Abstract
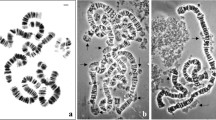
Allium sphaerocephalon is a species with a high number of secondary constrictions and with intra- and interindividual variation in their number. We analysed whether all the secondary constrictions are involved in nucleolus formation, whether the same variability exists in the number of NOR, and the mechanism(s) responsible for this variability. Our cytological data have shown that (i) all the secondary constrictions may be involved in the nucleolus organization, (ii) the variability in number is in accordance with the numbers of NOR, and (iii) the variability could be due to unequal exchanges among ribosomal genes on homologous and non-homologous chromosomes. Ribosomal DNA sequence homogeneity detected in this species supports this conclusion.
Similar content being viewed by others
Article PDF
References
Appels, R, Gerlach, W L, Dennis, E S, Swift, H, and Peacock, W J. 1980. Molecular and chromosomal organization of DNA sequences coding for the ribosomal RNAs in cereals. Chromosoma, 78, 293–311.
Arheim, N, Krystal, M, Schmickel, R, Wilson, G, Ryder, O, and Zimmer, E. 1980. Molecular evidence for genetic exchange among ribosomal genes on non-homologous chromosomes in man and apes. Proc Natl Acad Sci, USA, 77, 7323–7527.
Bougourd, S M, and Parker, J S. 1976. Nucleolar-organiser polymorphism in natural populations of Allium schoenoprasum. Chromosoma, 56, 301–307.
Carmona, M J, De Domenicis, R I, Salvi, G, and Maggini, F. 1984. Ribosomal RNA genes in biotypes of Scilla peruviana (Liliaceae). Pl Syst Evol, 146, 1–11.
Dellaporta, S L, Wood, J, and Hicks, J B. 1985. Maize DNA miniprep. In: Malmberg, R. Messing, J. and Sussex, I. (eds) Molecular Biology of Plants A Laboratory Course Manual. Cold Spring Harbor Laboratory Press, New York.
Delseny, M, Cooke, and Penon, P. 1983. Sequence heterogeneity in radish nuclear ribosomal RNA genes. Plant Sci Lett, 30, 107–119.
Ferraro, M, and Prantera, G. 1988. Human NORs show correlation between transcriptional activity, DNase I sensitivity, and hypomethylation. Cytogenet Cell Genet, 47, 58–61.
Gerlach, W L, and Bedbrook, J R. 1979. Cloning and characterization of ribosomal RNA genes from wheat and barley. Nucl Acids Res, 7, 1869–1885.
Givens, J F, and Phillips, R L. 1976. The nucleolus organizer regions of maize (Zea mays. L.) ribosomal RNA gene distribution and nucleolar interactions. Chromosoma, 57, 103–117.
Guillen, A, and Ruiz Rejon, M. 1984. The B-chromosome system of Allium sphaerocephalon L. (Liliaceae): Types, effects and origin. Caryologia, 37 (3), 259–267.
Iamilena, M, Ruiz Rejon, C, and Ruiz Rejon, M. 1990. Variation in the heterochromatin and nucleolar organizing regions of Allium subvillosum L. (Liliaceae). Genome, 33, 779–784.
Kodama, Y, Yoshida, M C, and Sasaki, M. 1980. An improved silver staining technique for nucleolus organizer regions by using a nylon cloth. Jpn J Hum Genet, 25, 229–233.
Loidl, J. 1988. SC-formation in some Allium species, and a discussion of the significance of SC-associated structures and of the mechanism for presynaptic alignment. Pl Syst Evol, 158, 117–131.
Loidl, J, and Greilhuber, J. 1983. Structural changes of Ag-stained nucleolus organizing regions and nucleoli during meiosis in Allium flavum. Can J Genet Cytol, 25, 524–529.
Maggini, F. 1975. Homologies of ribosomal RNA nucleotide sequences in monocots. J Mol Evol, 8, 317–322.
Maggini, F, and Carmona, M J. 1981. Sequence heterogeneity of the ribosomal DNA in Allium cepa (Liliaceae). Protoplasma, 108, 163–171.
Nicoloff, H, Anastassova-Kristeva, M, Rieger, R, and Künzel, G. 1979. ‘Nucleolar dominance’ as observed in barley translocation lines with specifically reconstructed SAT chromosomes. Theor Appl Genet, 55, 247–251.
Pastor, J. 1982. Karyology of Allium species from the Iberian Peninsula. Phyton, 22 (2), 171–200.
Phillips, R B, Pleyte, K A, and Hartley, S E. 1988. Stock-specific differences in the number and chromosome positions of the nucleolar organizer regions in arctic char (Salvelinus alpinus). Cytogenet Cell Genet, 48, 9–12.
Rayburn, A L, and Gill, B S. 1985. Use of biotin-labelled probes to map specific DNA sequences on wheat chromosomes. J Hered, 76, 78–81.
Sambrook, J, Fritsch, E F, and Maniatis, T. 1989. Molecular Cloning A Laboratory Manual. Cold Spring Harbor Laboratory Press, New York.
Sato, S. 1981. Cytological studies on satellited chromosomes of Allium cepa. Caryologia, 34 (4), 431–440.
Schubert, I, and Wobus, U. 1985. In situ hybridization confirms jumping nucleolus organizing regions in Allium. Chromosoma, 92, 143–148.
Suzuki, H, Kurihara, Y, Kanehisa, T, and Moriwaki, K. 1990. Variation in the distribution of silver-staining nucleolar organizer regions on the chromosomes of the wild mouse, Musmusculus. Mol Biol Evol, 7 (3), 271–282.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Garrido-Ramos, M., Jamilena, M., Lozano, R. et al. A cytogenetical and molecular analysis of the ribosomal cistrons of Allium sphaerocephalon L. (Liliaceae). Heredity 69, 43–49 (1992). https://doi.org/10.1038/hdy.1992.92
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1992.92
Keywords
This article is cited by
-
Cytogenetical evidences for hybrid structure and origin of diploid and triploid shallots (Allium cepa var.viviparum, Liliaceae) from Dalmatia (Croatia)
Plant Systematics and Evolution (1996)
-
rDNA site number polymorphism and NOR inactivation in natural populations ofAllium schoenoprasum
Genetica (1994)