Abstract
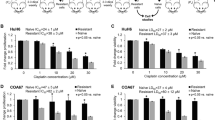
A tumor-selective non-lytic retroviral replicating vector (RRV), Toca 511, and an extended-release formulation of 5-fluorocytosine (5-FC), Toca FC, are currently being evaluated in clinical trials in patients with recurrent high-grade glioma (NCT01156584, NCT01470794 and NCT01985256). Tumor-selective propagation of this RRV enables highly efficient transduction of glioma cells with cytosine deaminase (CD), which serves as a prodrug activator for conversion of the anti-fungal prodrug 5-FC to the anti-cancer drug 5-fluorouracil (5-FU) directly within the infected cells. We investigated whether, in addition to its direct cytotoxic effects, 5-FU generated intracellularly by RRV-mediated CD/5-FC prodrug activator gene therapy could also act as a radiosensitizing agent. Efficient transduction by RRV and expression of CD were confirmed in the highly aggressive, radioresistant human glioblastoma cell line U87EGFRvIII and its parental cell line U87MG (U87). RRV-transduced cells showed significant radiosensitization even after transient exposure to 5-FC. This was confirmed both in vitro by a clonogenic colony survival assay and in vivo by bioluminescence imaging analysis. These results provide a convincing rationale for development of tumor-targeted radiosensitization strategies utilizing the tumor-selective replicative capability of RRV, and incorporation of radiation therapy into future clinical trials evaluating Toca 511 and Toca FC in brain tumor patients.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Rainov NG, Ren H . Clinical trials with retrovirus mediated gene therapy—what have we learned? J Neurooncol 2003; 65: 227–236.
Hiraoka K, Kimura T, Logg CR, Kasahara N . Tumor-selective gene expression in a hepatic metastasis model after locoregional delivery of a replication-competent retrovirus vector. Clin Cancer Res 2006; 12: 7108–7116.
Hiraoka K, Kimura T, Logg CR, Tai CK, Haga K, Lawson GW et al. Therapeutic efficacy of replication-competent retrovirus vector-mediated suicide gene therapy in a multifocal colorectal cancer metastasis model. Cancer Res 2007; 67: 5345–5353.
Kikuchi E, Menendez S, Ozu C, Ohori M, Cordon-Cardo C, Logg CR et al. Highly efficient gene delivery for bladder cancers by intravesically administered replication-competent retroviral vectors. Clin Cancer Res 2007; 13 (15 Pt 1): 4511–4518.
Kikuchi E, Menendez S, Ozu C, Ohori M, Cordon-Cardo C, Logg CR et al. Delivery of replication-competent retrovirus expressing Escherichia coli purine nucleoside phosphorylase increases the metabolism of the prodrug, fludarabine phosphate and suppresses the growth of bladder tumor xenografts. Cancer Gene Ther 2007; 14: 279–286.
Logg CR, Tai CK, Logg A, Anderson WF, Kasahara N . A uniquely stable replication-competent retrovirus vector achieves efficient gene delivery in vitro and in solid tumors. Hum Gene Ther 2001; 12: 921–932.
Tai CK, Wang W, Lai YH, Logg CR, Parker WB, Li YF et al. Enhanced efficiency of prodrug activation therapy by tumor-selective replicating retrovirus vectors armed with the Escherichia coli purine nucleoside phosphorylase gene. Cancer Gene Ther 2010; 17: 614–623.
Tai CK, Wang WJ, Chen TC, Kasahara N . Single-shot, multicycle suicide gene therapy by replication-competent retrovirus vectors achieves long-term survival benefit in experimental glioma. Mol Ther 2005; 12: 842–851.
Wang W, Tai CK, Kershaw AD, Solly SK, Klatzmann D, Kasahara N et al. Use of replication-competent retroviral vectors in an immunocompetent intracranial glioma model. Neurosurg Focus 2006; 20: E25.
Wang WJ, Tai CK, Kasahara N, Chen TC . Highly efficient and tumor-restricted gene transfer to malignant gliomas by replication-competent retroviral vectors. Hum Gene Ther 2003; 14: 117–127.
Hlavaty J, Jandl G, Liszt M, Petznek H, Konig-Schuster M, Sedlak J et al. Comparative evaluation of preclinical in vivo models for the assessment of replicating retroviral vectors for the treatment of glioblastoma. J Neurooncol 2011; 102: 59–69.
Ostertag D, Amundson KK, Lopez Espinoza F, Martin B, Buckley T, Galvao da Silva AP et al. Brain tumor eradication and prolonged survival from intratumoral conversion of 5-fluorocytosine to 5-fluorouracil using a nonlytic retroviral replicating vector. Neuro Oncol 2012; 14: 145–159.
Solly SK, Trajcevski S, Frisen C, Holzer GW, Nelson E, Clerc B et al. Replicative retroviral vectors for cancer gene therapy. Cancer Gene Ther 2003; 10: 30–39.
Perez OD, Logg CR, Hiraoka K, Diago O, Burnett R, Inagaki A et al. Design and selection of Toca 511 for clinical use: modified retroviral replicating vector with improved stability and gene expression. Mol Ther 2012; 20: 1689–1698.
McGinn CJ, Lawrence TS . Recent advances in the use of radiosensitizing nucleosides. Semin Radiat Oncol 2001; 11: 270–280.
Logg CR, Kasahara N . Retrovirus-mediated gene transfer to tumors: utilizing the replicative power of viruses to achieve highly efficient tumor transduction in vivo. Methods Mol Biol 2004; 246: 499–525.
Stripecke R, Koya RC, Ta HQ, Kasahara N, Levine AM . The use of lentiviral vectors in gene therapy of leukemia: combinatorial gene delivery of immunomodulators into leukemia cells by state-of-the-art vectors. Blood Cells Mol Dis 2003; 31: 28–37.
Wang MY, Lu KV, Zhu S, Dia EQ, Vivanco I, Shackleford GM et al. Mammalian target of rapamycin inhibition promotes response to epidermal growth factor receptor kinase inhibitors in PTEN-deficient and PTEN-intact glioblastoma cells. Cancer Res 2006; 66: 7864–7869.
Logg CR, Robbins JM, Jolly DJ, Gruber HE, Kasahara N . Retroviral replicating vectors in cancer. Methods Enzymol 2012; 507: 199–228.
Finch RE, Bending MR, Lant AF . Plasma levels of 5-fluorouracil after oral and intravenous administration in cancer patients. Br J Clin Pharmacol 1979; 7: 613–617.
Vermes A, Guchelaar HJ, Dankert J . Flucytosine: a review of its pharmacology, clinical indications, pharmacokinetics, toxicity and drug interactions. J Antimicrob Chemother 2000; 46: 171–179.
Kim K, Brush JM, Watson PA, Cacalano NA, Iwamoto KS, McBride WH . Epidermal growth factor receptor vIII expression in U87 glioblastoma cells alters their proteasome composition, function, and response to irradiation. Mol Cancer Res 2008; 6: 426–434.
Valdes G, Iwamoto KS . Re-evaluation of cellular radiosensitization by 5-fluorouracil: high-dose, pulsed administration is effective and preferable to conventional low-dose, chronic administration. Int J Radiat Biol 2013; 89: 851–862.
Block ER, Bennett JE . Pharmacological studies with 5-fluorocytosine. Antimicrob Agents Chemother 1972; 1: 476–482.
Kievit E, Bershad E, Ng E, Sethna P, Dev I, Lawrence TS et al. Superiority of yeast over bacterial cytosine deaminase for enzyme/prodrug gene therapy in colon cancer xenografts. Cancer Res 1999; 59: 1417–1421.
Acknowledgements
We thank Dr Christopher R Logg for advice and suggestions, and the UCLA Vector Core & Shared Resource Facility (supported by P30 DK041301 and P30 CA016042) for assistance with lentiviral vector production, and the UCLA Virology Core for assistance with p24 enzyme-linked immunosorbent assay. This work was supported in part by NIH grant no. U01 NS059821 and a research award from Tocagen. This work was also enabled by support to Tocagen from Accelerate Brain Cancer Cure, National Brain Tumor Society, American Brain Tumor Association, Musella Foundation and Voices Against Brain Cancer.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
HEG, JMR and DJJ are employees and/or shareholders of Tocagen. NK is a consultant, has ownership interest and is the recipient of a research grant from Tocagen. The research conducted under this grant award has been reviewed and approved by the UCLA Conflict of Interest Review Committee.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Takahashi, M., Valdes, G., Hiraoka, K. et al. Radiosensitization of gliomas by intracellular generation of 5-fluorouracil potentiates prodrug activator gene therapy with a retroviral replicating vector. Cancer Gene Ther 21, 405–410 (2014). https://doi.org/10.1038/cgt.2014.38
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2014.38
This article is cited by
-
Recent progress in the research of suicide gene therapy for malignant glioma
Neurosurgical Review (2021)
-
Antitumor efficacy of cytosine deaminase-armed vaccinia virus plus 5-fluorocytosine in colorectal cancers
Cancer Cell International (2020)
-
Stable knockdown of CREB, HIF-1 and HIF-2 by replication-competent retroviruses abrogates the responses to hypoxia in hepatocellular carcinoma
Cancer Gene Therapy (2017)
-
Adenoviral-mediated imaging of gene transfer using a somatostatin receptor-cytosine deaminase fusion protein
Cancer Gene Therapy (2015)