Abstract
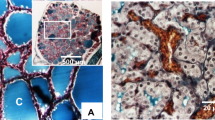
The purpose of this study was to evaluate whether the spontaneously hypertensive rat SHRSP.Z-Leprfa/IzmDmcr (SHRSP fatty) is a useful animal model to clarify molecular mechanisms that underlie metabolic syndrome. We investigated histopathologic changes in the cardiovascular organs and metabolic characteristics of SHRSP fatty rats, which are congenic rats from a cross between SHRSP and Zucker fatty (ZF) rats. The aortic wall and cardiac, carotid, and renal arteries from SHRSP and SHRSP fatty rats were thicker than those of ZF rats. The renal cortex in SHRSP and SHRSP fatty rats showed severe glomerulosclerosis. Pancreatic islands in SHRSP fatty and ZF rats showed marked hyperplasia. Steady-state plasma glucose concentrations were higher in SHRSP fatty than in ZF rats. Non-fasting triglyceride levels in SHRSP fatty rats were higher than in ZF rats. DNA synthesis in cultured vascular smooth muscle cells (VSMCs) from SHRSP fatty and SHRSP rats was significantly higher than that in VSMCs from Wistar-Kyoto (WKY) or ZF rats. Levels of platelet-derived growth factor A-chain and transforming growth factor-β1 mRNAs were higher in VSMCs from SHRSP fatty and SHRSP than from ZF rats. Microarray analysis identified five genes that were significantly upregulated and four genes that were significantly downregulated in visceral adipose tissue of SHRSP fatty rats compared with levels in control strains (SHRSP and ZF rats). These findings suggest that the combination of hypertension and obesity accelerates vascular remodeling, dyslipidemia, and insulin resistance in metabolic syndrome. The phenotype of SHRSP fatty is similar to that of human metabolic syndrome, and therefore, studies of these rats may help clarify the molecular mechanisms that underlie metabolic syndrome in humans.
Similar content being viewed by others
Article PDF
References
Landsberg L : Insulin resistance and hypertension. Clin Exp Hypertens 1999; 21: 885–894.
Rett K : The relation between insulin resistance and cardiovascular complications of the insulin resistance syndrome. Diabetes Obes Metab 1999; 1 ( Suppl 1): S8–S16.
Matsuzawa Y, Funahashi T, Nakamura T : Molecular mechanism of metabolic syndrome X: contribution of adipocytokines adipocyte-derived bioactive substances. Ann N Y Acad Sci 1999; 892: 146–154.
Eschwege E : The dysmetabolic syndrome, insulin resistance and increased cardiovascular (CV) morbidity and mortality in type 2 diabetes: aetiological factors in the development of CV complications. Diabetes Metab 2003; 29: 6 S19–6S27.
Schmieder RE, Hilgers KF, Schlaich MP, Schmidt BM : Renin-angiotensin system and cardiovascular risk. Lancet 2007; 369: 1208–1219.
Hiraoka-Yamamoto J, Nara Y, Yasui N, Onobayashi Y, Tsuchikura S, Ikeda K : Establishment of a new animal model of metabolic syndrome: SHRSP fatty (fa/fa) rats. Clin Exp Pharmacol Physiol 2004; 31: 107–109.
Raij L, Azar S, Keane W : Mesangial immune injury, hypertension, and progressive glomerular damage in Dahl rats. Kidney Int 1984; 26: 137–143.
Qian X, Jin L, Grande JP, Lloyd RV : Transforming growth factor-beta and p27 expression in pituitary cells. Endocrinology 1996; 137: 3051–3060.
Fukuda N, Kishioka H, Satoh C, et al: Role of long-form PDGF A-chain in the growth of vascular smooth muscle cells from spontaneously hypertensive rats. Am J Hypertens 1997; 10: 1117–1124.
el-Husseini AE, Paterson JA, Myal Y, Shiu RP : PCR detection of the rat brain basic fibroblast growth factor (bFGF) mRNA containing a unique 3′ untranslated region. Biochim Biophys Acta 1992; 1131: 314–316.
Hoof T, Riordan JR, Tummler B : Quantitation of mRNA by the kinetic polymerase chain reaction assay: a tool for monitoring P-glycoprotein gene expression. Anal Biochem 1991; 196: 161–169.
Baumbach GL, Walmsley JG, Hart MN : Composition and mechanics of cerebral arterioles in hypertensive rats. Am J Pathol 1988; 133: 464–471.
Ogata J, Fujishima M, Tamaki K, Nakatomi Y, Ishitsuka T, Omae T : Stroke-prone spontaneously hypertensive rats as an experimental model of malignant hypertension. I. A light- and electron-microscopic study of the brain. Acta Neuropathol (Berl) 1980; 51: 179–184.
Takaya K, Ogawa Y, Isse N, et al: Molecular cloning of rat leptin receptor isoform complementary DNAs—identification of a missense mutation in Zucker fatty (fa/fa) rats. Biochem Biophys Res Commun 1996; 225: 75–83.
Zucker TF, Zucker LM : Fat accretion and growth in the rat. Obes Res 1996; 4: 102–108.
Larsson LI, Boder GB, Shaw WN : Changes in the islets of langerhans in the obese Zucker rat. Lab Invest 1977; 36: 593–598.
Milburn JL Jr, Hirose H, Lee YH, et al: Pancreatic beta-cells in obesity. Evidence for induction of functional, morphologic, and metabolic abnormalities by increased long chain fatty acids. J Biol Chem 1995; 270: 1295–1299.
Folli F, Kahn CR, Hansen H, Bouchie JL, Feener EP : Angiotensin II inhibits insulin signaling in aortic smooth muscle cells at multiple levels. A potential role for serine phosphorylation in insulin/angiotensin II crosstalk. J Clin Invest 1997; 100: 2158–2169.
Velloso LA, Folli F, Sun XJ, White MF, Saad MJ, Kahn CR : Cross-talk between the insulin and angiotensin signaling systems. Proc Natl Acad Sci U S A 1996; 93: 12490–12495.
Fukuda N, Hu WY, Teng J, et al: Troglitazone inhibits growth and improves insulin signaling by suppression of angiotensin II action in vascular smooth muscle cells from spontaneously hypertensive rats. Atherosclerosis 2002; 163: 229–239.
Fukuda N, Satoh C, Hu WY, Nakayama M, Kishioka H, Kanmatsuse K : Endogenous angiotensin II suppresses insulin signaling in vascular smooth muscle cells from spontaneously hypertensive rats. J Hypertens 2001; 19: 1651–1658.
Higashiura K, Ura N, Miyazaki Y, Shimamoto K : Effect of an angiotensin II receptor antagonist, candesartan, on insulin resistance and pressor mechanisms in essential hypertension. J Hum Hypertens 1999; 13 ( Suppl 1): S71–S74.
Shimamoto K, Ura N, Nakagawa M, et al: The mechanisms of the improvement of insulin sensitivity by angiotensin converting enzyme inhibitor. Clin Exp Hypertens 1996; 18: 257–266.
Bahadir O, Aydin S, Caylan R : The effect on the middle-ear cavity of an absorbable gelatine sponge alone and with corticosteroids. Eur Arch Otorhinolaryngol 2003; 260: 19–23.
Yamaguchi K, Ura N, Murakami H, et al: Olmesartan ameliorates insulin sensitivity by modulating tumor necrosis factor-alpha and cyclic AMP in skeletal muscle. Hypertens Res 2005; 28: 773–778.
Fukuda N, Nakayama M, Jian T, et al: Leukocyte angiotensin II levels inpatients with essential hypertension: relation to insulin resistance. Am J Hypertens 2003; 16: 129–134.
Cai H, Li Z, Dikalov S, et al: NAD(P)H oxidase−derived hydrogen peroxide mediates endothelial nitric oxide production in response to angiotensin II. J Biol Chem 2002; 277: 48311–48317.
Griendling KK, Minieri CA, Ollerenshaw JD, Alexander RW : Angiotensin II stimulates NADH and NADPH oxidase activity in cultured vascular smooth muscle cells. Circ Res 1994; 74: 1141–1148.
Moriki N, Ito M, Seko T, et al: RhoA activation in vascular smooth muscle cells from stroke-prone spontaneously hypertensive rats. Hypertens Res 2004; 27: 263–270.
Negishi H, Ikeda K, Sagara M, Sawamura M, Yamori Y : Increased oxidative DNA damage in stroke-prone spontaneously hypertensive rats. Clin Exp Pharmacol Physiol 1999; 26: 482–484.
Mark AL, Shaffer RA, Correia ML, Morgan DA, Sigmund CD, Haynes WG : Contrasting blood pressure effects of obesity in leptin-deficient ob/ob mice and agouti yellow obese mice. J Hypertens 1999; 17: 1949–1953.
Verma S, Leung YM, Yao L, Battell M, Dumont AS, McNeill JH : Hyperinsulinemia superimposed on insulin resistance does not elevate blood pressure. Am J Hypertens 2001; 14: 429–432.
Fukuda N, Hu WY, Satoh C, et al: Contribution of synthetic phenotype on the enhanced angiotensin II−generating system in vascular smooth muscle cells from spontaneously hypertensive rats. J Hypertens 1999; 17: 1099–1107.
Fukuda N, Satoh C, Hu WY, et al: Production of angiotensin II by homogeneous cultures of vascular smooth muscle cells from spontaneously hypertensive rats. Arterioscler Thromb Vasc Biol 1999; 19: 1210–1217.
Hu WY, Fukuda N, Kanmatsuse K : Growth characteristics, angiotensin II generation, and microarray-determined gene expression in vascular smooth muscle cells from young spontaneously hypertensive rats. J Hypertens 2002; 20: 1323–1333.
Hu WY, Fukuda N, Satoh C, et al: Phenotypic modulation by fibronectin enhances the angiotensin II−generating system in cultured vascular smooth muscle cells. Arterioscler Thromb Vasc Biol 2000; 20: 1500–1505.
Satoh C, Fukuda N, Hu WY, Nakayama M, Kishioka H, Kanmatsuse K : Role of endogenous angiotensin II in the increased expression of growth factors in vascular smooth muscle cells from spontaneously hypertensive rats. J Cardiovasc Pharmacol 2001; 37: 108–118.
Rodriguez S, Gaunt TR, Day IN : Molecular genetics of human growth hormone, insulin-like growth factors and their pathways in common disease. Hum Genet 2007; 122: 1–21.
Beattie JH, Wood AM, Newman AM, et al: Obesity and hyperleptinemia in metallothionein (-I and -II) null mice. Proc Natl Acad Sci U S A 1998; 95: 358–363.
Hurowitz EH, Melnyk JM, Chen YJ, Kouros-Mehr H, Simon MI, Shizuya H : Genomic characterization of the human heterotrimeric G protein alpha, beta, and gamma subunit genes. DNA Res 2000; 7: 111–120.
Calabrese V, Boyd-Kimball D, Scapagnini G, Butterfield DA : Nitric oxide and cellular stress response in brain aging and neurodegenerative disorders: the role of vitagenes. In Vivo (Athens, Greece) 2004; 18: 245–267.
Katzmarzyk PT, Rankinen T, Perusse L, et al: Linkage and association of the sodium potassium-adenosine triphosphatase alpha2 and beta1 genes with respiratory quotient and resting metabolic rate in the Quebec Family Study. J Clin Endocrinol Metab 1999; 84: 2093–2097.
Adachi T, Ohta H, Yamada H, Futenma A, Kato K, Hirano K : Quantitative analysis of extracellular-superoxide dismutase in serum and urine by ELISA with monoclonal antibody. Clin Chim Acta 1992; 212: 89–102.
Nielsen J, Christiansen J, Lykke-Andersen J, Johnsen AH, Wewer UM, Nielsen FC : A family of insulin-like growth factor II mRNA-binding proteins represses translation in late development. Mol Cell Biol 1999; 19: 1262–1270.
Blachly-Dyson E, Zambronicz EB, Yu WH, et al: Cloning and functional expression in yeast of two human isoforms of the outer mitochondrial membrane channel, the voltage-dependent anion channel. J Biol Chem 1993; 268: 1835–1841.
Huq AH, Lovell RS, Ou CN, Beaudet AL, Craigen WJ : X-linked glycerol kinase deficiency in the mouse leads to growth retardation, altered fat metabolism, autonomous glucocorticoid secretion and neonatal death. Hum Mol Genet 1997; 6: 1803–1809.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ueno, T., Takagi, H., Fukuda, N. et al. Cardiovascular Remodeling and Metabolic Abnormalities in SHRSP.Z-Leprfa/IzmDmcr Rats as a New Model of Metabolic Syndrome. Hypertens Res 31, 1021–1031 (2008). https://doi.org/10.1291/hypres.31.1021
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.31.1021
Keywords
This article is cited by
-
Angiotensin II Type 1 Receptor Antagonist Azilsartan Restores Vascular Reactivity Through a Perivascular Adipose Tissue-Independent Mechanism in Rats with Metabolic Syndrome
Cardiovascular Drugs and Therapy (2019)
-
Renoprotective mechanisms of telmisartan on renal injury and inflammation in SHRSP.Z-Leprfa/IzmDmcr rats
Clinical and Experimental Nephrology (2013)
-
Guava leaf extracts alleviate fatty liver via expression of adiponectin receptors in SHRSP.Z-Leprfa/Izm rats
Nutrition & Metabolism (2012)