Abstract
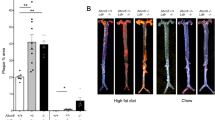
This study was designed to determine whether Bach1 gene ablation leads to suppression of atherosclerosis in apolipoprotein E (Apo E)/Bach1 double knockout (DKO) mice. Apo E/Bach1 DKO mice were generated by intercrossing Apo E knockout (KO) and Bach1 KO mice. The animals were fed a high-fat diet for 8 weeks, and the atherosclerotic plaques in the thoracic and abdominal aorta were visualized by oil red O staining. In DKO mice, the total plaque area was reduced by 32% compared with that in Apo E KO mice. In DKO mice, heme oxygenase-1 (HO-1) was upregulated in the endothelium and, to a lesser extent, in vascular smooth muscles. In atherosclerotic plaques in Apo E KO mice and DKO mice, HO-1 was abundantly expressed in the endothelium and macrophages. Urine excretion of 8-iso-prostaglandin (PG) F2α, a marker for lipid peroxidation, was reduced in DKO mice compared with that in Apo E KO mice. The effects of Bach1 ablation on the plaque area and 8-iso-PG F2α excretion were almost completely abolished by treating DKO mice with Sn protoporphyrin, an inhibitor of HO activity. Disruption of the Bach1 gene in Apo E KO mice caused inhibition of atherosclerosis through upregulation of HO-1. Inhibition of Bach1, conversely, may be a novel therapeutic strategy to treat atherosclerotic diseases.
Similar content being viewed by others
Article PDF
References
Coburn RF, Williams WJ, White P, Kahn SB : The production of carbon monoxide from hemoglobin in vivo. J Clin Invest 1967; 46: 346–356.
Yano Y, Ozono R, Oishi Y, et al: Genetic ablation of the transcription repressor Bach1 leads to myocardial protection against ischemia/reperfusion in mice. Genes Cells 2006; 11: 791–803.
Sun J, Hoshino H, Takaku K, et al: Hemoprotein Bach1 regulates enhancer availability of heme oxygenase-1 gene. EMBO J 2002; 21: 5216–5224.
Suzuki H, Tashiro S, Hira S, et al: Heme regulates gene expression by triggering Crm1-dependent nuclear export of Bach1. EMBO J 2004; 23: 2544–2553.
Saito K, Ishizaka N, Aizawa T, et al: Role of aberrant iron homeostasis in the upregulation of transforming growth factor-beta1 in the kidney of angiotensin II–induced hypertensive rats. Hypertens Res 2004; 27: 599–607.
Ozono R : New biotechnological methods to reduce oxidative stress in the cardiovascular system: focusing on the Bach1/heme oxygenase-1 pathway. Curr Pharm Biotechnol 2006; 7: 87–93.
Morita T, Mitsialis SA, Koike H, Liu Y, Kourembanas S : Carbon monoxide controls the proliferation of hypoxic vascular smooth muscle cells. J Biol Chem 1997; 272: 32804–32809.
Otterbein LE, Bach FH, Alam J, et al: Carbon monoxide has anti-inflammatory effects involving the mitogen-activated protein kinase pathway. Nat Med 2000; 6: 422–428.
Stocker R, Yamamoto Y, McDonagh AF, Glazer AN, Ames BN : Bilirubin is an antioxidant of possible physiological importance. Science 1987; 235: 1043–1046.
Poss KD, Tonegawa S : Reduced stress defense in heme oxygenase 1–deficient cells. Proc Natl Acad Sci U S A 1997; 94: 10925–10930.
Yet SF, Layne MD, Liu X, et al: Absence of heme oxygenase-1 exacerbates atherosclerotic lesion formation and vascular remodeling. FASEB J 2003; 17: 1759–1761.
Yachie A, Niida Y, Wada T, et al: Oxidative stress causes enhanced endothelial cell injury in human heme oxygenase-1 deficiency. J Clin Invest 1999; 103: 129–135.
Juan SH, Lee TS, Tseng KW, et al: Adenovirus-mediated heme oxygenase-1 gene transfer inhibits the development of atherosclerosis in apolipoprotein E–deficient mice. Circulation 2001; 104: 1519–1525.
Tulis DA, Durante W, Liu X, Evans AJ, Peyton KJ, Schafer AI : Adenovirus-mediated heme oxygenase-1 gene delivery inhibits injury-induced vascular neointima formation. Circulation 2001; 104: 2710–2715.
Brydun A, Watari Y, Yamamoto Y, et al: Reduced expression of heme oxygenase-1 in patients with coronary atherosclerosis. Hypertens Res 2007; 30: 341–348.
Itoh K, Chiba T, Takahashi S, et al: An Nrf2/small Maf heterodimer mediates the induction of phase II detoxifying enzyme genes through antioxidant response elements. Biochem Biophys Res Commun 1997; 236: 31–22.
Alam J, Stewart D, Touchard C, Boinapally S, Choi AM, Cook JL : Nrf2, a Cap’n’Collar transcription factor, regulates induction of the heme oxygenase-1 gene. J Biol Chem 1999; 274: 26071–26078.
Ishii T, Itoh K, Takahashi S, et al: Transcription factor Nrf2 coordinately regulates a group of oxidative stress–inducible genes in macrophages. J Biol Chem 2000; 275: 16023–16029.
Kataoka K, Handa H, Nishizawa M : Induction of cellular antioxidative stress genes through heterodimeric transcription factor Nrf2/small Maf by antirheumatic gold(I) compounds. J Biol Chem 2001; 276: 34074–34081.
Oyake T, Itoh K, Motohashi H, et al: Bach proteins belong to a novel family of BTB-basic leucine zipper transcription factors that interact with MafK and regulate transcription through the NF-E2 site. Mol Cell Biol 1996; 16: 6083–6095.
Sun J, Brand M, Zenke Y, Tashiro S, Groudine M, Igarashi K : Heme regulates the dynamic exchange of Bach1 and NF-E2–related factors in the Maf transcription factor network. Proc Natl Acad Sci U S A 2004; 101: 1461–1466.
Omura S, Suzuki H, Toyofuku M, Ozono R, Kohno N, Igarashi K : Effects of genetic ablation of bach1 upon smooth muscle cell proliferation and atherosclerosis after cuff injury. Genes Cells 2005; 10: 277–285.
Ogawa K, Sun J, Taketani S, et al: Heme mediates derepression of Maf recognition element through direct binding to transcription repressor Bach1. EMBO J 2001; 20: 2835–2843.
Kotite L, Bergeron N, Havel RJ : Quantification of apolipoproteins B-100, B-48, and E in human triglyceride-rich lipoproteins. J Lipid Res 1995; 36: 890–900.
Veillard NR, Steffens S, Burger F, Pelli G, Mach F : Differential expression patterns of proinflammatory and antiinflammatory mediators during atherogenesis in mice. Arterioscler Thromb Vasc Biol 2004; 24: 2339–2344.
Yang H, Roberts LJ, Shi MJ, et al: Retardation of atherosclerosis by overexpression of catalase or both Cu/Zn-superoxide dismutase and catalase in mice lacking apolipoprotein E. Circ Res 2004; 95: 1075–1081.
Lindenblatt N, Bordel R, Schareck W, Menger MD, Vollmar B : Vascular heme oxygenase-1 induction suppresses microvascular thrombus formation in vivo. Arterioscler Thromb Vasc Biol 2004; 24: 601–606.
Imai T, Morita T, Shindo T, et al: Vascular smooth muscle cell-directed overexpression of heme oxygenase-1 elevates blood pressure through attenuation of nitric oxide–induced vasodilation in mice. Circ Res 2001; 89: 55–62.
Shimosawa T, Ogihara T, Matsui H, Asano T, Ando K, Fujita T : Deficiency of adrenomedullin induces insulin resistance by increasing oxidative stress. Hypertension 2003; 41: 1080–1085.
Takaya T, Kawashima S, Shinohara M, et al: Angiotensin II type 1 receptor blocker telmisartan suppresses superoxide production and reduces atherosclerotic lesion formation in apolipoprotein E–deficient mice. Atherosclerosis 2006; 186: 402–410.
Tomoyori H, Kawata Y, Higuchi T, et al: Phytosterol oxidation products are absorbed in the intestinal lymphatics in rats but do not accelerate atherosclerosis in apolipoprotein E–deficient mice. J Nutr 2004; 134: 1690–1696.
Pratico D, Tangirala RK, Rader DJ, Rokach J, FitzGerald GA : Vitamin E suppresses isoprostane generation in vivo and reduces atherosclerosis in Apo E–deficient mice. Nat Med 1998; 4: 1189–1192.
Warnholtz A, Nickenig G, Schulz E, et al: NADH-oxidase–mediated superoxide production in the early stages of atherosclerosis: evidence for involvement of the renin-angiotensin system. Circulation 1999; 99: 2027–2033.
Shinomiya K, Fukunaga M, Kiyomoto H, et al: A role of oxidative stress–generated eicosanoid in the progression of arteriosclerosis in type 2 diabetes mellitus model rats. Hypertens Res 2002; 25: 91–98.
Ross R : Atherosclerosis—an inflammatory disease. N Engl J Med 1999; 340: 115–126.
Ohara Y, Peterson TE, Sayegh HS, Subramanian RR, Wilcox JN, Harrison DG : Dietary correction of hypercholesterolemia in the rabbit normalizes endothelial superoxide anion production. Circulation 1995; 92: 898–903.
Hu Q, Yu Z-X, Ferrans VJ, Takeda K, Irani K, Ziegelstein RC : Critical role of NADPH oxidase–derived reactive oxygen species in generating Ca2+ oscillations in human aortic endothelial cells stimulated by histamine. J Biol Chem 2002; 277: 32546–32551.
Kawamura K, Ishikawa K, Wada Y, et al: Bilirubin from heme oxygenase-1 attenuates vascular endothelial activation and dysfunction. Arterioscler Thromb Vasc Biol 2005; 25: 155–160.
Balla G, Jacob HS, Balla J, et al: Ferritin: a cytoprotective antioxidant strategem of endothelium. J Biol Chem 1992; 267: 18148–18153.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Watari, Y., Yamamoto, Y., Brydun, A. et al. Ablation of the Bach1 Gene Leads to the Suppression of Atherosclerosis in Bach1 and Apolipoprotein E Double Knockout Mice. Hypertens Res 31, 783–792 (2008). https://doi.org/10.1291/hypres.31.783
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1291/hypres.31.783
Keywords
This article is cited by
-
Regulatory mechanisms of heme regulatory protein BACH1: a potential therapeutic target for cancer
Medical Oncology (2021)
-
BACH transcription factors in innate and adaptive immunity
Nature Reviews Immunology (2017)
-
The Bach Family of Transcription Factors: A Comprehensive Review
Clinical Reviews in Allergy & Immunology (2016)