Abstract
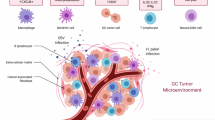
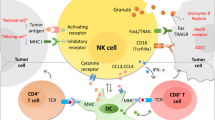
Gastrointestinal stromal tumors (GIST) contain tumor-infiltrating immune cells and their presence provides an opportunity and rationale for developing effective forms of immunotherapy. The types of tumor-infiltrating inflammatory cells and relevant immune checkpoint inhibitors are the focus of active investigation. The most numerous tumor-infiltrating inflammatory cells are tumor-associated macrophages (TAMs) and CD3+ T cells. Studies have shown that patients with GISTs that harbor increased numbers of CD3+ T cells have better outcomes. However, the clinical behavior of GIST has not been shown to correlate with the number of TAMs. The biological significance of other less frequent tumor-infiltrating immune cells including tumor-infiltrating neurtrophils (TINs), natural killer cells (NKs), B cells, dendritic cells (DCs) remains unclear. The immune checkpoint inhibitors CTLA-4, PD1/PDL1 and TIM3/galectin-9 are molecules that can be targeted by synthesized antibodies. Clinical and pre-clinical trials using this approach against immune checkpoint inhibitors, anti-KIT antibody and the generation of chimeric antigen receptor (CAR) T-cells have shown promising results. The treatment of GIST with immunotherapy is complex and evolving; this article reviews its current status for patients with GISTs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Barnett CM, Corless CL, Heinrich MC . Gastrointestinal stromal tumors: molecular markers and genetic subtypes. Hematol Oncol Clin North Am 2013; 27: 871–888.
Trent JC, Subramanian MP . Managing GIST in the imatinib era: optimization of adjuvant therapy. Expert Rev Anticancer Ther 2014; 14: 1–15.
Cheng CT, Tsai CY, Yeh CN, Chiang KC, Chen YY, Wang SY et al. Clinical significance of pathological complete response in patients with metastatic gastrointestinal stromal tumors after imatinib mesylate treatment - lessons learned. Anticancer Res 2014; 34: 6617–6625.
Vadakara J, von Mehren M . Gastrointestinal stromal tumors: management of metastatic disease and emerging therapies. Hematol Oncol Clin North Am 2013; 27: 905–920.
Tan Y, Garcia-Buitrago MT, Trent JC, Rosenberg AE . The immune system and gastrointestinal stromal tumor: a wealth of opportunities. Curr Opin Oncol 2015; 27: 338–342.
van Dongen M, Savage ND, Jordanova ES, Briaire-de Bruijn IH, Walburg KV, Ottenhoff TH et al. Anti-inflammatory M2 type macrophages characterize metastasized and tyrosine kinase inhibitor-treated gastrointestinal stromaltumors. Int J Cancer 2010; 127: 899–909.
Cameron S, Gieselmann M, Blaschke M, Ramadori G, Fuzesi L . Immune cells in primary and metastatic gastrointestinal stromal tumors (GIST). Int J Clin Exp Pathol 2014; 7: 3563–3579.
Rusakiewicz S, Semeraro M, Sarabi M, Desbois M, Locher C, Mendez R et al. Immune infiltrates are prognostic factors in localized gastrointestinal stromaltumors. Cancer Res 2013; 73: 3499–3510.
Delahaye NF, Rusakiewicz S, Martins I, Menard C, Roux S, Lyonnet L et al. Alternatively spliced NKp30 isoforms affect the prognosis of gastrointestinal stromal tumors. Nat Med 2011; 17: 700–U91.
Balachandran VP, Cavnar MJ, Zeng S, Bamboat ZM, Ocuin LM, Obaid H et al. Imatinib potentiates antitumor T cell responses in gastrointestinal stromal tumor through the inhibition of Ido. Nat Med 2011; 17: 1094–1100.
Prendergast GC, Smith C, Thomas S, Mandik-Nayak L, Laury-Kleintop L, Metz R et al. Indoleamine 2,3-dioxygenase pathways of pathogenic inflammation and immune escape in cancer. Cancer Immunol Immunother 2014; 63: 721–735.
Burgess M, Tawbi H . Immunotherapeutic approaches to sarcoma. Curr Treat Options Oncol 2015; 16: 26.
Shoushtari AN, Van Tine BA, Schwartz GK . Novel treatment targets in sarcoma: more than just the GIST. Am Soc Clin Oncol Educ Book. 2014; 34: e488–e495.
Bertucci F, Finetti P, Mamessier E, Pantaleo MA, Astolfi A, Ostrowski J et al. PDL1 expression is an independent prognostic factor in localized GIST. Oncoimmunology 2015; 4: e1002729.
Seifert AM, Zeng S, Zhang JQ, Kim TS, Cohen NA, Beckman MJ et al. PD-1/PD-L1 blockade enhances T cell activity and antitumor efficacy of imatinib in gastrointestinal stromal tumors. Clin Cancer Res 2016 (e-pub ahead of print).
da Silva IP, Gallois A, Jimenez-Baranda S, Khan S, Anderson AC, Kuchroo VK et al. Reversal of NK-cell exhaustion in advanced melanoma by Tim-3 blockade. Cancer Immunol Res 2014; 2: 410–422.
Komita H, Koido S, Hayashi K, Kan S, Ito M, Kamata Y et al. Expression of immune checkpoint molecules of T cell immunoglobulin and mucin protein 3/galectin-9 for NK cell suppression in human gastrointestinal stromal tumors. Oncol Rep 2015; 34: 2099–2105.
Chen LL, Chen X, Choi H, Sang H, Chen LC, Zhang H et al. Exploiting antitumor immunity to overcome relapse and improve remission duration. Cancer Immunol Immunother 2012; 61: 1113–1124.
Edris B, Willingham SB, Weiskopf K, Volkmer AK, Volkmer JP, Muhlenberg T et al. Anti-KIT monoclonal antibody inhibits imatinib-resistant gastrointestinal stromal tumor growth. Proc Natl Acad Sci USA 2013; 110: 3501–3506.
Katz SC, Burga RA, Naheed S, Licata LA, Thorn M, Osgood D et al. Anti-KIT designer T cells for the treatment of gastrointestinal stromal tumor. J Transl Med 2013; 11: 46.
Perez D, Hauswirth F, Jager D, Metzger U, Samartzis EP, Went P et al. Protein expression of cancer testis antigens predicts tumor recurrence and treatment response to imatinib in gastrointestinal stromal tumors. Int J Cancer 2011; 128: 2947–2952.
Ghadban T, Perez DR, Vashist YK, Bockhorn M, Koenig AM, El Gammal AT et al. Expression of cancer testis antigens CT10 (MAGE-C2) and GAGE in gastrointestinal stromal tumors. Eur J Surg Oncol 2014; 40: 1307–1312.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Tan, Y., Trent, J., Wilky, B. et al. Current status of immunotherapy for gastrointestinal stromal tumor. Cancer Gene Ther 24, 130–133 (2017). https://doi.org/10.1038/cgt.2016.58
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2016.58
This article is cited by
-
Advances in immunology and immunotherapy for mesenchymal gastrointestinal cancers
Molecular Cancer (2023)
-
Immune Cell Infiltration and the Expression of PD-1 and PD-L1 in Primary PDGFRA-Mutant Gastrointestinal Stromal Tumors
Journal of Gastrointestinal Surgery (2021)
-
Translating Knowledge About the Immune Microenvironment of Gastrointestinal Stromal Tumors into Effective Clinical Strategies
Current Treatment Options in Oncology (2021)
-
Gastrointestinal stromal tumor: a review of current and emerging therapies
Cancer and Metastasis Reviews (2021)
-
Clinicopathological implications of TIM3+ tumor-infiltrating lymphocytes and the miR-455-5p/Galectin-9 axis in skull base chordoma patients
Cancer Immunology, Immunotherapy (2019)