- NEWS AND VIEWS
Neuronal plasticity in nematode worms
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 553, 159-160 (2018)
doi: https://doi.org/10.1038/d41586-017-09031-5
References
Holtmaat, A. & Svoboda, K. Nature Rev. Neurosci. 10, 647–658 (2009).
Hart, M. P. & Hobert, O. Nature 553, 165–170 (2018).
Jin, Y. & Qi, Y. B. Curr. Opin. Neurobiol. 48, 97–105 (2017).
White, J. G., Southgate, E., Thomson, J. N. & Brenner, S. Phil. Trans. R. Soc. B 314, 1–340 (1986).
Emmons, S. W. Phil. Trans. R. Soc. B 370, 20140309 (2015).
Jarrell, T. A. et al. Science 337, 437–444 (2012).
LeBoeuf, B. & Garcia, L. R. G3 Genes Genomes Genet. 7, 647–662 (2017).
Williams, M. E., de Wit, J. & Ghosh, A. Neuron 68, 9–18 (2010).
Südhof, T. C. Cell 171, 745–769 (2017).
Portman, D. S. J. Neurosci. Res. 95, 527–538 (2017).

 Read the paper: Neurexin controls plasticity of a mature, sexually dimorphic neuron
Read the paper: Neurexin controls plasticity of a mature, sexually dimorphic neuron
 Forty years of cellular clues from worms
Forty years of cellular clues from worms
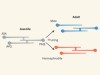 Wired for sex
Wired for sex