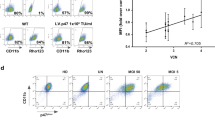
Abstract
Proto-oncogene activation caused by retroviral vector integration can cause malignancies in gene therapy trials. This has led investigators to search for less genotoxic vectors with minimal enhancer activity and a decreased risk of influencing neighboring chromosomal gene expression after integration. We previously showed that foamy virus (FV) vectors expressing the canine CD18 gene from an internal murine stem cell virus (MSCV) promoter could cure canine leukocyte adhesion deficiency (LAD). Here, we have repeated these studies using a FV vector expressing canine CD18 from a phosphoglycerate kinase (PGK) gene promoter. In vitro analysis showed that this vector did not contain an enhancer that activated neighboring genes, and it expressed CD18 efficiently in canine neutrophils and CD34+ cells. However, dogs that received hematopoietic stem cells transduced with the PGK-CD18 vector continued to suffer from LAD, and sometimes died prematurely of the disease. These studies show that the PGK promoter cannot effectively replace the MSCV promoter in CD18-expressing FV vectors, and they suggest that vectors containing a strong promoter–enhancer may be necessary for the treatment of human LAD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Gaspar HB, Parsley KL, Howe S, King D, Gilmour KC, Sinclair J et al. Gene therapy of X-linked severe combined immunodeficiency by use of a pseudotyped gammaretroviral vector. Lancet 2004; 364: 2181–2187.
Cavazzana-Calvo M, Hacein-Bey S, de Saint Basile G, Gross F, Yvon E, Nusbaum P et al. Gene therapy of human severe combined immunodeficiency (SCID)-X1 disease. Science 2000; 288: 669–672.
Aiuti A, Slavin S, Aker M, Ficara F, Deola S, Mortellaro A et al. Correction of ADA-SCID by stem cell gene therapy combined with nonmyeloablative conditioning. Science 2002; 296: 2410–2413.
Cartier N, Hacein-Bey-Abina S, Bartholomae CC, Veres G, Schmidt M, Kutschera I et al. Hematopoietic stem cell gene therapy with a lentiviral vector in X-linked adrenoleukodystrophy. Science 2009; 326: 818–823.
Ott MG, Schmidt M, Schwarzwaelder K, Stein S, Siler U, Koehl U et al. Correction of X-linked chronic granulomatous disease by gene therapy, augmented by insertional activation of MDS1-EVI1, PRDM16 or SETBP1. Nat Med 2006; 12: 401–409.
Howe SJ, Mansour MR, Schwarzwaelder K, Bartholomae C, Hubank M, Kempski H et al. Insertional mutagenesis combined with acquired somatic mutations causes leukemogenesis following gene therapy of SCID-X1 patients. J Clin Invest 2008; 118: 3143–3150.
Hacein-Bey-Abina S, Von Kalle C, Schmidt M, McCormack MP, Wulffraat N, Leboulch P et al. LMO2-associated clonal T cell proliferation in two patients after gene therapy for SCID-X1. Science 2003; 302: 415–419.
Wu X, Li Y, Crise B, Burgess SM . Transcription start regions in the human genome are favored targets for MLV integration. Science 2003; 300: 1749–1751.
Modlich U, Bohne J, Schmidt M, von Kalle C, Knoss S, Schambach A et al. Cell-culture assays reveal the importance of retroviral vector design for insertional genotoxicity. Blood 2006; 108: 2545–2553.
Zychlinski D, Schambach A, Modlich U, Maetzig T, Meyer J, Grassman E et al. Physiological promoters reduce the genotoxic risk of integrating gene vectors. Mol Therapy 2008; 16: 718–725.
Schroder AR, Shinn P, Chen H, Berry C, Ecker JR, Bushman F . HIV-1 integration in the human genome favors active genes and local hotspots. Cell 2002; 110: 521–529.
McBurney MW, Sutherland LC, Adra CN, Leclair B, Rudnicki MA, Jardine K . The mouse Pgk-1 gene promoter contains an upstream activator sequence. Nucleic Acids Res 1991; 19: 5755–5761.
Hendrie PC, Huo Y, Stolitenko RB, Russell DW . A rapid and quantitative assay for measuring neighboring gene activation by vector proviruses. Mol Therapy 2008; 16: 534–540.
Montini E, Cesana D, Schmidt M, Sanvito F, Ponzoni M, Bartholomae C et al. Hematopoietic stem cell gene transfer in a tumor-prone mouse model uncovers low genotoxicity of lentiviral vector integration. Nat Biotechnol 2006; 24: 687–696.
Montini E, Cesana D, Schmidt M, Sanvito F, Bartholomae CC, Ranzani M et al. The genotoxic potential of retroviral vectors is strongly modulated by vector design and integration site selection in a mouse model of HSC gene therapy. J Clin Invest 2009; 119: 964–975.
Falcone V, Schweizer M, Neumann-Haefelin D . Replication of primate foamy viruses in natural and experimental hosts. Curr Top Microbiol Immunol 2003; 277: 161–180.
Trobridge G, Josephson N, Vassilopoulos G, Mac J, Russell DW . Improved foamy virus vectors with minimal viral sequences. Mol Therapy 2002; 6: 321–328.
Heinkelein M, Dressler M, Jarmy G, Rammling M, Imrich H, Thurow J et al. Improved primate foamy virus vectors and packaging constructs. J Virol 2002; 76: 3774–3783.
Mergia A, Heinkelein M . Foamy virus vectors. Curr Top Microbiol Immunol 2003; 277: 131–159.
Josephson NC, Trobridge G, Russell DW . Transduction of long-term and mobilized peripheral blood-derived NOD/SCID repopulating cells by foamy virus vectors. Hum Gene Therapy 2004; 15: 87–92.
Josephson NC, Vassilopoulos G, Trobridge GD, Priestley GV, Wood BL, Papayannopoulou T et al. Transduction of human NOD/SCID-repopulating cells with both lymphoid and myeloid potential by foamy virus vectors. Proc Natl Acad Sci USA 2002; 99: 8295–8300.
Vassilopoulos G, Trobridge G, Josephson NC, Russell DW . Gene transfer into murine hematopoietic stem cells with helper-free foamy virus vectors. Blood 2001; 98: 604–609.
Kiem HP, Allen J, Trobridge G, Olson E, Keyser K, Peterson L et al. Foamy virus-mediated gene transfer to canine repopulating cells. Blood 2007; 109: 65–70.
Leurs C, Jansen M, Pollok KE, Heinkelein M, Schmidt M, Wissler M et al. Comparison of three retroviral vector systems for transduction of nonobese diabetic/severe combined immunodeficiency mice repopulating human CD34(+) cord blood cells. Hum Gene Therapy 2003; 14: 509–519.
Trobridge GD, Allen JM, Peterson L, Ironside CG, Russell D, Kiem HP . Foamy and lentiviral vectors transduce canine long-term repopulating cells at similar efficiency. Hum Gene Therapy 2009; 20: 519–523.
Zucali JR, Ciccarone T, Kelley V, Park J, Johnson CM, Mergia A . Transduction of umbilical cord blood CD34+ NOD/SCID-repopulating cells by simian foamy virus type 1 (SFV-1) vector. Virology 2002; 302: 229–235.
Trobridge GD, Miller DG, Jacobs MA, Allen JM, Kiem HP, Kaul R et al. Foamy virus vector integration sites in normal human cells. Proc Natl Acad Sci USA 2006; 103: 1498–1503.
Bauer Jr TR, Allen JM, Hai M, Tuschong LM, Khan IF, Olson EM et al. Successful treatment of canine leukocyte adhesion deficiency by foamy virus vectors. Nat Med 2008; 14: 93–97.
Anderson DC, Smith CW . Leukocyte adhesion deficiencies. In: Scriver CR, Sly WS (eds). The Metabolic and Molecular Bases of Inherited Disease. McGraw-Hill: New York, NY, 2001, pp 4829–4856.
Kijas JM, Bauer Jr TR, Gafvert S, Marklund S, Trowald-Wigh G, Johannisson A et al. A missense mutation in the beta-2 integrin gene (ITGB2) causes canine leukocyte adhesion deficiency. Genomics 1999; 61: 101–107.
Gu YC, Bauer Jr TR, Ackermann MR, Smith CW, Kehrli Jr ME, Starost MF et al. The genetic immunodeficiency disease, leukocyte adhesion deficiency, in humans, dogs, cattle, and mice. Comp Med 2004; 54: 363–372.
Hawley RG, Lieu FH, Fong AZ, Hawley TS . Versatile retroviral vectors for potential use in gene therapy. Gene Therapy 1994; 1: 136–138.
Uetsuki T, Naito A, Nagata S, Kaziro Y . Isolation and characterization of the human chromosomal gene for polypeptide chain elongation factor-1 alpha. J Biol Chem 1989; 264: 5791–5798.
Ye ZQ, Qiu P, Burkholder JK, Turner J, Culp J, Roberts T et al. Cytokine transgene expression and promoter usage in primary CD34+ cells using particle-mediated gene delivery. Hum Gene Therapy 1998; 9: 2197–2205.
Schambach A, Bohne J, Chandra S, Will E, Margison GP, Williams DA et al. Equal potency of gammaretroviral and lentiviral SIN vectors for expression of O6-methylguanine-DNA methyltransferase in hematopoietic cells. Mol Therapy 2006; 13: 391–400.
Sumiyoshi T, Holt NG, Hollis RP, Ge S, Cannon PM, Crooks GM et al. Stable transgene expression in primitive human CD34(+) hematopoietic stem/progenitor cells, using the sleeping beauty transposon system. Hum Gene Therapy 2009; 20: 1607–1626.
Gunji Y, Nakamura M, Hagiwara T, Hayakawa K, Matsushita H, Osawa H et al. Expression and function of adhesion molecules on human hematopoietic stem cells: CD34+ LFA-1- cells are more primitive than CD34+ LFA-1+ cells. Blood 1992; 80: 429–436.
Bauer Jr TR, Creevy KE, Gu YC, Tuschong LM, Donahue RE, Metzger ME et al. Very low levels of donor CD18+ neutrophils following allogeneic hematopoietic stem cell transplantation reverse the disease phenotype in canine leukocyte adhesion deficiency. Blood 2004; 103: 3582–3589.
Nelson EJ, Tuschong LM, Hunter MJ, Bauer Jr TR, Burkholder TH, Hickstein DD . Lentiviral vectors incorporating a human elongation factor 1alpha promoter for the treatment of canine leukocyte adhesion deficiency. Gene Therapy 2010; 17: 672–677.
Baum C, Kustikova O, Modlich U, Li Z, Fehse B . Mutagenesis and oncogenesis by chromosomal insertion of gene transfer vectors. Hum Gene Therapy 2006; 17: 253–263.
Acknowledgements
This research was supported by US National Institutes of Health grants HL53750 and HL085107 and the Intramural Research Program of the US National Institutes of Health, National Cancer Institute, Center for Cancer Research. We thank Raisa Stolitenko for technical assistance, William Telford and Veena Kapoor for help with flow cytometry, and Dennis Hickstein for support and advice.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Bauer, T., Olson, E., Huo, Y. et al. Treatment of canine leukocyte adhesion deficiency by foamy virus vectors expressing CD18 from a PGK promoter. Gene Ther 18, 553–559 (2011). https://doi.org/10.1038/gt.2010.169
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/gt.2010.169
Keywords
This article is cited by
-
CRISPR-Cas9 fusion to dominant-negative 53BP1 enhances HDR and inhibits NHEJ specifically at Cas9 target sites
Nature Communications (2019)
-
Evidence for the in vivo safety of insulated foamy viral vectors
Gene Therapy (2017)
-
Long-Term Follow-up of Foamy Viral Vector-Mediated Gene Therapy for Canine Leukocyte Adhesion Deficiency
Molecular Therapy (2013)
-
Current progress on gene therapy for primary immunodeficiencies
Gene Therapy (2013)
-
Immunisation with foamy virus Bet fusion proteins as novel strategy for HIV-1 epitope delivery
Immunologic Research (2013)