Abstract
Objectives:
We investigated the correlation between Japanese apricot (JA) intake and Helicobacter pylori-related chronic atrophic gastritis (CAG).
Methods:
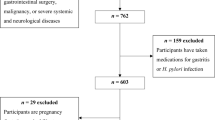
A questionnaire was administered and serum anti-H. pylori IgG antibodies measured in 1358 asymptomatic adults. The subjects were divided into high-intake and low-intake groups. Histological and serological evaluation of H. pylori-related CAG was performed in 68 non-elderly volunteers.
Results:
The H. pylori-negative rate did not differ significantly between the high-intake and low-intake groups. Mean antibody titers were lower in the high-intake group, but the difference was not significant. There was no significant difference in the rate of H. pylori infection on the basis of JA intake when subjects were stratified by age. Among H. pylori-positive non-elderly subjects, antibody titers were significantly lower in the high-intake group (P=0.041). Endoscopic tissue biopsy from the 68 volunteers showed less H. pylori bacterial load and mononuclear infiltration irrespective of gastric site in the high-intake group. In the high-intake group, antral neutrophil infiltration was significantly less pronounced and corporal atrophy was less extensive. Serological evaluation using serum PG levels also confirmed these histopathological data.
Conclusions:
Our findings strongly indicate a preventive effect of JA intake on CAG by inhibiting H. pylori infection and reducing active mucosal inflammation.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Becker S, Dossus L, Kaaks R (2009). Obesity related hyperinsulinaemia and hyperglycaemia and cancer development. Arch Physiol Biochem 115, 86–96.
Chen TS, Chang FY, Lee SD (1997). Serodiagnosis of Helicobacter pylori infection: comparison and correlation between enzyme-linked immunosorbent assay and rapid serological test results. J Clin Microbiol 35, 184–186.
Correa P, Houghton J (2007). Carcinogenesis of Helicobacter pylori. Gastroenterol 133, 659–672.
Dixon MF, Genta RM, Yardley JH, Correa P (1996). Classification and grading of gastritis. The updated Sydney System. International Workshop on the Histopathology of Gastritis, Houston 1994. Am J Surg Pathol 20, 1161–1181.
Eaton KA, Krakowka S (1992). Chronic active gastritis due to Helicobacter pylori in immunized gnotobiotic piglets. Gastroenterol 103, 1580–1586.
El-Omar EM, Carrington M, Chow WH, McColl KE, Bream JH, Young HA et al. (2000). Interleukin-1 polymorphisms associated with increased risk of gastric cancer. Nature 404, 398–402.
Fujita K, Hasegawa M, Fujita M, Kobayashi I, Ozasa K, Watanabe Y (2002). Anti-Helicobacter pylori effects of Bainiku-ekisu (concentrate of Japanese apricot juice). Nippon Shokakibyo Gakkai Zasshi 99, 379–385.
Hashimoto T, Yagami F, Owada M, Sugawara T, Kawamura M (2008). Salt preference according to a questionnaire vs dietary salt intake estimated by a spot urine method in participants at a health check-up center. Intern Med 47, 399–403.
Hatakeyama M (2004). Oncogenic mechanisms of the Helicobacter pylori CagA protein. Nat Rev Cancer 4, 688–694.
Ichinose M, Miki K, Furihata C, Kageyama T, Niwa H, Oka H et al. (1982). Radioimmunoassay of group II pepsinogen in human serum. Clin Chim Acta 122, 61–69.
Kabir S (2009). Effect of Helicobacter pylori eradication on incidence of gastric cancer in human and animal models: underlying biochemical and molecular events. Helicobacter 14, 159–171.
Kneller RW, You WC, Chang YS, Liu WD, Zhang L, Zhao L et al. (1992). Cigarette smoking and other risk factors for progression of precancerous stomach lesions. J Natl Cancer Inst 84, 1261–1266.
Kobayashi M, Tsubono Y, Sasazuki S, Sasaki S, Tsugane S (2002). Vegetables, fruit and risk of gastric cancer in Japan: a 10-year follow-up of the JPHC Study Cohort I. Int J Cancer 102, 39–44.
Loffeld RJ, Werdmuller BF, Kusters JG, Kuipers EJ (2000). IgG antibody titer against Helicobacter pylori correlates with presence of cytotoxin associated gene A-positive H.pylori strains. FEMS Immunol Med Microbiol 28, 139–141.
Mardh E, Mardh S, Mardh B, Borch K (2002). Diagnosis of gastritis by means of a combination of serological analyses. Clin Chim Acta 320, 17–27.
Miki K, Ichinose M, Shimizu A, Huang SC, Oka H, Furihata C et al. (1987). Serum pepsinogens as a screening test of extensive chronic gastritis. Gastroenterol Jpn 22, 133–141.
Miyazawa M, Utsunomiya H, Inada K, Yamada T, Okuno Y, Tanaka H et al. (2006). Inhibition of Helicobacter pylori motility by (+)-syringaresinol from unripe Japanese apricot. Biol Pharm Bull 29, 172–173.
Nozaki K, Shimizu N, Inada K, Tsukamoto T, Inoue M, Kumagai T et al. (2002). Synergistic promoting effects of Helicobacter pylori infection and high-salt diet on gastric carcinogenesis in Mongolian gerbils. Jpn J Cancer Res 93, 1083–1089.
Ohata H, Kitauchi S, Yoshimura N, Mugitani K, Iwane M, Nakamura H et al. (2004). Progression of chronic atrophic gastritis associated with Helicobacter pylori infection increases risk of gastric cancer. Int J Cancer 109, 138–143.
Ohta Y, Tsuchihashi T, Ueno M, Kajioka T, Onaka U, Tominaga M et al. (2004). Relationship between the awareness of salt restriction and the actual salt intake in hypertensive patients. Hypertension Res 27, 243–246.
Otsuka T (2005). Suppressive effect of fruit-juice concentrate of Prunus mume Sieb. et et Zucc. (Japanese apricot, Ume) on Helicobacter pylori-induced glandular stomach lesion in Mongolian gerbils. Asian Pacific J Cancer Prev 6, 337–341.
Plebani M, Basso D, Cassaro M, Brigato L, Scrigner M, Toma A et al. (1996). Helicobacter pylori serology in patients with chronic gastritis. Am J Gastroenterol 91, 954–958.
Samloff IM, Varis K, Ihamaki T, Siurala M, Rotter JI (1982). Relationships among serum pepsinogen I, serum pepsinogen II, and gastric mucosal histology. A study in relatives of patients with pernicious anemia. Gastroenterol 83, 204–209.
Shikata K, Kiyohara Y, Kubo M, Yonemoto K, Ninomiya T, Shirota T et al. (2006). A prospective study of dietary salt intake and gastric cancer incidence in a defined Japanese population: the Hisayama study. Int J Cancer 119, 196–201.
Shimizu N, Inada K, Nakanishi H, Tsukamoto T, Ikehara Y, Kaminishi M et al. (1999). Helicobacter pylori infection enhances glandular stomach carcinogenesis in Mongolian gerbils treated with chemical carcinogens. Carcinogenesis 20, 669–676.
Tredaniel J, Boffetta P, Buiatti E, Saracci R, Hirsch A (1997). Tobacco smoking and gastric cancer: review and meta-analysis. Int J Cancer 72, 565–573.
Utsunomiya H, Yamakawa T, Kamei J, Kadonosono K, Tanaka S (2005). Anti-hyperglycemic effects of plum in a rat model of obesity and type 2 diabetes, Wistar fatty rat. Biomed Res 26, 193–200.
Yamagata H, Kiyohara Y, Nakamura S, Kubo M, Tanizaki Y, Matsumoto T et al. (2005). Impact of fasting plasma glucose levels on gastric cancer incidence in a general Japanese population: the Hisayama study. Diabetes Care 28, 789–794.
Yanaoka K, Oka M, Mukoubayashi C, Yoshimura N, Enomoto S, Iguchi M et al. (2008a). Cancer high-risk subjects identified by serum pepsinogen tests: outcomes after 10-year follow-up in asymptomatic middle-aged males. Cancer Epidemiol Biomarkers Prev 17, 838–845.
Yanaoka K, Oka M, Yoshimura N, Mukoubayashi C, Enomoto S, Iguchi M et al. (2008b). Risk of gastric cancer in asymptomatic, middle-aged Japanese subjects based on serum pepsinogen and Helicobacter pylori antibody levels. Int J Cancer 123, 917–926.
Yanaoka K, Oka M, Ohata H, Yoshimura N, Deguchi H, Mukoubayashi C et al. (2009). Eradication of Helicobacter pylori prevents cancer development in subjects with mild gastric atrophy identified by serum pepsinogen levels. Int J Cancer 125, 2697–2703.
Yu GP, Hsieh CC, Wang LY, Yu SZ, Li XL, Jin TH (1995). Green-tea consumption and risk of stomach cancer: a population-based case–control study in Shanghai, China. Cancer Causes Control 6, 532–538.
Acknowledgements
This work was supported in part by a Grant-in-Aid for Cancer Research from the Ministry of Health, Labor, and Welfare of Japan. We express our deepest thanks to Ms Kazu Konishi for excellent secretarial assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Enomoto, S., Yanaoka, K., Utsunomiya, H. et al. Inhibitory effects of Japanese apricot (Prunus mume Siebold et Zucc.; Ume) on Helicobacter pylori-related chronic gastritis. Eur J Clin Nutr 64, 714–719 (2010). https://doi.org/10.1038/ejcn.2010.70
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ejcn.2010.70
Keywords
This article is cited by
-
Komplementärmedizinische Behandlungsansätze bei Gastritis und funktionellen Magenbeschwerden während der Tumortherapie
Der Onkologe (2020)
-
Antioxidant activities and beneficial health effects of some dried fruits commonly consumed in Algeria: a review
Euro-Mediterranean Journal for Environmental Integration (2019)
-
Biological and epidemiological evidence of anti-allergic effects of traditional Japanese food ume (Prunus mume)
Scientific Reports (2018)
-
Safety and adherence of Umezu polyphenols in the Japanese plum (Prunus mume) in a 12-week double-blind randomized placebo-controlled pilot trial to evaluate antihypertensive effects
Environmental Health and Preventive Medicine (2014)
-
Japanese apricots reported to inhibit inflammation and gastritis progression related to Helicobacter pylori infection
Nature Reviews Gastroenterology & Hepatology (2010)