Abstract
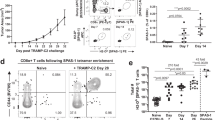
How tumor-infiltrating lymphocytes (TILs) that are tumor-specific but functionally tolerant persist in the antigen-expressing tumor tissue is largely unknown. We have previously developed a modified TRansgenic Adenocarcinoma of the Mouse Prostate (TRAMP) model where prostate cancer cells express the T-cell epitope SIYRYYGL (SIY) recognized by CD8 T cells expressing the 2C T-cell receptor (TCR) (referred to as TRP-SIY mice). In TRP-SIY mice, activated 2C T cells rapidly become tolerant following infiltration into the prostate tumor. In this study, we show that tolerant 2C T cells persist in the prostate tumor of TRP-SIY mice by proliferating slowly in a tumor-dependent, but antigen-, interleukin (IL)-7- and IL-15-independent manner. We also show that disappearance of 2C T cells from the lymphoid organs of TRP-SIY mice are due to antigen-induced T-cell contraction rather than altered trafficking or generalized T-cell depletion in the mice. Finally, we show that clonal T cells unreactive to SIY are equally capable of persisting in the prostate tumor. These findings suggest that while functional tolerance of TILs is induced by antigen, persistence of tolerant TILs in the tumor tissue is mediated by a novel mechanism: slow proliferation independent of antigen and homeostatic cytokines. These results also allow CD8 T-cell survival in the tumor environment to be compared with T-cell survival in chronic infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Husby G, Hoagland PM, Strickland RG, Williams RC Jr . Tissue T and B cell infiltration of primary and metastatic cancer. J Clin Invest 1976; 57: 1471–1482.
von Hanwehr RI, Hofman FM, Taylor CR, Apuzzo ML . Mononuclear lymphoid populations infiltrating the microenvironment of primary CNS tumors. Characterization of cell subsets with monoclonal antibodies. J Neurosurg 1984; 60: 1138–1147.
Miescher S, Whiteside TL, Carrel S, von Fliedner V . Functional properties of tumor-infiltrating and blood lymphocytes in patients with solid tumors: effects of tumor cells and their supernatants on proliferative responses of lymphocytes. J Immunol 1986; 136: 1899–1907.
Whitford P, Mallon EA, George WD, Campbell AM . Flow cytometric analysis of tumour infiltrating lymphocytes in breast cancer. Br J Cancer 1990; 62: 971–975.
Ademmer K, Ebert M, Muller-Ostermeyer F, Friess H, Buchler MW, Schubert W et al. Effector T lymphocyte subsets in human pancreatic cancer: detection of CD8+CD18+ cells and CD8+CD103+ cells by multi-epitope imaging. Clin Exp Immunol 1998; 112: 21–26.
Boon T, Coulie PG, van den Eynde BJ, van der Bruggen P . Human T cell responses against melanoma. Annu Rev Immunol 2006; 24: 175–208.
Zippelius A, Batard P, Rubio-Godoy V, Bioley G, Lienard D, Lejeune F et al. Effector function of human tumor-specific CD8 T cells in melanoma lesions: a state of local functional tolerance. Cancer Res 2004; 64: 2865–2873.
Harlin H, Kuna TV, Peterson AC, Meng Y, Gajewski TF . Tumor progression despite massive influx of activated CD8+ T cells in a patient with malignant melanoma ascites. Cancer Immunol Immunother 2006; 55: 1185–1197.
Bronte V, Kasic T, Gri G, Gallana K, Borsellino G, Marigo I et al. Boosting antitumor responses of T lymphocytes infiltrating human prostate cancers. J Exp Med 2005; 201: 1257–1268.
Greenberg NM, DeMayo F, Finegold MJ, Medina D, Tilley WD, Aspinall JO et al. Prostate cancer in a transgenic mouse. Proc Natl Acad Sci USA 1995; 92: 3439–3443.
Anderson MJ, Shafer-Weaver K, Greenberg NM, Hurwitz AA . Tolerization of tumor-specific T cells despite efficient initial priming in a primary murine model of prostate cancer. J Immunol 2007; 178: 1268–1276.
Bai A, Higham E, Eisen HN, Wittrup KD, Chen J . Rapid tolerization of virus-activated tumor-specific CD8+ T cells in prostate tumors of TRAMP mice. Proc Natl Acad Sci USA 2008; 105: 13003–13008.
Shen CH, Ge Q, Talay O, Eisen HN, Garcia-Sastre A, Chen J . Loss of IL-7R and IL-15R expression is associated with disappearance of memory T cells in respiratory tract following influenza infection. J Immunol 2008; 180: 171–178.
Higham EM, Shen CH, Wittrup KD, Chen J . Cutting edge: delay and reversal of T cell tolerance by intratumoral injection of antigen-loaded dendritic cells in an autochthonous tumor model. J Immunol 2010; 184: 5954–5958.
Dudley ME, Wunderlich JR, Yang JC, Sherry RM, Topalian SL, Restifo NP et al. Adoptive cell transfer therapy following non-myeloablative but lymphodepleting chemotherapy for the treatment of patients with refractory metastatic melanoma. J Clin Oncol 2005; 23: 2346–2357.
Dudley ME, Yang JC, Sherry R, Hughes MS, Royal R, Kammula U et al. Adoptive cell therapy for patients with metastatic melanoma: evaluation of intensive myeloablative chemoradiation preparative regimens. J Clin Oncol 2008; 26: 5233–5239.
Kondratiev S, Sabo E, Yakirevich E, Lavie O, Resnick MB . Intratumoral CD8+ T lymphocytes as a prognostic factor of survival in endometrial carcinoma. Clin Cancer Res 2004; 10: 4450–4456.
Thomas DL, Kim M, Bowerman NA, Narayanan S, Kranz DM, Schreiber H et al. Recurrence of intracranial tumors following adoptive T cell therapy can be prevented by direct and indirect killing aided by high levels of tumor antigen cross-presented on stromal cells. J Immunol 2009; 183: 1828–1837.
Ohlen C, Kalos M, Cheng LE, Shur AC, Hong DJ, Carson BD et al. CD8+ T cell tolerance to a tumor-associated antigen is maintained at the level of expansion rather than effector function. J Exp Med 2002; 195: 1407–1418.
Tanchot C, Lemonnier FA, Perarnau B, Freitas AA, Rocha B . Differential requirements for survival and proliferation of CD8 naive or memory T cells. Science 1997; 276: 2057–2062.
Nesic D, Vukmanovic S . MHC class I is required for peripheral accumulation of CD8+ thymic emigrants. J Immunol 1998; 160: 3705–3712.
Murali-Krishna K, Lau LL, Sambhara S, Lemonnier F, Altman J, Ahmed R . Persistence of memory CD8 T cells in MHC class I-deficient mice. Science 1999; 286: 1377–1381.
Schluns KS, Kieper WC, Jameson SC, Lefrancois L . Interleukin-7 mediates the homeostasis of naive and memory CD8 T cells in vivo. Nat Immunol 2000; 1: 426–432.
Tan JT, Dudl E, LeRoy E, Murray R, Sprent J, Weinberg KI et al. IL-7 is critical for homeostatic proliferation and survival of naive T cells. Proc Natl Acad Sci USA 2001; 98: 8732–8737.
Lodolce JP, Boone DL, Chai S, Swain RE, Dassopoulos T, Trettin S et al. IL-15 receptor maintains lymphoid homeostasis by supporting lymphocyte homing and proliferation. Immunity 1998; 9: 669–676.
Kennedy MK, Glaccum M, Brown SN, Butz EA, Viney JL, Embers M et al. Reversible defects in natural killer and memory CD8 T cell lineages in interleukin 15-deficient mice. J Exp Med 2000; 191: 771–780.
Zhang X, Sun S, Hwang I, Tough DF, Sprent J . Potent and selective stimulation of memory-phenotype CD8+ T cells in vivo by IL-15. Immunity 1998; 8: 591–599.
Ku CC, Murakami M, Sakamoto A, Kappler J, Marrack P . Control of homeostasis of CD8+ memory T cells by opposing cytokines. Science 2000; 288: 675–678.
Virgin HW, Wherry EJ, Ahmed R . Redefining chronic viral infection. Cell 2009; 138: 30–50.
Shin H, Blackburn SD, Blattman JN, Wherry EJ . Viral antigen and extensive division maintain virus-specific CD8 T cells during chronic infection. J Exp Med 2007; 204: 941–949.
Fodor E, Devenish L, Engelhardt OG, Palese P, Brownlee GG, Garcia-Sastre A . Rescue of influenza A virus from recombinant DNA. J Virol 1999; 73: 9679–9682.
Stevenson PG, Efstathiou S, Doherty PC, Lehner PJ . Inhibition of MHC class I-restricted antigen presentation by gamma 2-herpesviruses. Proc Natl Acad Sci USA 2000; 97: 8455–8460.
Hurwitz AA, Foster BA, Allison JP, Greenberg NM, Kwon ED . The TRAMP Mouse as a Model for Prostate Cancer. Current Protocols in Immunology. New York John Wiley & Sons, Inc. 2001: 20.5.1–20.5.23.
Shen CH, Talay O, Mahajan VS, Leskov IB, Eisen HN, Chen J . Antigen-bearing dendritic cells regulate the diverse pattern of memory CD8 T-cell development in different tissues. Proc Natl Acad Sci USA 2010; 107: 22587–22592.
Wherry EJ, Ahmed R . Memory CD8 T-cell differentiation during viral infection. J Virol 2004; 78: 5535–5545.
Di Carlo E, D'Antuono T, Pompa P, Giuliani R, Rosini S, Stuppia L et al. The lack of epithelial interleukin-7 and BAFF/BLyS gene expression in prostate cancer as a possible mechanism of tumor escape from immunosurveillance. Clin Cancer Res 2009; 15: 2979–2987.
Bromley SK, Thomas SY, Luster AD . Chemokine receptor CCR7 guides T cell exit from peripheral tissues and entry into afferent lymphatics. Nat Immunol 2005; 6: 895–901.
Gallatin WM, Weissman IL, Butcher EC . A cell-surface molecule involved in organ-specific homing of lymphocytes. 1983. J Immunol 2006; 177: 5–9.
Becker TC, Wherry EJ, Boone D, Murali-Krishna K, Antia R, Ma A et al. Interleukin 15 is required for proliferative renewal of virus-specific memory CD8 T cells. J Exp Med 2002; 195: 1541–1548.
Schluns KS, Williams K, Ma A, Zheng XX, Lefrancois L . Cutting edge: requirement for IL-15 in the generation of primary and memory antigen-specific CD8 T cells. J Immunol 2002; 168: 4827–4831.
Kurts C, Kosaka H, Carbone FR, Miller JF, Heath WR . Class I-restricted cross-presentation of exogenous self-antigens leads to deletion of autoreactive CD8+ T cells. J Exp Med 1997; 186: 239–245.
Hernandez J, Aung S, Redmond WL, Sherman LA . Phenotypic and functional analysis of CD8+ T cells undergoing peripheral deletion in response to cross-presentation of self-antigen. J Exp Med 2001; 194: 707–717.
Wherry EJ, Blattman JN, Murali-Krishna K, van der Most R, Ahmed R . Viral persistence alters CD8 T-cell immunodominance and tissue distribution and results in distinct stages of functional impairment. J Virol 2003; 77: 4911–4927.
Zajac AJ, Blattman JN, Murali-Krishna K, Sourdive DJ, Suresh M, Altman JD et al. Viral immune evasion due to persistence of activated T cells without effector function. J Exp Med 1998; 188: 2205–2213.
Acknowledgements
We thank Dr Paul Matsudaira for input and support for the project and Dr Herman N. Eisen and Dr Eileen M. Higham for carefully reviewing this manuscript, as well as Eileen M. Higham, Oezcan Talay, Ilya Leskov, Kristyn Ferber, Maria F. Fragoso, Carol McKinley and Camille Jusino for reagents and technical assistance. This work was supported by grants from the National Institutes of Health (F31-AI080286 to MO and CA100875 to JC), UNCF-Merck Graduate Research and NSF Graduate Research Fellowships (to MO) and Singapore-MIT Alliance and Koch Research Fund (to JC).
Author information
Authors and Affiliations
Corresponding author
Additional information
Supplementary information accompanies the paper on Cellular & Molecular Immunology's website.
Supplementary information
Rights and permissions
About this article
Cite this article
Olurinde, M., Shen, CH., Drake, A. et al. Persistence of tumor-infiltrating CD8 T cells is tumor-dependent but antigen-independent. Cell Mol Immunol 8, 415–423 (2011). https://doi.org/10.1038/cmi.2011.18
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cmi.2011.18
Keywords
This article is cited by
-
Activated cytotoxic lymphocytes promote tumor progression by increasing the ability of 3LL tumor cells to mediate MDSC chemoattraction via Fas signaling
Cellular & Molecular Immunology (2015)