Abstract
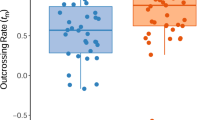
Evolutionary theory predicts a negative correlation between the rate of self-fertilization and the magnitude of inbreeding depression in natural plant populations. We examined the mating system and inbreeding depression at four life stages in a diploid population of Epilobium angustifolium (Onagraceae). The multilocus rate of self-fertilization, inferred from the segregation of allozymes at two polymorphic loci (Pgi-2, Mdh-2) among seedlings from 22 open-pollinated plants, was 0.06 (SE±0.10). However, after adjusting for differential mortality of selfed progeny prior to the seedling stage, the selfing rate was 0.45. The parental inbreeding coefficient (F) was -0.26 (SE ± 0.103) indicating more heterozygotes than in a randomly mating population. The magnitude of inbreeding depression at seed maturation, seed germination, juvenile survival and dry mass at 8 weeks was measured in a greenhouse experiment. Thirty-four plants, grown from open-pollinated seed, were each self-fertilized and randomly outcrossed. We observed significant inbreeding depression at all four stages, although its magnitude differed significantly among maternal plants and among stages. Inbreeding depression was largest during seed maturation (0.87), where seed-set in outcrossed flowers (65.7 per cent) was eight times higher than in selfed flowers (8.3 per cent). Inbreeding depression for dry mass after 8 weeks (0.50) was at least twice that for germination (0.25) and survival (0.15). The magnitude of inbreeding depression for each of the 34 maternal parents was not significantly correlated across life stages, suggesting that its genetic basis varies across the life cycle. Our results indicate that E. angustifolium exhibits high levels of inbreeding depression, much of which is expressed early in the life cycle, during seed maturation, consistent with results for other long-lived, outcrossing species. However, given its moderate rate of self-fertilization, the total inbreeding depression (0.95) maintained in E. angustifolium is very high in comparison to other angiosperms.
Similar content being viewed by others
Article PDF
References
Ågren, J, and Schemske, D W. 1993. Outcrossing rate and inbreeding depression in two annual monoecious herbs, Begonia hirsuta and B. semiovata. Evolution, 47, 125–135.
Ashman, T-L. 1992. The relative importance of inbreeding and maternal sex in determining progeny fitness in Sidalcea oregana ssp. spicata, a gynodioecious plant. Evolution, 46, 1862–1874.
Bertin, R I, Barnes, C, and Guttman, S I. 1989. Self-sterility and cryptic self-fertility in Campsis radicans (Bignoniaceae). Bot Gaz, 150, 397–403.
Bishir, J, and Namkoong, G. 1987. Unsound seeds in conifers: estimation of numbers of lethal alleles and of magnitudes of effects associated with the maternal parent. Silvae Genet, 36, 180–185.
Brown, A H D, and Allard, R W. 1970. Estimation of the mating system in open-pollinated maize populations using isozyme polymorphisms. Genetics, 66, 133–145.
Broyles, S B, and Wyatt, R. 1993. The consequences of self-pollination in Asclepias exaltata, a self-incompatible milkweed. Am J Bot, 80, 41–44.
Charlesworth, D, and Charlesworth, B. 1979. The evolutionary genetics of sexual systems in flowering plants. Proc R Soc B, 205, 513–530.
Charlesworth, D, and Charlesworth, B. 1987. Inbreeding depression and its evolutionary consequences. Ann Rev Ecol Syst, 18, 237–268.
Charlesworth, D, Morgan, M T, and Charlesworth, B. 1990. Inbreeding depression, genetic load, and the evolution of outcrossing rates in a multilocus system with no linkage. Evolution, 44, 1469–1489.
Darwin, C R. 1876. The Effects of Cross and Self-fertilization in the Vegetable Kingdom. John Murray, London.
Dudash, M R. 1990. Relative fitness of selfed and outcrossed progeny in a self-compatible protandrous species, Sabatia angularis L. (Gentianaceae): a comparison of three environments. Evolution, 44, 1129–1139.
Flint, E P. 1980. Ecology and Distribution of Diploid and Tetraploid Epilobium angustifolium (Fireweed) in the Beartooth Mountains of Wyoming and Montana. Ph.D. Thesis, Duke University.
Fowler, D P, and Park, Y S. 1983. Population studies of white spruce. I. Effects of self-pollination. Can J Forest Res, 13, 1133–1138.
Fowler, D P. 1965b. Effects of inbreeding in red pine, Pinus resinosa. Ait. IV. Comparison with other northeastern Pinus species. Silvae Genet, 14, 76–81.
Fowler, D P, and Park, Y S. 1983. Population studies of white spruce. I. Effects of self-pollination. Can J Forest Res, 13, 1133–1138.
Franklin, E C. 1969. Inbreeding depression in metrical traits of loblolly pine (Pinus taeda L.) as a result of self-pollination. Technical Rep. NO. 40, School of Forest Resources, N. Carolina State Univ. Raleigh, 19 pp.
Galen, C, and Plowright, R C. 1988. Contrasting movement patterns of nectar-collecting and pollen-collecting bumble bees (Bombus terricola) on fireweed (Chamaenerion angustifolium) inflorescences. Ecol Entomol, 10, 9–17.
Haldane, J B S. 1949. The association of characters as a result of inbreeding and linkage. Ann Eugen, 15, 23.
Holtsford, T P, and Ellstrand, N C. 1990. Inbreeding effects in Clarkia tembloriensis (Onagraceae) populations with different natural outcrossing rates. Evolution, 44, 2031–2046.
Husband, B C, and Schemske, D W. 1995. Evolution of the magnitude and timing of inbreeding depression in plants. Evolution, in press.
Johnston, M O. 1992. Effects of cross and self-fertilization and progeny fitness in Lobelia cardinalis and L. syphilitica. Evolution, 46, 688–702.
Johnston, M O, and Schoen, D J. 1994. On the measurement of inbreeding depression. Evolution, in press.
Karoly, K. 1991. The Causes and Consequences of Intra-populational Mating System Variation in Lupinus nanus (Leguminosae). Ph.D. Thesis, University of Chicago.
Kenrick, J, Kaul, V, and Williams, E G. 1986. Self-incompatibility in Acacia retinodes: site of pollen-tube arrest is the nucellus. Planta, 169, 245–250.
Kimura, M. 1958. Zygotic frequencies in a partially self-fertilizing population. Ann Report Nat Instil Genetics, Japan, 8, 104–105.
Krebs, S L, and Hancock, J F. 1991. Embryonic genetic load in the highbush blueberry Vaccinium corymbosum (Ericaceae). Am J Bot, 78, 1427–1437.
Lande, R, and Schemske, D W. 1985. The evolution of self-fertilization and inbreeding depression in plants. I. Genetic models. Evolution, 39, 24–40.
Lande, R, Schemske, D W, and Schultz, S T. 1994. High inbreeding depression, selective interference among loci, and the threshold selfing rate for purging recessive lethal mutations. Evolution, 48, 965–978.
Lloyd, D G. 1979. Some reproductive factors affecting the selection of self-fertilization in plants. Am Nat, 113, 67–79.
Lynch, M. 1988. Design and analysis of experiments on random drift and inbreeding depression. Genetics, 120, 791–807.
Maki, M. 1993. Outcrossing and fecundity advantage of females in gynodioecious Chionographis japonica var. kurohimensis (Liliaceae). Am J Bot, 80, 629–634.
Meinke, D W. 1991. Perspective on genetic analysis of plant embryogenesis. PI Cell, 3, 857–866.
Miller, R J. Jr. 1986. Beyond ANOVA, Basics of Applied Statistics. John Wiley and Sons, New York.
Montalvo, A M. 1992. Relative success of self and outcross pollen comparing mixed- and single-donor pollinations in Aquilegia caerulea. Evolution, 46, 1181–1198.
Mosquin, T. 1967. Evidence for autopolyploidy in Epilobium angustifolium (Onagraceae). Evolution, 21, 713–719.
Park, Y S, and Fowler, D P. 1984. Inbreeding in black spruce (Picea mariana (Mill.) B.S.P.): self-fertility, genetic load, and performance. Can J Forest Res, 14, 17–21.
Parker, I M, Nakamura, R R, and Schemske, D W. 1995. Reproductive allocation and the fitness consequences of selfing in two sympatric species of Epilobium (Onagraceae) with contrasting mating systems. Am J Bot, in press.
Raven, P H. 1979. A survey of reproductive biology in Onagraceae. N Z J Bot, 17, 575–593.
Raven, P H. 1988. Onagraceae as a model of plant evolution. In: Gottlieb, L. D. and Jain, S. K. (eds) Plant Evolutionary Biology, pp. 85–107. Chapman & Hall, New York.
Ritland, K. 1989. A series of FORTRAN computer programs for estimating plant mating systems. J Hered, 81, 235–237.
Ritland, K, and Jain, S K. 1981. A model for the estimation of outcrossing rate and gene frequencies using n independent loci. Heredity, 47, 35–52.
Roach, D A, and Wulff, R D. 1987. Maternal effects in plants. Ann Rev Ecol Syst, 18, 209–235.
Schemske, D W. 1983. Breeding system and habitat effects on fitness components in three neotropical Costus (Zingiber-aceae). Evolution, 37, 523–539.
Schoen, D J. 1983. Relative fitnesses of selfed and outcrossed progeny in Gilia achilleifolia (Polemoniaceae). Evolution, 37, 292–301.
Schoen, D J, and Clegg, M T. 1984. Estimation of mating parameters when outcrossing events are correlated. Proc Natl Acad Sci USA, 81, 5258–5262.
Scribailo, R W, and Barrett, S C H. 1991. Pollen-pistil interactions in tristylous Pontederia sagittata (Pontederiaceae). II. Patterns of pollen tube growth. Am J Bot, 78, 1662–1682.
Seavey, S R, and Bawa, K S. 1986. Late-acting self-incompatibility in angiosperms. Bot Rev, 52, 195–219.
Seavey, S R, and Carter, S K. 1994. Self-sterility in Epilobium obcordatum (Onagraceae). Am J Bot, 81, 331–338.
Snyder, E B. 1968. Seed yield and nursery performance of self-pollinated pines. Forest Sci, 14, 68–74.
Sokal, R R, and Rohlf, F J. 1981. Biometry, 2nd edn. Freeman and CO., New York.
Soltis, D E, Haufler, C H, Darrow, D C, and Gastony, G J. 1983. Starch gel electrophoresis of ferns: a compilation of grinding buffers, gel and electrode buffers and staining schedules. Am Fern J, 73, 9–27.
Sorensen, F C, and Miles, R S. 1974. Self-pollination effects on Douglas-fir and ponderosa pine seeds and seedlings. Sil vae Genet, 23, 135–138.
Stevens, J P, and Bougourd, S M. 1988. Inbreeding depression and the outcrossing rate in natural populations of Allium schoenoprasum L. Heredity, 60, 257–261.
Toppings, P. 1989. The Significance of Inbreeding Depression to the Evolution of Self-fertilization in Eichhornia paniculata (Spreng.) Solms. (Pontederiaceae) M.Sc. Thesis, University of Toronto, Canada.
Wiens, D, Calvin, C L, Wilson, C A, Davern, C I, Frank, D, and Seavey, S R. 1987. Reproductive success, spontaneous embryo abortion and genetic load in flowering plants. Oecologia, 71, 501–509.
Wolfe, L M. 1993. Inbreeding depression in Hydrophyllum appendiculatum: role of maternal effects, crowding and parental mating history. Evolution, 47, 374–386.
Acknowledgements
We thank S. Curtis, J. Ross, M. Conrath and D. Ewing for technical assistance. This study was supported by a Royalty Research Fund grant to D.S. and a Postdoctoral Fellowship from the Natural Sciences and Engineering Research Council of Canada to B.H.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Husband, B., Schemske, D. Magnitude and timing of inbreeding depression in a diploid population of Epilobium angustifolium (Onagraceae). Heredity 75, 206–215 (1995). https://doi.org/10.1038/hdy.1995.125
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1995.125
Keywords
This article is cited by
-
A genetic view on the role of prolonged drought stress and mating systems on post-drought recovery, persistence and drought memory of orchardgrass (Dactylis glomerata L.)
Euphytica (2020)
-
In vitro propagation of Epipactis flava Seidenf., an endangered rheophytic orchid: a first study on factors affecting asymbiotic seed germination, seedling development and greenhouse acclimatization
Plant Cell, Tissue and Organ Culture (PCTOC) (2018)
-
Different levels of inbreeding depression between outcrossing and selfing Serapias species
Biologia plantarum (2009)
-
Effects of population size on performance and inbreeding depression in Lupinus perennis
Oecologia (2008)
-
Sexual interference within flowers of Chamerion angustifolium
Evolutionary Ecology (2006)