Abstract
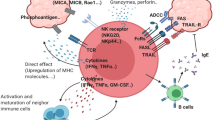
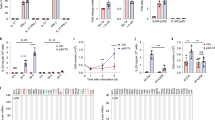
Interleukin 4 (IL-4) has a variety of immune functions, including helper T-cell (Th-cell) differentiation and innate immune-response processes. However, the impact of IL-4 on gamma delta (γδ) T cells remains unclear. In this study, we investigate the effects of IL-4 on the activation and proliferation of γδ T cells and the balance between variable delta 1 (Vδ1) and Vδ2 T cells in humans. The results show that IL-4 inhibits the activation of γδ T cells in the presence of γδ T-cell receptor (TCR) stimulation in a STAT6-dependent manner. IL-4 promoted the growth of activated γδ T cells and increased the levels of Vδ1 T cells, which in turn inhibited Vδ2 T-cell growth via significant IL-10 secretion. Vδ1 T cells secreted significantly less interferon gamma (IFNγ) and more IL-10 relative to Vδ2. Furthermore, Vδ1 T cells showed relatively low levels of Natural Killer Group 2D (NKG2D) expression in the presence of IL-4, suggesting that Vδ1 T cells weaken the γδ T cell-mediated anti-tumor immune response. For the first time, our findings demonstrate a negative regulatory role of IL-4 in γδ T cell-mediated anti-tumor immunity.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Kabelitz D, Wesch D, He W . Perspectives of gammadelta T cells in tumor immunology. Cancer Res 2007; 67: 5–8.
Thedrez A, Sabourin C, Gertner J, Devilder MC, Allain-Maillet S, Fournie JJ et al. Self/non-self discrimination by human gammadelta T cells: simple solutions for a complex issue? Immunol Rev 2007; 215: 123–135.
Hayday AC . [gamma][delta] cells: a right time and a right place for a conserved third way of protection. Annu Rev Immunol 2000; 18: 975–1026.
Willcox CR, Pitard V, Netzer S, Couzi L, Salim M, Silberzahn T et al. Cytomegalovirus and tumor stress surveillance by binding of a human gammadelta T cell antigen receptor to endothelial protein C receptor. Nat Immunol 2012; 13: 872–879.
Gober HJ, Kistowska M, Angman L, Jeno P, Mori L, de Libero G . Human T cell receptor gammadelta cells recognize endogenous mevalonate metabolites in tumor cells. J Exp Med 2003; 197: 163–168.
Bonneville M, O'Brien RL, Born WK . Gammadelta T cell effector functions: a blend of innate programming and acquired plasticity. Nat Rev Immunol 2010; 10: 467–478.
Champagne E . gammadelta T cell receptor ligands and modes of antigen recognition. Arch Immunol Ther Exp (Warsz) 2011; 59: 117–137.
Wu H, Knight JF, Alexander SI . Regulatory gamma delta T cells in Heymann nephritis express an invariant Vgamma6/Vdelta1 with a canonical CDR3 sequence. Eur J Immunol 2004; 34: 2322–2230.
Kuhl AA, Pawlowski NN, Grollich K, Blessenohl M, Westermann J, Zeitz M et al. Human peripheral gammadelta T cells possess regulatory potential. Immunology 2009; 128: 580–588.
Correia DV, Fogli M, Hudspeth K, da Silva MG, Mavilio D, Silva-Santos B . Differentiation of human peripheral blood Vdelta1+ T cells expressing the natural cytotoxicity receptor NKp30 for recognition of lymphoid leukemia cells. Blood 2011; 118: 992–1001.
Groh V, Steinle A, Bauer S, Spies T . Recognition of stress-induced MHC molecules by intestinal epithelial gammadelta T cells. Science 1998; 279: 1737–1740.
Knight A, Mackinnon S, Lowdell MW . Human Vdelta1 gamma-delta T cells exert potent specific cytotoxicity against primary multiple myeloma cells. Cytotherapy 2012; 14: 1110–1118.
Maeurer MJ, Martin D, Walter W, Liu K, Zitvogel L, Halusczcak K et al. Human intestinal Vdelta1+ lymphocytes recognize tumor cells of epithelial origin. J Exp Med 1996; 183: 1681–1696.
Poggi A, Venturino C, Catellani S, Clavio M, Miglino M, Gobbi M et al. Vdelta1 T lymphocytes from B-CLL patients recognize ULBP3 expressed on leukemic B cells and up-regulated by trans-retinoic acid. Cancer Res 2004; 64: 9172–9179.
Dechanet J, Merville P, Lim A, Retiere C, Pitard V, Lafarge X et al. Implication of gammadelta T cells in the human immune response to cytomegalovirus. J Clin Invest 1999; 103: 1437–1449.
Fenoglio D, Poggi A, Catellani S, Battaglia F, Ferrera A, Setti M et al. Vdelta1 T lymphocytes producing IFN-gamma and IL-17 are expanded in HIV-1-infected patients and respond to Candida albicans. Blood 2009; 113: 6611–6618.
Halary F, Pitard V, Dlubek D, Krzysiek R, de la Salle H, Merville P et al. Shared reactivity of Vδ2− γδ T cells against cytomegalovirus-infected cells and tumor intestinal epithelial cells. J Exp Med 2005; 201: 1567–1578.
Peng G, Wang HY, Peng W, Kiniwa Y, Seo KH, Wang RF . Tumor-infiltrating gammadelta T cells suppress T and dendritic cell function via mechanisms controlled by a unique Toll-like receptor signaling pathway. Immunity 2007; 27: 334–348.
Li Y, Koshiba T, Yoshizawa A, Yonekawa Y, Masuda K, Ito A et al. Analyses of peripheral blood mononuclear cells in operational tolerance after pediatric living donor liver transplantation. Am J Transplant 2004; 4: 2118–2125.
Martinez-Llordella M, Puig-Pey I, Orlando G, Ramoni M, Tisone G, Rimola A et al. Multiparameter immune profiling of operational tolerance in liver transplantation. Am J Transplant 2007; 7: 309–319.
Zhao X, Li Y, Ohe H, Nafady-Hego H, Uemoto S, Bishop GA et al. Intragraft Vdelta1 gammadelta T cells with a unique T-cell receptor are closely associated with pediatric semiallogeneic liver transplant tolerance. Transplantation 2013; 95: 192–202.
Hao J, Dong S, Xia S, He W, Jia H, Zhang S et al. Regulatory role of Vgamma1 gammadelta T cells in tumor immunity through IL-4 production. J Immunol 2011; 187: 4979–4986.
Wen L, Peakman M, Mieli-Vergani G, Vergani D . Elevation of activated gamma delta T cell receptor bearing T lymphocytes in patients with autoimmune chronic liver disease. Clin Exp Immunol 1992; 89: 78–82.
Rajan AJ, Asensio VC, Campbell IL, Brosnan CF . Experimental autoimmune encephalomyelitis on the SJL mouse: effect of gamma delta T cell depletion on chemokine and chemokine receptor expression in the central nervous system. J Immunol 2000; 164: 2120–2130.
Huber SA, Stone JE, Wagner DH Jr, Kupperman J, Pfeiffer L, David C et al. gamma delta+ T cells regulate major histocompatibility complex class II(IA and IE)-dependent susceptibility to coxsackievirus B3-induced autoimmune myocarditis. J Virol 1999; 73: 5630–5636.
Agrati C, Castilletti C, de Santis R, Cimini E, Bordi L, Malkovsky M et al. Interferon-gamma-mediated antiviral immunity against orthopoxvirus infection is provided by gamma delta T cells. J Infect Dis 2006; 193: 1606–1607; author reply 1607–1608.
Petermann F, Rothhammer V, Claussen MC, Haas JD, Blanco LR, Heink S et al. gammadelta T cells enhance autoimmunity by restraining regulatory T cell responses via an interleukin-23-dependent mechanism. Immunity 2010; 33: 351–363.
Veldhoen M, Uyttenhove C, van Snick J, Helmby H, Westendorf A, Buer J et al. Transforming growth factor-beta ‘reprograms’ the differentiation of T helper 2 cells and promotes an interleukin 9-producing subset. Nat Immunol 2008; 9: 1341–1346.
Dardalhon V, Awasthi A, Kwon H, Galileos G, Gao W, Sobel RA et al. IL-4 inhibits TGF-beta-induced Foxp3+ T cells and, together with TGF-beta, generates IL-9+ IL-10+ Foxp3− effector T cells. Nat Immunol 2008; 9: 1347–1355.
Onishi T, Ohishi Y, Imagawa K, Ohmoto Y, Murata K . An assessment of the immunological environment based on intratumoral cytokine production in renal cell carcinoma. BJU Int 1999; 83: 488–492.
Kawakami M, Kawakami K, Stepensky VA, Maki RA, Robin H, Muller W et al. Interleukin 4 receptor on human lung cancer: a molecular target for cytotoxin therapy. Clin Cancer Res 2002; 8: 3503–3511.
Kioi M, Takahashi S, Kawakami M, Kawakami K, Kreitman RJ, Puri RK . Expression and targeting of interleukin-4 receptor for primary and advanced ovarian cancer therapy. Cancer Res 2005; 65: 8388–8396.
Leland P, Taguchi J, Husain SR, Kreitman RJ, Pastan I, Puri RK . Human breast carcinoma cells express type II IL-4 receptors and are sensitive to antitumor activity of a chimeric IL-4–Pseudomonas exotoxin fusion protein in vitro and in vivo. Mol Med 2000; 6: 165–178.
Kawakami K, Kawakami M, Husain SR, Puri RK . Targeting interleukin-4 receptors for effective pancreatic cancer therapy. Cancer Res 2002; 62: 3575–3580.
Yi Y, He HW, Wang JX, Cai XY, Li YW, Zhou J et al. The functional impairment of HCC-infiltrating gammadelta T cells, partially mediated by regulatory T cells in a TGFbeta- and IL-10-dependent manner. J Hepatol 2013; 58: 977–983.
Seo N, Tokura Y, Takigawa M, Egawa K . Depletion of IL-10- and TGF-beta-producing regulatory gamma delta T cells by administering a daunomycin-conjugated specific monoclonal antibody in early tumor lesions augments the activity of CTLs and NK cells. J Immunol 1999; 163: 242–249.
Ye J, Ma C, Hsueh EC, Eickhoff CS, Zhang Y, Varvares MA et al. Tumor-derived gammadelta regulatory T cells suppress innate and adaptive immunity through the induction of immunosenescence. J Immunol 2013; 190: 2403–2414.
Catellani S, Poggi A, Bruzzone A, Dadati P, Ravetti JL, Gobbi M et al. Expansion of Vdelta1 T lymphocytes producing IL-4 in low-grade non-Hodgkin lymphomas expressing UL-16-binding proteins. Blood 2007; 109: 2078–2085.
Kang N, Zhou J, Zhang T, Wang L, Lu F, Cui Y et al. Adoptive immunotherapy of lung cancer with immobilized anti-TCRgammadelta antibody-expanded human gammadelta T-cells in peripheral blood. Cancer Biol Ther 2009; 8: 1540–1549.
Li X, Kang N, Zhang X, Dong X, Wei W, Cui L et al. Generation of human regulatory gammadelta T cells by TCRgammadelta stimulation in the presence of TGF-beta and their involvement in the pathogenesis of systemic lupus erythematosus. J Immunol 2011; 186: 6693–6700.
Kabelitz D, Wesch D, Pitters E, Zoller M . Characterization of tumor reactivity of human V gamma 9V delta 2 gamma delta T cells in vitro and in SCID mice in vivo. J Immunol 2004; 173: 6767–6776.
Kong Y, Cao W, Xi X, Ma C, Cui L, He W . The NKG2D ligand ULBP4 binds to TCRgamma9/delta2 and induces cytotoxicity to tumor cells through both TCRgammadelta and NKG2D. Blood 2009; 114: 310–317.
Dai Y, Chen H, Mo C, Cui L, He W . Ectopically-expressed human tumor biomarker MutS homologue 2 is a novel endogenous ligand that is recognized by human gammadelta T cells to induce innate anti-tumor/virus immunity. J Biol Chem 2012; 287: 16812–16819.
Lanca T, Correia DV, Moita CF, Raquel H, Neves-Costa A, Ferreira C et al. The MHC class Ib protein ULBP1 is a nonredundant determinant of leukemia/lymphoma susceptibility to gammadelta T-cell cytotoxicity. Blood 2010; 115: 2407–2411.
Rincon-Orozco B, Kunzmann V, Wrobel P, Kabelitz D, Steinle A, Herrmann T . Activation of V gamma 9V delta 2 T cells by NKG2D. J Immunol 2005; 175: 2144–2151.
Girardi M, Oppenheim DE, Steele CR, Lewis JM, Glusac E, Filler R et al. Regulation of cutaneous malignancy by gammadelta T cells. Science 2001; 294: 605–609.
Das H, Groh V, Kuijl C, Sugita M, Morita CT, Spies T et al. MICA engagement by human Vgamma2Vdelta2 T cells enhances their antigen-dependent effector function. Immunity 2001; 15: 83–93.
Lu J, Aggarwal R, Kanji S, Das M, Joseph M, Pompili V et al. Human ovarian tumor cells escape gammadelta T cell recognition partly by down regulating surface expression of MICA and limiting cell cycle related molecules. PLoS One 2011; 6: e23348.
Kim YJ, Han MK, Broxmeyer HE . 4-1BB regulates NKG2D costimulation in human cord blood CD8+ T cells. Blood 2008; 111: 1378–1386.
Brady J, Carotta S, Thong RP, Chan CJ, Hayakawa Y, Smyth MJ et al. The interactions of multiple cytokines control NK cell maturation. J Immunol 2010; 185: 6679–6688.
Ventre E, Brinza L, Schicklin S, Mafille J, Coupet CA, Marcais A et al. Negative regulation of NKG2D expression by IL-4 in memory CD8 T cells. J Immunol 2012; 189: 3480–3489.
He W, Hao J, Dong S, Gao Y, Tao J, Chi H et al. Naturally activated V gamma 4 gamma delta T cells play a protective role in tumor immunity through expression of eomesodermin. J Immunol 2010; 185: 126–133.
Dokouhaki P, Han M, Joe B, Li M, Johnston MR, Tsao MS et al. Adoptive immunotherapy of cancer using ex vivo expanded human gammadelta T cells: a new approach. Cancer Lett 2010; 297: 126–136.
Siegers GM, Dhamko H, Wang XH, Mathieson AM, Kosaka Y, Felizardo TC et al. Human Vdelta1 gammadelta T cells expanded from peripheral blood exhibit specific cytotoxicity against B-cell chronic lymphocytic leukemia-derived cells. Cytotherapy 2011; 13: 753–764.
Siegers GM, Ribot EJ, Keating A, Foster PJ . Extensive expansion of primary human gamma delta T cells generates cytotoxic effector memory cells that can be labeled with Feraheme for cellular MRI. Cancer Immunol Immunother 2013; 62: 571–583.
Schilbach K, Frommer K, Meier S, Handgretinger R, Eyrich M . Immune response of human propagated gammadelta-T-cells to neuroblastoma recommend the Vdelta1+ subset for gammadelta-T-cell-based immunotherapy. J Immunother 2008; 31: 896–905.
Grivennikov SI, Greten FR, Karin M . Immunity, inflammation, and cancer. Cell 2010; 140: 883–899.
Acknowledgements
We thank Dr Mingxiao He (Duke University for Immunology Research) for critical reading of this manuscript. This work was supported by the Research Special Fund for Public Welfare Industry of Health (Grant Nos. 201202004 and 201302018), the National Program for Key Basic Research Projects, Ministry of Science and Technology, China (2013CB530503 and 2014CB542103), the Beijing Natural Science Foundation (Grant No. 5122031), the National Natural Science Foundation of China (Grant No. 30972776) and the National High Technology Research and Development Program (863 Program) (Grant No. 2007AA021109). We thank Dr Austin Cape for careful reading and feedback.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Mao, Y., Yin, S., Zhang, J. et al. A new effect of IL-4 on human γδ T cells: promoting regulatory Vδ1 T cells via IL-10 production and inhibiting function of Vδ2 T cells. Cell Mol Immunol 13, 217–228 (2016). https://doi.org/10.1038/cmi.2015.07
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2015.07
Keywords
This article is cited by
-
Gamma delta T-cell-based immune checkpoint therapy: attractive candidate for antitumor treatment
Molecular Cancer (2023)
-
The emerging roles of γδ T cells in cancer immunotherapy
Nature Reviews Clinical Oncology (2023)
-
Innate lymphoid cells and innate-like T cells in cancer — at the crossroads of innate and adaptive immunity
Nature Reviews Cancer (2023)
-
Anti-PD-1 antibody armored γδ T cells enhance anti-tumor efficacy in ovarian cancer
Signal Transduction and Targeted Therapy (2023)
-
γδ T cells: origin and fate, subsets, diseases and immunotherapy
Signal Transduction and Targeted Therapy (2023)