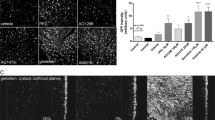
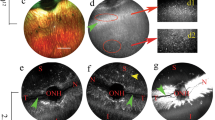
Abstract
We previously designed a modified channelrhodopsin-1 (mVChR1) protein chimera with a broader action than that of Chlamydomonas channelrhodopsin-2 and reported that its transduction into retinal ganglion cells can restore visual function in genetically blind, dystrophic Royal College of Surgeons (RCS) rats, with photostimuli ranging from 486 to 640 nm. In the current study, we sought to investigate the safety and influence of mVChR1 transgene expression. Adeno-associated virus type 2 encoding mVChR1 was administered by intravitreous injection into dystrophic RCS rats. Reverse-transcription PCR was used to monitor virus and transgene dissemination and the results demonstrated that their expression was restricted specifically within the eye tissues, and not in non-target organs. Moreover, examination of the blood, plasma and serum revealed that no excess immunoreactivity was present, as determined using standard clinical hematological parameters. Serum antibodies targeting the recombinant adeno-associated virus (rAAV) capsid increased after the injection; however, no increase in mVChR1 antibody was detected during the observation period. In addition, retinal histological examination showed no signs of inflammation in rAAV-injected rats. In conclusion, our results demonstrate that mVChR1 can be exogenously expressed without harmful immunological reactions in vivo. These findings will aid in studies of AAV gene transfer to restore vision in late-stage retinitis pigmentosa.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Hartong DT, Berson EL, Dryja TP . Retinitis pigmentosa. Lancet 2006; 368: 1795–1809.
Leveillard T, Sahel JA . Rod-derived cone viability factor for treating blinding diseases: from clinic to redox signaling. Sci Transl Med 2010; 2: 26ps16.
Dalkara D, Kolstad KD, Guerin KI, Hoffmann NV, Visel M, Klimczak RR et al. AAV mediated GDNF secretion from retinal glia slows down retinal degeneration in a rat model of retinitis pigmentosa. Mol Ther 2011; 19: 1602–1608.
Nagel G, Szellas T, Huhn W, Kateriya S, Adeishvili N, Berthold P et al. Channelrhodopsin-2, a directly light-gated cation-selective membrane channel. Proc Natl Acad Sci USA 2003; 100: 13940–13945.
Bi A, Cui J, Ma YP, Olshevskaya E, Pu M, Dizhoor AM et al. Ectopic expression of a microbial-type rhodopsin restores visual responses in mice with photoreceptor degeneration. Neuron 2006; 50: 23–33.
Zhang Y, Ivanova E, Bi A, Pan ZH . Ectopic expression of multiple microbial rhodopsins restores ON and OFF light responses in retinas with photoreceptor degeneration. J Neurosci 2009; 29: 9186–9196.
Osawa S, Iwasaki M, Hosaka R, Matsuzaka Y, Tomita H, Ishizuka T et al. Optogenetically induced seizure and the longitudinal hippocampal network dynamics. PLoS One 2013; 8: e60928.
Semo M, Gias C, Ahmado A, Sugano E, Allen AE, Lawrence JM et al. Dissecting a role for melanopsin in behavioural light aversion reveals a response independent of conventional photoreception. PLoS One 2010; 5: e15009.
Busskamp V, Duebel J, Balya D, Fradot M, Viney TJ, Siegert S et al. Genetic reactivation of cone photoreceptors restores visual responses in retinitis pigmentosa. Science 2010; 329: 413–417.
Lagali PS, Balya D, Awatramani GB, Munch TA, Kim DS, Busskamp V et al. Light-activated channels targeted to ON bipolar cells restore visual function in retinal degeneration. Nature Neurosci 2008; 11: 667–675.
Wang H, Sugiyama Y, Hikima T, Sugano E, Tomita H, Takahashi T et al. Molecular determinants differentiating photocurrent properties of two channelrhodopsins from chlamydomonas. J Biol Chem 2009; 284: 5685–5696.
Tomita H, Sugano E, Murayama N, Ozaki T, Nishiyama F, Tabata K et al. Restoration of the majority of the visual spectrum by using modified Volvox channelrhodopsin-1. Mol Ther 2014; 22: 1434–1440.
Mace E, Caplette R, Marre O, Sengupta A, Chaffiol A, Barbe P et al. Targeting channelrhodopsin-2 to ON-bipolar cells with vitreally administered AAV restores ON and OFF visual responses in blind mice. Mol Ther 2015; 23: 7–16.
Tomita H, Sugano E, Yawo H, Ishizuka T, Isago H, Narikawa S et al. Restoration of visual response in aged dystrophic RCS rats using AAV-mediated channelopsin-2 gene transfer. Invest Ophthalmol Vis Sci 2007; 48: 3821–3826.
Tomita H, Sugano E, Fukazawa Y, Isago H, Sugiyama Y, Hiroi T et al. Visual properties of transgenic rats harboring the channelrhodopsin-2 gene regulated by the thy-1.2 promoter. PLoS One 2009; 4: e7679.
Tomita H, Sugano E, Isago H, Tamai M . Channelrhodopsins provide a breakthrough insight into strategies for curing blindness. J Genet 2009; 88: 409–415.
Tomita H, Sugano E, Isago H, Hiroi T, Wang Z, Ohta E et al. Channelrhodopsin-2 gene transduced into retinal ganglion cells restores functional vision in genetically blind rats. Exp Eye Res 2010; 90: 429–436.
Isago H, Sugano E, Wang Z, Murayama N, Koyanagi E, Tamai M et al. Age-dependent differences in recovered visual responses in Royal College of Surgeons rats transduced with the Channelrhodopsin-2 gene. J Mol Neurosci 2012; 46: 393–400.
Sugano E, Isago H, Wang Z, Murayama N, Tamai M, Tomita H . Immune responses to adeno-associated virus type 2 encoding channelrhodopsin-2 in a genetically blind rat model for gene therapy. Gene Ther 2011; 18: 266–274.
Zhang F, Prigge M, Beyriere F, Tsunoda SP, Mattis J, Yizhar O et al. Red-shifted optogenetic excitation: a tool for fast neural control derived from Volvox carteri. Nat Neurosci 2008; 11: 631–633.
Prusky GT, Alam NM, Beekman S, Douglas RM . Rapid quantification of adult and developing mouse spatial vision using a virtual optomotor system. Invest Ophthalmol Vis Sci 2004; 45: 4611–4616.
Matsuzawa T, Nomura M, Unno T . Clinical pathology reference ranges of laboratory animals. Working Group II, Nonclinical Safety Evaluation Subcommittee of the Japan Pharmaceutical Manufacturers Association. The Journal of Veterinary Medical Science/The Japanese Society of Veterinary Science 1993; 55: 351–362.
Mainous AG 3rd, Pearson WS . Aspirin and ibuprofen: potential mediators of the cardiovascular risk due to smoking? Fam Med 2003; 35: 112–118.
Verma S, Szmitko PE, Ridker PM . C-reactive protein comes of age. Nat Clin Pract Cardiovasc Med 2005; 2: 29–36; quiz 58.
Sun H, Koike T, Ichikawa T, Hatakeyama K, Shiomi M, Zhang B et al. C-reactive protein in atherosclerotic lesions: its origin and pathophysiological significance. Am J Pathol 2005; 167: 1139–1148.
Szalai AJ, McCrory MA, Xing D, Hage FG, Miller A, Oparil S et al. Inhibiting C-reactive protein for the treatment of cardiovascular disease: promising evidence from rodent models. Mediators Inflamm 2014; 2014: 353614.
Dehghan A, van Hoek M, Sijbrands EJ, Stijnen T, Hofman A, Witteman JC . Risk of type 2 diabetes attributable to C-reactive protein and other risk factors. Diabetes Care 2007; 30: 2695–2699.
Zhang YC, Powers M, Wasserfall C, Brusko T, Song S, Flotte T et al. Immunity to adeno-associated virus serotype 2 delivered transgenes imparted by genetic predisposition to autoimmunity. Gene Ther 2004; 11: 233–240.
Simpson E . A historical perspective on immunological privilege. Immunol Rev 2006; 213: 12–22.
Willett K, Bennett J . Immunology of AAV-mediated gene transfer in the eye. Front Immunol 2013; 4: 261.
Li W, Kong F, Li X, Dai X, Liu X, Zheng Q et al. Gene therapy following subretinal AAV5 vector delivery is not affected by a previous intravitreal AAV5 vector administration in the partner eye. Mol Vis 2009; 15: 267–275.
Li Q, Miller R, Han PY, Pang J, Dinculescu A, Chiodo V et al. Intraocular route of AAV2 vector administration defines humoral immune response and therapeutic potential. Mol Vis 2008; 14: 1760–1769.
Takuwa H, Matsuura T, Obata T, Kawaguchi H, Kanno I, Ito H . Hemodynamic changes during somatosensory stimulation in awake and isoflurane-anesthetized mice measured by laser-Doppler flowmetry. Brain Res 2012; 1472: 107–112.
Donsante A, Miller DG, Li Y, Vogler C, Brunt EM, Russell DW et al. AAV vector integration sites in mouse hepatocellular carcinoma. Science 2007; 317: 477.
Li H, Malani N, Hamilton SR, Schlachterman A, Bussadori G, Edmonson SE et al. Assessing the potential for AAV vector genotoxicity in a murine model. Blood 2011; 117: 3311–3319.
Black S, Kushner I, Samols D . C-reactive protein. J Biol Chem 2004; 279: 48487–48490.
Yeh ET . A new perspective on the biology of C-reactive protein. Circ Res 2005; 97: 609–611.
Nunomura W . C-reactive protein in rat: in development, pregnancy and effect of sex hormones. Comp Biochem Physiol A Comp Physiol 1990; 96: 489–493.
Hellstrom M, Ruitenberg MJ, Pollett MA, Ehlert EM, Twisk J, Verhaagen J et al. Cellular tropism and transduction properties of seven adeno-associated viral vector serotypes in adult retina after intravitreal injection. Gene Ther 2009; 16: 521–532.
Sugano E, Murayama N, Takahashi M, Tabata K, Tamai M, Tomita H . Essential role of thioredoxin 2 in mitigating oxidative stress in retinal epithelial cells. J Ophthalmol 2013; 2013: 185825.
Sugano E, Tomita H, Ishiguro S, Isago H, Tamai M . Nitric oxide-induced accumulation of lipofuscin-like materials is caused by inhibition of cathepsin S. Curr Eye Res 2006; 31: 607–616.
Dhingra R, Gona P, Nam BH, D'Agostino RB Sr., Wilson PW, Benjamin EJ et al. C-reactive protein, inflammatory conditions, and cardiovascular disease risk. Am J Med 2007; 120: 1054–1062.
Hong T, Tan AG, Mitchell P, Wang JJ . A review and meta-analysis of the association between C-reactive protein and age-related macular degeneration. Surv Ophthalmol 2011; 56: 184–194.
Nita M, Grzybowski A, Ascaso FJ, Huerva V . Age-related macular degeneration in the aspect of chronic low-grade inflammation (pathophysiological parainflammation). Mediators Inflamm 2014; 2014: 930671.
Vine AK, Stader J, Branham K, Musch DC, Swaroop A . Biomarkers of cardiovascular disease as risk factors for age-related macular degeneration. Ophthalmology 2005; 112: 2076–2080.
Auricchio A, Hildinger M, O'Connor E, Gao GP, Wilson JM . Isolation of highly infectious and pure adeno-associated virus type 2 vectors with a single-step gravity-flow column. Hum Gene Ther 2001; 12: 71–76.
Auricchio A, O'Connor E, Hildinger M, Wilson JM . A single-step affinity column for purification of serotype-5 based adeno-associated viral vectors. Mol Ther 2001; 4: 372–374.
Sugano E, Tomita H, Ishiguro S, Abe T, Tamai M . Establishment of effective methods for transducing genes into iris pigment epithelial cells by using adeno-associated virus type 2. Invest Ophthalmol Vis Sci 2005; 46: 3341–3348.
Mandal MN, Moiseyev GP, Elliott MH, Kasus-Jacobi A, Li X, Chen H et al. Alpha-phenyl-N-tert-butylnitrone (PBN) prevents light-induced degeneration of the retina by inhibiting RPE65 protein isomerohydrolase activity. J Biol Chem 2011; 286: 32491–32501.
Acknowledgements
This work was partly supported by Grants-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan (nos. 25462747, 24390393 and 26670750); the Program for the Promotion of Fundamental Studies in Health Sciences of the National Institute of Biomedical Innovation (no. 10-06, NIBIO); Translational Research Network program (no. 3805140342); and the Japanese Retinitis Pigmentosa Society. We express our heartfelt appreciation to Ichiro Hagimori of Narita Animal Science Laboratory, whose enormous support and insightful comments were invaluable during the course of this study. We also express our heartfelt appreciation to Chikako Yamada of Center for Developmental Biology, RIKEN for technical advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Sugano, E., Tabata, K., Takahashi, M. et al. Local and systemic responses following intravitreous injection of AAV2-encoded modified Volvox channelrhodopsin-1 in a genetically blind rat model. Gene Ther 23, 158–166 (2016). https://doi.org/10.1038/gt.2015.99
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2015.99
This article is cited by
-
Development of an optogenetic gene sensitive to daylight and its implications in vision restoration
npj Regenerative Medicine (2021)
-
A 2020 vision of ocular gene therapy
Gene Therapy (2021)