Illustration by Peter Horvath; Photos: Getty
In September last year, a handful of researchers were sitting around a computer monitor in chemist Kenchiro Itami’s lab at Japan’s Nagoya University as one loaded a file showing the results of an X-ray crystallography scan. Within seconds, the room erupted: scientists were on their feet, cheering and exchanging high fives. In front of them was a 3D representation of a carbon nanobelt — a new molecule of carbon that the team had successfully synthesized after 12 years of painstaking effort.
“Without these data, we couldn’t prove its structure 100%,” says Itami, director of the Institute of Transformative Bio-Molecules at Nagoya University. “It was the most exciting moment I ever had in my life.”
Itami thinks he has good reason to be so excited. “The discovery of a new form of carbon has always opened up new science and technology — fullerenes are a great example,” he says, referring to the all-carbon molecular spheres created by scientists at Rice University in Houston, Texas, in 1985.
Chemists have indeed tried many things when it comes to constructing exotic forms, or allotropes, of carbon: nanobelts are only the latest and it’s significant that a Japanese team made them. The country has enjoyed a rich history of manipulating carbon — an atom with the stability and strength to build complex molecules — into new materials with useful properties, and it has built a strong industrial sector from those basic research efforts.
But in the past decade or so, Japan has found itself beset by international competition, as progress in the field has shifted abroad. Now, Japanese researchers are fighting to maintain their global prominence.
No small pedigree
The history of nanocarbons is nearly as intricate as the structures themselves. Smiths have forged carbon with metal to make sharp, resilient weapons for thousands of years, with some astonishing results: a 2006 analysis of Damascus sabre steel from the seventeenth century1 revealed that the material contains microstructures such as carbon nanotubes — lattices of carbon atoms arranged to create a pipe with a diameter of 0.7–50 nanometres.
Modern synthesis of carbon nanotubes is widely attributed to Japanese scientists Morinobu Endo and Sumio Iijima. When Endo was working at the French National Centre for Scientific Research (CNRS) in the 1970s, he synthesized carbon fibres that featured hollow tubes arranged in concentric sheets like the rings of a tree. Sixteen years later, Iijima published a letter in Nature entitled ‘Helical microtubules of graphitic carbon’; he is often cited as the scientist who discovered carbon nanotubes2.
But who first made the discovery is still a topic of discussion. A 2006 editorial in Carbon3 suggested that Russian scientists L. V. Radushkevich and V. M. Lukyanovich should be credited for their 1952 paper4, which reported that “carbon filaments could be hollow and have a nanometre-size diameter”. The paper, published in Russian, was little-known outside the Soviet Union, and it was Japanese researchers who were responsible for opening up the field to the rest of the world. “From the beginning of the discovery, I was confident in the importance of nanotubes,” says Iijima, now a chemist at Meijo University in Nagoya.
He turned out to be right. The strength-to-weight ratio of the tiny molecules is at least 30 times greater than for Kevlar, the material used in bulletproof vests. Adding nanotube fibres to just about any material — from vehicle parts to golf-club shafts to elevator cables — will strengthen it enormously. And because they can conduct electricity better than copper, nanotubes have given rise to products such as transparent conductive films and even prototype computers.
As with many new materials, some applications can be unexpected. For instance, a nanotube yarn originally designed for lightweight data cables was re-engineered for use in space. NASA scientists think that the yarn — 200 times stronger and 5 times more elastic than steel — could significantly lower the overall mass of a spacecraft.
Itami’s carbon nanobelt is, essentially, the single repeating unit of a relatively tiny carbon nanotube (see ‘Nanocarbon molecules: a glossary’). But the simplicity of the nanobelt is misleading — synthesizing one is supremely difficult owing to the high degree of strain placed on the 12 benzene rings that must be joined together to create the belt.
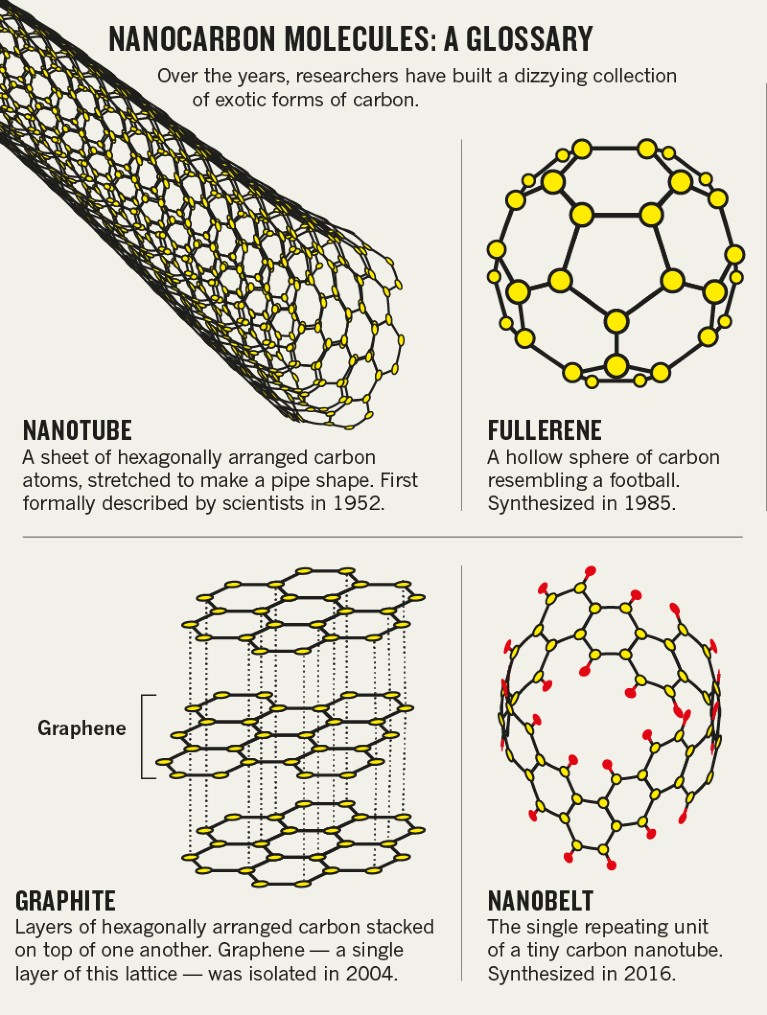
The effort could well be worthwhile, however. Nanobelts have been described as ‘dream molecules’ by researchers, because of their potential roles in everything from semiconductors to photonics equipment. They might even serve as “seeds” for growing “structurally well-defined carbon nanotubes”, Itami and his colleagues reported this year5.
Itami hopes that carbon nanobelts will become available to businesses in a matter of months. If they do, they will be making their way into a commercial field that is steadily filling up with exotic forms of carbon. A material known as Vantablack comprises millions of vertically aligned nanotubes, each with a diameter of about 20 nanometres. The nanotubes absorb 99.96% of incident radiation, according to Vantablack’s developers, Surrey NanoSystems in Newhaven, UK. That makes Vantablack the darkest synthetic substance in the world. As such, it could be used to protect telescopes from stray light, making it easier to see faint stars, or to improve the optical performance of satellites, among other applications.
And in 2013, researchers at Stanford University in California reported6 that they had made a basic computer from carbon-nanotube transistors and simple electronic circuits. The device drew attention as a possible replacement for silicon and as a way to facilitate the development of ever-smaller transistors.
Barriers to entry
The promise of nanotubes has been somewhat overshadowed by their potential health dangers. The Japanese government has been evaluating nanotubes for possible carcinogenic effects, and some scientists have likened the tubes to asbestos in terms of health risk, In 2015, one joint French–US study7 found nanotubes similar to those used in vehicle exhaust systems in tissue samples taken from 69 children in Paris with asthma. “These results strongly suggest that humans are routinely exposed to nanotubes,” the authors wrote.
Another roadblock to the application of nanotubes is price — 1 gram can cost as much as US$250, around 6 times the price of gold. Mass production would bring down that cost considerably, so an important area in nanotube research is the development of alternatives to the expensive, high-energy methods currently used, such as arc electrical discharge and chemical vapour deposition.
The possible effects on human health and the high price tag may have dampened global enthusiasm for nanocarbon products, and could have slowed down Japan’s academic output. The country also faces challenges in terms of its academic workforce. “Excellent chemists and physicists are still in Japan but the trend for training young Japanese scientists is decreasing because students are not motivated to pursue PhDs any more — instead they want to finish a master’s, as companies do not value PhDs,” says Mauricio Terrones, a nanotechnologist at Pennsylvania State University in University Park and an editor at Carbon. “Therefore, Japan needs a new policy to motivate industry to hire PhDs from Japanese universities. In this way, the next generation of chemists and physicists will continue to be first-class and competitive worldwide.”
Japan’s research performance doesn’t reflect the nuances of its relative strengths in the field. Japanese researchers excel in some areas, such as developing chemical techniques to grow nanotubes, but underperform in others, such as thermal engineering, says Takashi Kodama, a thermophysicist at the University of Tokyo.
Kodama returned to Japan early last year, after ten years at Stanford. “I’ve experienced both environments, the United States and Japan, and Japan’s advantage is its high-tech equipment,” he says. “Researchers here have easy access to excellent equipment at low cost, compared to the old, expensive equipment in the United States. Part of the reason is that it’s easier to spend research funds in Japan on equipment instead of just human resources.”
Economies of scale
Although its academic output may be smaller than that of other science superpowers, Japan has continued to make significant contributions to the development and applications of nanotubes and carbon fibres, with state-backed research centres working closely with private companies to produce high-tech commercial products. Toray Industries in Tokyo announced in 2014 that it would be the sole supplier of carbon fibres for Boeing’s upcoming 777X jet, extending its existing supply contract for the 787 Dreamliner, which made aviation history for its composite fuselage of carbon fibre coated with epoxy resin. Fujitsu, based in Tokyo and known for its supercomputers and servers, is looking into how nanotubes and graphene, another nanomaterial that has attracted immense interest, could help to miniaturize electronics.
Meanwhile, research organizations are continuing to expand understanding of the basic science of nanotubes while working on practical applications. Earlier this year, for example, the Tsukuba-based CNT-Application Research Center reported that it had developed a flexible, wearable nanotube strip that can sense and capture the hand movements of humans and robots. As a gesture-tracking device, it has potential health-care, information-technology and military applications.
In 2005, Yoshinori Ando, a materials scientist at Meijo University, set up Meijo Nano Carbon to manufacture nanotubes. In 2013, collaboration between Meijo Nano Carbon and Japan’s National Institute of Advanced Industrial Science and Technology (AIST) led to a joint production plant that uses a technology known as enhanced direct injection pyrolytic synthesis. According to AIST, manufacturing speeds are 100 times greater than those achieved previously, with higher-quality end products. The company’s hope is that mass production of high-quality nanotubes for the research and development market will accelerate commercial applications.
They face competition. In 2015, ZEON in Toyko began what it says was the first mass production of nanotubes using another technique developed at AIST, termed Super-growth. It expects to supply manufacturers of conductive and rubber materials and high-performance capacitors. Meanwhile, AIST is working with companies such as electronics maker Furukawa Electric in Tokyo, which is looking into how nanotubes could help to reduce the size of vehicle engines and therefore reduce carbon dioxide emissions.
Iijima is encouraged by the wide range of applications being explored. “Anyone can make a profit based on nanotube research in Japan or other countries,” he says. And he isn’t alone in his enthusiasm for Japan’s market potential. “I believe Japan is still leading carbon science and technology — just look around and the major applications of carbon materials are being developed there,” says Terrones. The fruits of this labour can be seen in the Boeing jets, sporting goods and oil-exploration equipment, he says. But he notes that companies are less willing than academic organizations to publicize their progress owing to intellectual-property concerns in a competitive sector.
As for Itami, the next step is to grow a nanobelt into a structurally pure nanotube. He also hopes to use nanobelts for small-molecule components in optoelectronic devices such as organic light-emitting diodes and solar cells.
“As in the history of fullerenes, one cannot really predict the extraordinary properties and functions of a new form of carbon,” Itami says. “Perhaps trying to discover them is going to be one of the most exciting journeys for scientists.”