Abstract
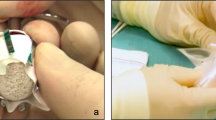
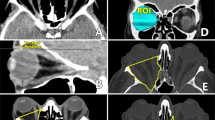
The hydroxyapatite orbital implant was first released for use as an orbital implant in humans in August 1989. It has been shown to be well tolerated, providing good motility of the artificial eye with a low complication rate when used as a primary implant.1 This prospective study evaluated the hydroxyapatite orbital implant used as both a primary and a secondary implant. Sixty patients were implanted between October 1992 and November 1994, 28 being implanted as a primary procedure at the time of enucleation or evisceration, and 32 as a secondary procedure. Seven patients underwent second-stage drilling and pegging of the implant. The mean follow-up time was 13 months (range 2-26 months). A standardised operative and post-operative protocol was followed. The patients were evaluated post-operatively for the amount of enophthalmos, degree of upper lid sulcus deformity, motility of the prosthesis, location of the implant in the socket, socket status and the presence or absence of discharge, position of the drill hole and coverage of the implant. Complications and their management were documented. Both patient and surgeon made a subjective assessment of cosmesis and the patient's satisfaction with the overall result was noted. The results of this study show the hydroxyapatite orbital implant to provide excellent motility of the artificial eye and good cosmesis with a low rate of complications when used both as a primary and as a secondary implant.
Similar content being viewed by others
Article PDF
References
Shields CL, Shields JA, De Potter P, Singh AD . Problems with the hydroxyapatite orbital implant: experience with 250 consecutive cases. Br J Ophthalmol 1994; 78: 702–6.
Mules PH . Evisceration of the globe with artificial vitreous. Trans Ophthalmol Soc UK 1885; 5: 200–6.
Leatherbarrow B, Kwartz J, Sunderland S, Brammar R, Nichol E . The ‘baseball’ orbital implant: a prospective study. Eye 1994; 8: 569–76.
Tyers AG, Collin JRO . Baseball orbital implants: a review of 39 patients. Br J Ophthalmol 1985; 69: 438–42.
Perry AC . Advances in enucleation. Ophthalmic Clin North Am 1991; 1: 173–82.
Ferrone PJ, Dutton JJ . Rate of vascularisation of coralline hydroxyapatite ocular implants. Ophthalmology 1992; 99: 376–9.
Goldberg RA, Holds JB, Ebrahimpour J . Exposed hydroxyapatite orbital implants: a report of six cases. Ophthalmology 1992; 99: 831–6.
De Potter P, Shields CL, Shields JA, Singh AD . Use of the hydroxyapatite ocular implant in the paediatric population. Arch Ophthalmol 1994; 112: 208–12.
Kim Y-D, Goldberg RA, Shorr N, Steinsapir KD . Management of exposed hydroxyapatite orbital implants. Ophthalmology 1994; 101: 1709–15.
Buettner H, Bartley GB . Tissue breakdown and exposure associated with orbital hydroxyapatite implants. Am J Ophthalmol 1992; 113: 669–73.
Dutton JJ . Coralline hydroxyapatite as an ocular implant. Ophthalmology 1991; 98: 370–7.
De Potter P, Shields CL, Shields JA, Flanders AE, Rao VM . The role of magnetic resonance imaging in the evaluation of the hydroxyapatite orbital implant. Ophthalmology 1992; 99: 824–30.
Shields CL, Shields JA, De Potter P . Hydroxyapatite orbital implant after enucleation. Arch Ophthalmol 1992; 110: 333–8.
Shields JA, Shields CL, De Potter P . Hydroxyapatite orbital implant after enucleation: experience with 200 cases. Mayo Clin Proc 1993; 68: 1191–5.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ashworth, J., Rhatigan, M., Sampath, R. et al. The hydroxyapatite orbital implant: A prospective study. Eye 10, 29–37 (1996). https://doi.org/10.1038/eye.1996.4
Issue Date:
DOI: https://doi.org/10.1038/eye.1996.4
Keywords
This article is cited by
-
Synthetic hydroxyapatite orbital implants: A clinical and MRI evaluation
Eye (1999)
-
The hydroxyapatite orbital implant: Post-operative pain
Eye (1998)
-
A study of the hydroxyapatite orbital implant drilling procedure
Eye (1998)
-
Letter
Eye (1997)
-
Orbital implants: Food for thought
Eye (1996)