Abstract
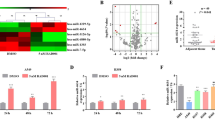
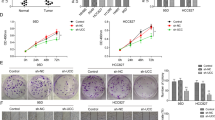
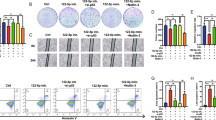
Preliminary studies showed that miR-21 is overexpressed in some human cancers. However, the role of miR-21 in cancer is still unclear and even controversial. Our purpose was to investigate the biological effects of miR-21 on A549 non-small cell lung cancer (NSCLC) cells and the underlying mechanisms of those effects. The expression of miR-21 was quantified in serum samples from patients with NSCLC. A549 cells were transfected with miR-NC-sponge or miR-21-sponge only, or with miR-21-sponge plus PDCD4 small-interfering RNA (siRNA). The expression of miR-21 and PDCD4 mRNA in transfected cells was quantified by real-time polymerase chain reaction and the expression of PDCD4 protein by Western blot. Cell proliferation, apoptosis, migration, and invasion assays were performed to determine the biological effects of miR-21 expression and PDCD4 inhibition. miR-21 was overexpressed in serum from patients with NSCLC. Reduced miR-21 expression was observed following transfection with miR-21-sponge in A549 NSCLC cells. Co-transfection of miR-21-sponge with PDCD4 siRNA upregulated miR-21 expression in these cells. PDCD4 mRNA and protein levels were increased 2.14-fold and 2.16-fold, respectively, following inhibition of miR-21 expression. Inhibition of miR-21 expression following transfection of miR-21-sponge reduced cell proliferation, migration, and invasion of A549 cells. Depletion of PDCD4 by siRNA transfection reversed the reduction of cell proliferation, migration, and invasion induced by inhibition of miR-21 in A549 cells. It indicates that miR-21 is highly expressed in patients with NSCLC and inhibition of miR-21 expression reduces proliferation, migration, and invasion of A549 cells by upregulating PDCD4 expression. Modulation of miR-21 or PDCD4 expression may provide a potentially novel therapeutic approach for NSCLC.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Siegel R, Naishadham D, Jemal A . Cancer statistics, 2013. CA: a cancer journal for clinicians 2013; 63: 11–30.
Wood AJ, Spira A, Ettinger DS . Multidisciplinary management of lung cancer. New England Journal of Medicine 2004; 350: 379–392.
Ponn RB, Lo Cicero J III, Daly BD . Surgical treatment of non-small cell lung cancer. General thoracic surgery 2005; 6: 1548–1587.
Soon YY, Stockler MR, Askie LM, Boyer MJ . Duration of chemotherapy for advanced non-small-cell lung cancer: a systematic review and meta-analysis of randomized trials. Journal of clinical oncology: official journal of the American Society of Clinical Oncology 2009; 27: 3277–3283.
Almeida MI, Reis RM, Calin GA . MicroRNA history: discovery, recent applications, and next frontiers. Mutation research 2011; 717: 1–8.
Munker R, Calin GA . MicroRNA profiling in cancer. Clinical science 2011; 121: 141–158.
Krol J, Loedige I, Filipowicz W . The widespread regulation of microRNA biogenesis, function and decay. Nature Reviews Genetics 2010; 11: 597–610.
Garzon R, Calin GA, Croce CM . MicroRNAs in cancer. Annual review of medicine 2009; 60: 167–179.
Cortez MA, Bueso-Ramos C, Ferdin J, Lopez-Berestein G, Sood AK, Calin GA . MicroRNAs in body fluids—the mix of hormones and biomarkers. Nature reviews. Clinical oncology 2011; 8: 467–477.
Di Leva G, Croce CM . Roles of small RNAs in tumor formation. Trends in molecular medicine 2010; 16: 257–267.
Hu Z, Chen X, Zhao Y, Tian T, Jin G, Shu Y et al. Serum MicroRNA signatures identified in a genome-wide serum MicroRNA expression profiling predict survival of non–small-cell lung cancer. Journal of Clinical Oncology 2010; 28: 1721–1726.
Volinia S, Calin GA, Liu C-G, Ambs S, Cimmino A, Petrocca F et al. A microRNA expression signature of human solid tumors defines cancer gene targets. Proceedings of the National Academy of Sciences of the United States of America 2006; 103: 2257–2261.
Krichevsky AM, Gabriely G . miR-21: a small multi-faceted RNA. Journal of cellular and molecular medicine 2009; 13: 39–53.
Ribas J, Ni X, Haffner M, Wentzel EA, Salmasi AH, Chowdhury WH et al. miR-21: an androgen receptor-regulated microRNA that promotes hormone-dependent and hormone-independent prostate cancer growth. Cancer research 2009; 69: 7165–7169.
Darido C, Georgy SR, Wilanowski T, Dworkin S, Auden A, Zhao Q et al. Targeting of the tumor suppressor GRHL3 by a miR-21-dependent proto-oncogenic network results in PTEN loss and tumorigenesis. Cancer cell 2011; 20: 635–648.
Folini M, Gandellini P, Longoni N, Profumo V, Callari M, Pennati M et al. miR-21: an oncomir on strike in prostate cancer. Molecular cancer 2010; 9: 12.
Lankat‐Buttgereit B, Göke R . The tumour suppressor Pdcd4: recent advances in the elucidation of function and regulation. Biology of the Cell 2009; 101: 309–317.
McDonald JS, Milosevic D, Reddi HV, Grebe SK, Algeciras-Schimnich A . Analysis of circulating microRNA: preanalytical and analytical challenges. Clin Chem 2011; 57: 833–840.
Ebert MS, Neilson JR, Sharp PA . MicroRNA sponges: competitive inhibitors of small RNAs in mammalian cells. Nature methods 2007; 4: 721–726.
Lu J, Ji J, Meng H, Wang D, Jiang B, Liu L et al. The protective effect and underlying mechanism of metformin on neointima formation in fructose-induced insulin resistant rats. Cardiovasc Diabetol 2013; 12: 58.
Yu S-L, Chen H-Y, Chang G-C, Chen C-Y, Chen H-W, Singh S et al. MicroRNA signature predicts survival and relapse in lung cancer. Cancer cell 2008; 13: 48–57.
Zhang JG, Wang JJ, Zhao F, Liu Q, Jiang K, Yang GH . MicroRNA-21 (miR-21) represses tumor suppressor PTEN and promotes growth and invasion in non-small cell lung cancer (NSCLC). Clinica chimica acta; international journal of clinical chemistry 2010; 411: 846–852.
Liu Z-L, Wang H, Liu J, Wang Z-X . MicroRNA-21 (miR-21) expression promotes growth, metastasis, and chemo-or radioresistance in non-small cell lung cancer cells by targeting PTEN. Molecular and cellular biochemistry 2013; 372: 35–45.
Volinia S, Galasso M, Costinean S, Tagliavini L, Gamberoni G, Drusco A et al. Reprogramming of miRNA networks in cancer and leukemia. Genome research 2010; 20: 589–599.
Wang Q, Zhang Y, Yang HS . Pdcd4 knockdown up-regulates MAP4K1 expression and activation of AP-1 dependent transcription through c-Myc. Biochimica et biophysica acta 2012; 1823: 1807–1814.
Bitomsky N, Bohm M, Klempnauer KH . Transformation suppressor protein Pdcd4 interferes with JNK-mediated phosphorylation of c-Jun and recruitment of the coactivator p300 by c-Jun. Oncogene 2004; 23: 7484–7493.
Ma X, Kumar M, Choudhury SN, Buscaglia LEB, Barker JR, Kanakamedala K et al. Loss of the miR-21 allele elevates the expression of its target genes and reduces tumorigenesis. Proceedings of the National Academy of Sciences 2011; 108: 10144–10149.
Chang JH, Cho YH, Sohn SY, Choi JM, Kim A, Kim YC et al. Crystal structure of the eIF4A–PDCD4 complex. Proceedings of the National Academy of Sciences 2009; 106: 3148–3153.
Asangani IA, Rasheed SA, Nikolova DA, Leupold JH, Colburn NH, Post S et al. MicroRNA-21 (miR-21) post-transcriptionally downregulates tumor suppressor Pdcd4 and stimulates invasion, intravasation and metastasis in colorectal cancer. Oncogene 2008; 27: 2128–2136.
Gaur AB, Holbeck SL, Colburn NH, Israel MA . Downregulation of Pdcd4 by mir-21 facilitates glioblastoma proliferation in vivo. Neuro-oncology 2011; 13: 580–590.
Hatley ME, Patrick DM, Garcia MR, Richardson JA, Bassel-Duby R, van Rooij E et al. Modulation of K-Ras-dependent lung tumorigenesis by MicroRNA-21. Cancer Cell 2010; 18: 282–293.
Leupold JH, Yang HS, Colburn NH, Asangani I, Post S, Allgayer H . Tumor suppressor Pdcd4 inhibits invasion/intravasation and regulates urokinase receptor (u-PAR) gene expression via Sp-transcription factors. Oncogene 2007; 26: 4550–4562.
Jung E-J, Calin GA . The meaning of 21 in the microRNA world: perfection rather than destruction? Cancer cell 2010; 18: 203–205.
Acknowledgements
This study was supported by grants from the National Natural Science Foundation of China (81170257) and Zhejiang Provincial Natural Science Foundation (Y2110513).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Rights and permissions
About this article
Cite this article
Yang, Y., Meng, H., Peng, Q. et al. Downregulation of microRNA-21 expression restrains non-small cell lung cancer cell proliferation and migration through upregulation of programmed cell death 4. Cancer Gene Ther 22, 23–29 (2015). https://doi.org/10.1038/cgt.2014.66
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2014.66
This article is cited by
-
The emerging role of MicroRNA-182 in tumorigenesis; a promising therapeutic target
Cancer Cell International (2023)
-
KLF5-trancripted miR-125b-5p is involved in enhancing the radio-sensitivity of breast cancer cells by targeting BRCA1
Molecular & Cellular Toxicology (2022)
-
miR-21 regulates immunosuppression mediated by myeloid-derived suppressor cells by impairing RUNX1-YAP interaction in lung cancer
Cancer Cell International (2020)
-
Application of magnetic nanoparticles in nucleic acid detection
Journal of Nanobiotechnology (2020)
-
miR-21a in exosomes from Lewis lung carcinoma cells accelerates tumor growth through targeting PDCD4 to enhance expansion of myeloid-derived suppressor cells
Oncogene (2020)