Abstract
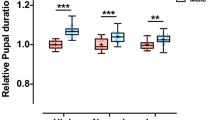
The developmental time of period mutants in Drosophila melanogaster was monitored under different environmental conditions. We observed that the pers mutants, which have short 19 h circadian cycles, develop faster from eggs to adult than the wild-type: perL mutants, which have long 28 h circadian rhythms, complete development more slowly than the wild-type. These results suggest that endogenous timers may be involved in regulating the development time of D. melanogaster.
Similar content being viewed by others
Article PDF
References
Bargiello, C A, and Young, M W. 1984. Molecular Genetics of A Biological Clock in Drosophila. Proc. Natl Acad. Sei. Usa., 81, 2142–2146.
Bargiello, T A, Jackson, F R, and Young, M W. 1984. Restoration of circadian behavioural rythms by gene transfer in Drosophila. Nature, 312, 752–754.
Bargiello, T A, Saez, T A, Baylies, M K, Gasic, G, Young, M W, and Spray, D C. 1987. The Drosophila gene per affects intercellular junctional communication. Nature, 328, 686–691.
Baylies, M K, Bargiello, T A, Jackson, F R, and Young, M W. 1987. Changes in abundance or structure of the per gene product can alter periodicity of the Drosophila clock. Nature, 326, 390–392.
Borteaux, A, Goldbeater, A, and Hess, B. 1975. Control of Oscillating Glycolysis of Yeast By Stochastic, Periodic and Steady Source of Substrate: A Model and Experimental Study. Proc. Natl Acad. Sei. Usa., 72, 3829–3833.
Chance, B, Eastbrook, R W, and Ghosh, A. 1964. Damped Sinusoidal Oscillations of Cytoplasmic Reduced Pyridine Nucleoutides in Yeast Cells. Proc Natl Acad Sei Usa, 51, 1244–1251.
Cooke, J, and Zeeman, E C. 1976. A clock and wavefront model for control of the number of repeated structures during animal morphogenesis. J Theoret Biol, 58, 455–476.
Dowse, H B, Hall, J C, and Ringo, J M. 1987. Circadian and ultradian rhythms in period mutants of Drosophila melanogaster. Behav Genet, 17, 19–35.
Dowse, H B, and Ringo, J M. 1987. Further evidence that circadian rhythms in Drosophila are a population of coupled ultradian oscillators. J Biol Rhythms, 2, 65–76.
Dowse, H B, Kass, L, and Ringo, J M. 1988. Studies on a congenital heart defect in Drosophila. Behav. Genet, 18, S714–715.
Ewer, J, Rosbash, M, and Hall, J C. 1988. An inducible promotor fused to the period gene in Drosophila conditionally rescues adult per-mutant arrhythmicity. Nature, 333, 82–84.
Frank, K D, and Zimmerman, W F. 1969. Action spectra for phase shifts of a circadian rhythm in Drosophila. Science, 168, 688–689.
Hall, J C, and Rosbash, M. 1988. Mutations and molecules influencing biological rhythms. Ann Rev Neurosci, 11, 373–393.
Hamblen, M, Zehring, W A, Kyriacou, C P, Reddy, P, Yu, Q, Wheeler, D A, Zweibel, O L J, Konopka, R J, Rosbash, M, and Hall, J C. 1986. Germ-line transformation involving DNA from the period locus in Drosophila melanogaster. Overlapping genomic fragments that restore circadian and ultradian rhythmicity to pero and per mutants. J Neurogenet, 3, 249–291.
Jackson, F R, Bargiello, T A, Yun, S H, and Young, M W. 1986. Product of per of Drosophila shares homology with proteoglycans. Nature, 320, 185–188.
James, A A, Ewer, J, Reddy, P, Hall, J C, and Rosbash, M. 1986. Embryonic Expression of the Period Clock Gene of Drosophila Melanogaster. Embo J. 5, 2313–2320.
Konopka, R J. 1972. Circadian Clock Mutants of Drosophila melanogaster. Ph.D. Thesis. California Institute of Technology.
Konopka, R J, and Benzer, S. 1971. Clock Mutants of Drosophila Melanogaster. Proc. Natl Acad. Sei. Usa, 68, 2112–2116.
Kyriacou, C P, and Hall, J C. 1980. Circadian Rhythm Mutations in Drosophila Affect Short-Term Fluctuations in The Male'S Courtship Song. Proc Natl Acad Sei Usa, 77, 6929–6933.
Kyriacou, C P. 1985. Long term ebony polymorphisms: a comparison of the contributions of behavioural and nonbehavioural fitness characters. Behav Genet, 15, 165–180.
Kyriacou, C P, and Burnet, B. 1979. Genetic analysis of phototaxis near the upper limit of the visual spectrum in D. melanogaster. Behav Genet, 123–128.
Liu, X, Lorenz, L, Yu, Q, Hall, J C, and Rosbash, M. 1988. Spatial and Temporal Expression of The Period Gene in Drosophila Melanogaster. 1988. Genes & Development, 2, 228–238.
Livingstone, M S. 1981. Two mutations in Drosophila affect the synthesis of octopamine, dopamine and serotonin by altering the activities of two different amino-acid decarboxylases. Neurosci Abst, 8, 384.
Lloyd, D, and Edwards, S W. 1984. Epigenetic oscillation during the cell cycles of lower eukaryotes are coupled to a clock. In Edmunds, L. N. (ed.) Cell Cycle Clocks, Marcel Dekker, New York, pp. 27–46.
Lo, C W, and Gilula, N B. 1979. Gap Junctional Communication in The Post-Implantation Mouse Embryo. Cell, 18, 411–422.
Pittendrigh, C S. 1974. Circadian oscillations in cells and the circadian organisation of multicellular systems. In Schmit, F. O. and Worden, F. G. (eds) The Neurosciences: Third Study Program, Cambridge, MIT Press, pp. 437–458.
Reddy, P, Jacquier, A C, Abovich, N, Petersen, G, and Rosbash, M. 1986. The period clock locus of D. melanogaster codes for a proteoglycan. Cell, 46, 53–61.
Serras, F, and Van Den Bigelaar, J A M. 1987. Is a mosaic embryo also a mosaic of communciation compartments Dev Biol, 120, 132–138.
Siegel, S. 1956. Nonparametric Statistics for the Behavioural Sciences. Tokyo, McGraw-Hill.
Siwicki, K K, Eastman, C, Petersen, G, Rosbash, M, and Hall, J C. 1988. Antibodies to the period gene product of Drosophila reveal diverse tissue distribution and rhythmic changes in the visual system. Neuron, 1, 141–150.
Snow, M H L, and Tam, P L. 1980. Timing in embryological development. Nature, 286, 107.
Spray, D C, Fujita, M, Saez, J C, Choi, H, Watanabe, T, Heitzberg, L, Rosenberg, C, and Reid, L M. 1987. Proteoglycans and glycasominoglycans induce gap junction synthesis and function in primary liver cell cultures. J Cell Biol, 105, 541–551.
Warner, A. 1985. The role of gap junctions in amphibian development. J Embr Exp Morph Suppl, 89, 365–380.
Yu, Q, Jacquier, A C, Citri, Y, and Colot, H M. 1987. Molecular mapping of point mutations in the period gene that stop or speed up biological clocks in Drosophila melanogaster. Proc. Natl Acad. Sei. USA, 84, 784–788.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kyriacou, C., Oldroyd, M., Wood, J. et al. Clock mutations alter developmental timing in Drosophila. Heredity 64, 395–401 (1990). https://doi.org/10.1038/hdy.1990.50
Received:
Issue Date:
DOI: https://doi.org/10.1038/hdy.1990.50
This article is cited by
-
Impact of disabled circadian clock on yellow fever mosquito Aedes aegypti fitness and behaviors
Scientific Reports (2022)
-
Environmental cycles regulate development time via circadian clock mediated gating of adult emergence
BMC Developmental Biology (2018)
-
Life-history traits of Drosophila melanogaster populations exhibiting early and late eclosion chronotypes
BMC Evolutionary Biology (2016)
-
Interaction of light regimes and circadian clocks modulate timing of pre-adult developmental events in Drosophila
BMC Developmental Biology (2014)
-
Circadian clocks of faster developing fruit fly populations also age faster
Biogerontology (2014)