patients, we first performed multiple stepwise regression analysis with PWV as the dependent variable in the study population and found that besides BP parameters, independent determinants of PWV included age and diabetes mellitus. Since age and the prevalence of diabetes mellitus did not differ significantly between normotensive patients and MUCH, we first compared PWV between groups without any adjustment. To further account for the potential confounding effect of cardiovascular risk factors and to test the stability of the model, we then adjusted extensively for these covariates (including age, sex, body mass index (BMI), smoking status, interdialytic weight gain, diabetes mellitus, history of cardiovascular disease, use of antihypertensive medication, use of statins, total cholesterol, high-density lipoprotein cholesterol and log-transformed C-reactive protein). Clinical variables, including predialysis BP, with a P-value ⩽0.10 in the crude comparison between patients with and without MUCH were entered into a multivariable logistic regression model using the stepwise forward method for the determination of independent correlates of MUCH. All statistical analyses were performed using SPSS 19.0 (IBM SPSS, Chicago, IL, USA). Figures were generated by GraphPad Prism 6.0 (GraphPad Software, San Diego, CA, USA). A two-tailed P-value <0.05 was considered statistically significant.
Results
A total of 145 subjects with controlled predialysis BP were selected from 368 participants of the cohort for the current analysis (Figure 1). Compared with the original cohort, subjects included in the current analysis showed lower predialysis and ambulatory BP (predialysis systolic BP: 124.7±11.1 mm Hg vs. 140.5±19.7 mm Hg; predialysis diastolic BP: 75.2±8.6 mm Hg vs. 83.0±11.9 mm Hg; ambulatory systolic BP: 124.0±16.4 mm Hg vs. 138.7±22.9 mm Hg; and ambulatory diastolic BP: 77.5±9.5 mm Hg vs. 84.5±12.8 mm Hg; all P<0.001), decreased PWV (9.7±2.7 m s−1 vs. 10.6±3.5 m s−1; P=0.005) and were more likely to be female (53.1 vs. 43.5%; P=0.049) and less likely to use antihypertensive medication (55.9 vs. 69.6%; P=0.003).
Using the three definitions described above, the prevalence of MUCH in these clinic BP controlled patients was 43.4% (definition-1), 55.9% (definition-2) and 74.5% (definition-3; Figure 2).
General characteristics of normotensive patients and MUCH patients, who were diagnosed according to the various definitions, are summarized in Table 1. Age and sex ratios were comparable between normotensive patients and patients with MUCH. BMI was significantly higher in normotensive patients under definition-2 and -3 (P=0.04 and 0.009, respectively). Patients with MUCH had lower interdialytic weight gain (all P⩽0.03) and hemoglobin levels (all P⩽0.05), and the prevalence of antihypertensive drug use was greater (all P<0.001). There was a marginal increase in high-density-lipoprotein cholesterol level and serum parathyroid hormone concentration in patients with MUCH under definition-3 (both P=0.04). No additional significant differences between groups were observed regarding other parameters.
Predialysis and ambulatory BP are presented in Table 2. Compared with normotensive patients, patients with MUCH had significantly higher predialysis BP (all P⩽0.001), and as expected, they also had significantly higher ambulatory BP (including total, daytime and nighttime BP; all P<0.001).
We next explored clinical associates of MUCH using multivariable logistic regression analysis, the results of which are shown in Table 3. The variables independently associated with MUCH were varied across different definitions. The use of antihypertensive medication was the most consistent factor in all three models (OR=4.28, 5.83 and 8.07 for definition-1, 2 and 3, respectively; all P<0.001). Predialysis systolic BP (both OR>1, P⩽0.04), predialysis diastolic BP (both OR>1, P⩽0.001) and hemoglobin (both OR<1, P=0.02) were all significantly associated with MUCH in two models. Interdialytic weight gain (OR=0.52, P=0.02) was associated with MUCH under definition-2, and BMI (OR=0.86, P=0.03) was associated with MUCH under definition-3.
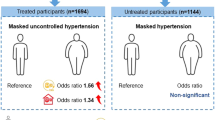
PWV was compared between the two groups with or without adjustment (Figure 3). In the crude comparison, patients with MUCH had significantly elevated PWV compared with their counterparts under all three definitions of MUCH (all P⩽0.01). After adjusting for age, sex, BMI, smoking status, interdialytic weight gain, diabetes mellitus, history of cardiovascular disease, use of antihypertensive drugs, use of statins, total cholesterol, high-density lipoprotein cholesterol and log-transformed C-reactive protein, the results remained qualitatively unchanged (all P⩽0.03).
Elevated pulse wave velocity in patients with masked uncontrolled hypertension. (a) Unadjusted PWV. (b) PWV adjusted for age, sex, body mass index, smoking status, interdialytic weight gain, diabetes mellitus, history of cardiovascular disease, use of antihypertensive drugs, use of statins, total cholesterol, high-density lipoprotein cholesterol and log-transformed CRP. CRP, C-reactive protein; PWV, pulse wave velocity.
Discussions
In a group of dialysis patients with controlled predialysis BP, we demonstrated the following: (1). The prevalence of MUCH was high and varied from 43.4 to 74.5% depending on the different definitions; (2). Patients on antihypertensive medication and those with higher predialysis BP were more susceptible to MUCH; and (3). MUCH was associated with increased PWV.
The definitions of MUCH varied in previous studies. When Banegas et al. first proposed the term MUCH in their 2014 report, they defined it as 24-hour BP⩾130/80 mm Hg in people with treated and controlled clinic BP (<140/90 mm Hg), which corresponds to definition-2 in our analysis, and they observed a prevalence of 31.1% in 14 840 treated hypertensive patients.3 Before their introduction of this concept, masked hypertension was instead used to describe a similar phenomenon, and it was usually diagnosed based on daytime ambulatory BP⩾135/85 mm Hg, which corresponds to definition-1 in the present study.20 Based on this definition, ~1/5 subjects in the general population had MUCH in two independent studies (23% by Selenta et al. and 20.6% by Franklin et al.).21, 22 Noticeably, the aforementioned studies reported the prevalence of MUCH (or masked hypertension) in those with controlled (treated) or normal (treatment-naive) clinic BP, while in other studies, the prevalence of MUCH was determined within subjects with either normal or elevated clinic BP. With the definition of masked hypertension as office BP<140/90 mm Hg and 24-hour BP⩾130/80 mm Hg, Gorostidi et al. and Drawz et al. reported the prevalence of MUCH as 32.1 and 36.1%, respectively, in CKD patients with controlled clinic BP.19, 23 In patients on hemodialysis, the prevalence was 30.5% in those with normal dialysis-unit BP when masked hypertension was defined as 44-hour ambulatory BP⩾135/85 mm Hg and median midweek intradialytic BP<140/80 mm Hg.24 Our analysis demonstrated that, based on various definitions, the prevalence ranged from 43.4 to 74.5%, indicating that at least >2/5 dialysis patients with controlled dialysis-unit BP actually have elevated ambulatory BP, and the ratio could be as high as ~3/4 given a broader definition. This finding was similar to that shown by Agarwal et al. in dialysis-independent CKD patients: the prevalence was 26.7% by daytime ambulatory BP and increased to 56.1% by either daytime or nighttime ambulatory BP.5
Our data also showed that the occurrence of MUCH in these patients can be predicted by several clinical characteristics. The most effective predictor of MUCH in these dialysis patients was the use of antihypertensive medications. The reasons for this probably include the following: (1) normal clinic BP without the need for antihypertensive medication may indicate that the patient is naïve from hypertension and is thus less likely to have elevated ambulatory BP; and (2) the drugs used to achieve controlled clinic BP are incapable of achieving controlled BP within a whole 24-hour period, especially during the nighttime.25, 26, 27 Higher predialysis BP was also independently associated with MUCH. This result is consistent with that reported by Agarwal et al. in CKD patients. They found that the prevalence of MUCH positively increased with the levels of clinic BP.5 It is worth noting that intravenous use of erythropoietin can increase interdialytic BP and may thus contribute to MUCH.28 However, we did not observe a difference in erythropoietin use between NT and MUCH patients. Further studies should attempt to specify whether erythropoietin use during dialysis just before ABPM contributes to MUCH.
According to our analysis, MUCH was associated with reduced hemoglobin level and lower BMI, which are both predictors of adverse outcome in dialysis patients.29, 30 Lower interdialytic weight gain was also a predictor for MUCH. Although volume status is usually a factor associated with BP, previous data have suggested that interdialytic weight gain has a very modest effect on hypertension in dialysis patients.31 The significant association observed in the present study may be a false positive finding since it was present in only one of the three models.
Arterial stiffness, as measured by PWV, is a classical sign of hypertensive target-organ damage. Prior studies have confirmed its role in predicting prognosis in dialysis patients.32, 33 In the current analysis, we demonstrated that MUCH in dialysis patients was associated with elevated PWV even after extensive adjustments with cardiovascular covariates. This finding highlights the detrimental effect of MUCH on the cardiovascular system and is line with previous data suggesting that ambulatory BP is more accurate in predicting outcome than dialysis-unit BP.11
Some limitations of our study should be noted for the accurate interpretation of our data. First, the cross-sectional design precludes any causality inference, and the sample size was limited. Second, we extracted dialysis-unit BP data from dialysis records instead of performing standardized predialysis BP measurements. This may be less precise in evaluating predialysis BP levels. However, this approach reflects every-day clinical practice, using data that physicians would consider in their determination of hypertension status in dialysis patients.
In conclusion, MUCH affects a large proportion of dialysis patients with controlled predialysis BP and was associated with increased PWV. Patients on antihypertensive medication and with higher predialysis BP were more likely to have MUCH. Attention should be paid to these patients in clinical practice.
References
Mancia G, Facchetti R, Bombelli M, Grassi G, Sega R . Long-term risk of mortality associated with selective and combined elevation in office, home, and ambulatory blood pressure. Hypertension 2006; 47: 846–853.
Fagard RH, Cornelissen VA . Incidence of cardiovascular events in white-coat, masked and sustained hypertension versus true normotension: a meta-analysis. J Hypertens 2007; 25: 2193–2198.
Banegas JR, Ruilope LM, de la Sierra A, de la Cruz JJ, Gorostidi M, Segura J, Martell N, Garcia-Puig J, Deanfield J, Williams B . High prevalence of masked uncontrolled hypertension in people with treated hypertension. Eur Heart J 2014; 35: 3304–3312.
Sabater-Hernandez D, Sendra-Lillo J, Jimenez-Monleon JJ, Benrimoj SI, Durks D, Martinez-Martinez F, de la Sierra A, MEPAFAR study workgroup. Identifying masked uncontrolled hypertension in the community pharmacy setting. Blood Press Monit 2015; 20: 138–143.
Agarwal R, Pappas MK, Sinha AD . Masked uncontrolled hypertension in CKD. J Am Soc Nephrol 2016; 27: 924–932.
Leiria LF, Severo MD, Ledur PS, Becker AD, Aguiar FM, Massierer D, Freitas VC, Schaan BD, Gus M . White coat effect and masked uncontrolled hypertension in treated hypertensive-diabetic patients: prevalence and target organ damage. J Diabetes 2015; 7: 699–707.
Naser N, Dzubur A, Durak A, Kulic M, Naser N . Blood pressure control in hypertensive patients, cardiovascular risk profile and the prevalence of masked uncontrolled hypertension (MUCH). Med Arch 2016; 70: 274–279.
Gorostidi M, Vinyoles E, Banegas JR, de la Sierra A . Prevalence of white-coat and masked hypertension in national and international registries. Hypertens Res 2015; 38: 1–7.
Workgroup KD . K/DOQI clinical practice guidelines for cardiovascular disease in dialysis patients. Am J Kidney Dis 2005; 45: S1–153.
Agarwal R, Peixoto AJ, Santos SF, Zoccali C . Pre- and postdialysis blood pressures are imprecise estimates of interdialytic ambulatory blood pressure. Clin J Am Soc Nephrol 2006; 1: 389–398.
Agarwal R . Blood pressure and mortality among hemodialysis patients. Hypertension 2010; 55: 762–768.
Santos SF, Mendes RB, Santos CA, Dorigo D, Peixoto AJ . Profile of interdialytic blood pressure in hemodialysis patients. Am J Nephrol 2003; 23: 96–105.
Amar J, Vernier I, Rossignol E, Bongard V, Arnaud C, Conte JJ, Salvador M, Chamontin B . Nocturnal blood pressure and 24-hour pulse pressure are potent indicators of mortality in hemodialysis patients. Kidney Int 2000; 57: 2485–2491.
Tripepi G, Fagugli RM, Dattolo P, Parlongo G, Mallamaci F, Buoncristiani U, Zoccali C . Prognostic value of 24-hour ambulatory blood pressure monitoring and of night/day ratio in nondiabetic, cardiovascular events-free hemodialysis patients. Kidney Int 2005; 68: 1294–1302.
Cecelja M, Chowienczyk P . Dissociation of aortic pulse wave velocity with risk factors for cardiovascular disease other than hypertension: a systematic review. Hypertension 2009; 54: 1328–1336.
Laszlo A, Reusz G, Nemcsik J . Ambulatory arterial stiffness in chronic kidney disease: a methodological review. Hypertens Res 2016; 39: 192–198.
Tientcheu D, Ayers C, Das SR, McGuire DK, de Lemos JA, Khera A, Kaplan N, Victor R, Vongpatanasin W . Target organ complications and cardiovascular events associated with masked hypertension and white-coat hypertension: analysis from the Dallas heart study. J Am College Cardiol 2015; 66: 2159–2169.
Hanninen MR, Niiranen TJ, Puukka PJ, Kesaniemi YA, Kahonen M, Jula AM . Target organ damage and masked hypertension in the general population: the Finn-Home study. J Hypertens 2013; 31: 1136–1143.
Drawz PE, Alper AB, Anderson AH, Brecklin CS, Charleston J, Chen J, Deo R, Fischer MJ, He J, Hsu CY, Huan Y, Keane MG, Kusek JW, Makos GK, Miller ER 3rd, Soliman EZ, Steigerwalt SP, Taliercio JJ, Townsend RR, Weir MR, Wright JT Jr, Xie D, Rahman M, Chronic Renal Insufficiency Cohort Study I. Masked hypertension and elevated nighttime blood pressure in CKD: prevalence and association with target organ damage. Clin J Am Soc Nephrol 2016; 11: 642–652.
Pickering TG, Davidson K, Gerin W, Schwartz JE . Masked hypertension. Hypertension 2002; 40: 795–796.
Selenta C, Hogan BE, Linden W . How often do office blood pressure measurements fail to identify true hypertension? An exploration of white-coat normotension. Arch Family Med 2000; 9: 533–540.
Franklin SS, Thijs L, Li Y, Hansen TW, Boggia J, Liu Y, Asayama K, Bjorklund-Bodegard K, Ohkubo T, Jeppesen J, Torp-Pedersen C, Dolan E, Kuznetsova T, Stolarz-Skrzypek K, Tikhonoff V, Malyutina S, Casiglia E, Nikitin Y, Lind L, Sandoya E, Kawecka-Jaszcz K, Filipovsky J, Imai Y, Wang J, Ibsen H, O'Brien E, Staessen JA, International Database on Ambulatory blood pressure in Relation to Cardiovascular Outcomes Investigators. Masked hypertension in diabetes mellitus: treatment implications for clinical practice. Hypertension 2013; 61: 964–971.
Gorostidi M, Sarafidis PA, de la Sierra A, Segura J, de la Cruz JJ, Banegas JR, Ruilope LM Spanish ABPM Registry Investigators. Differences between office and 24-hour blood pressure control in hypertensive patients with CKD: a 5693-patient cross-sectional analysis from Spain. Am J Kidney Dis 2013; 62: 285–294.
Agarwal R, Sinha AD, Light RP . Toward a definition of masked hypertension and white-coat hypertension among hemodialysis patients. Clin J Am Soc Nephrol 2011; 6: 2003–2008.
Myers MG . Evaluation of once-daily antihypertensive therapy. Heart Dis 2000; 2: S7–12.
Hermida RC, Ayala DE, Chayan L, Mojon A, Fernandez JR . Administration-time-dependent effects of olmesartan on the ambulatory blood pressure of essential hypertension patients. Chronobiol Int 2009; 26: 61–79.
Zannad F . Practical relevance of the 24-hour trough: peak ratio of antihypertensive drugs. J Hypertens 1995; 13: S109–S112.
Krapf R, Hulter HN . Arterial hypertension induced by erythropoietin and erythropoiesis-stimulating agents (ESA). Clin J Am Soc Nephrol 2009; 4: 470–480.
Pisoni RL, Bragg-Gresham JL, Young EW, Akizawa T, Asano Y, Locatelli F, Bommer J, Cruz JM, Kerr PG, Mendelssohn DC, Held PJ, Port FK . Anemia management and outcomes from 12 countries in the Dialysis Outcomes and Practice Patterns Study (DOPPS). Am J Kidney Dis 2004; 44: 94–111.
Abbott KC, Glanton CW, Trespalacios FC, Oliver DK, Ortiz MI, Agodoa LY, Cruess DF, Kimmel PL . Body mass index, dialysis modality, and survival: analysis of the United States Renal Data System Dialysis Morbidity and Mortality Wave II Study. Kidney Int 2004; 65: 597–605.
Inrig JK, Patel UD, Gillespie BS, Hasselblad V, Himmelfarb J, Reddan D, Lindsay RM, Winchester JF, Stivelman J, Toto R, Szczech LA . Relationship between interdialytic weight gain and blood pressure among prevalent hemodialysis patients. Am J Kidney Dis 2007; 50: 108–118 118 e101-104.
Verbeke F, Van Biesen W, Honkanen E, Wikstrom B, Jensen PB, Krzesinski JM, Rasmussen M, Vanholder R, Rensma PL, CORD Study Investigators. Prognostic value of aortic stiffness and calcification for cardiovascular events and mortality in dialysis patients: outcome of the calcification outcome in renal disease (CORD) study. Clin J Am Soc Nephrol 2011; 6: 153–159.
Avramovski P, Janakievska P, Sotiroski K, Zafirova-Ivanovska B, Sikole A . Aortic pulse wave velocity is a strong predictor of all—cause and cardiovascular mortality in chronic dialysis patients. Ren Fail 2014; 36: 176–186.
Acknowledgements
We are grateful to the patients who took part in this study. Our efforts would be meaningless without their contribution. We are grateful to Ms Li Changhong for performing the PWV measurements. This study was supported by a grant for clinical research from the Jiangsu Science and Technology Department to Dr Junwei Yang (Grant Number: BL2013037).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Hypertension Research website
Supplementary information
Rights and permissions
About this article
Cite this article
Liu, W., Wang, L., Sun, Z. et al. Masked uncontrolled hypertension in patients on maintenance hemodialysis. Hypertens Res 40, 819–824 (2017). https://doi.org/10.1038/hr.2017.48
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/hr.2017.48
Keywords
This article is cited by
-
Characteristics of visit-to-visit blood pressure variability in hemodialysis patients
Hypertension Research (2019)
-
Who removes the mask of hypertension?
Hypertension Research (2017)