Abstract
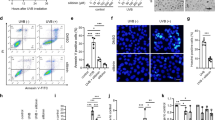
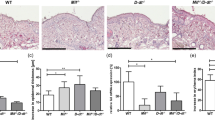
Interferon-gamma (IFN-γ) exhibits biological activities that are considered to have important roles in tumor suppression. Therefore, the IFN-γ gene is a potential candidate for in vivo cytokine gene therapy against skin cancer. The present study evaluated the efficacy of a hydrodynamics-based IFN-γ gene transfection for skin cancer treatment, in which the plasmid DNA encoding IFN-γ was administered into the tail vein of mice following 7,12-dimethylbenz[a]anthracene and 12-O-tetradecanoylphorbol-13-acetate-induced skin carcinogenesis. Serum levels of IFN-γ were substantially elevated without liver toxicity. The mice injected with IFN-γ plasmid DNA displayed a marked reduction in papilloma numbers, suppressed proliferation of epidermal cells and induction of caspase-3-mediated apoptosis. These results suggest that the hydrodynamics-based transfection of IFN-γ plasmid DNA is a convenient and efficient means of skin cancer gene therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Accession codes
References
Hara-Chikuma M, Verkman AS . Prevention of skin tumorigenesis and impairment of epidermal cell proliferation by targeted aquaporin-3 gene disruption. Mol Cell Biol 2008; 28: 326–332.
Lansbury L, Leonardi-Bee J, Perkins W, Goodacre T, Tweed JA, Bath-Hextall FJ . Interventions for non-metastatic squamous cell carcinoma of the skin. Cochrane Database Syst Rev 2010; 4: CD007869.
Cusack Jr JC, Tanabe KK . Cancer gene therapy. Surg Oncol Clin N Am 1998; 7: 421–469.
Chawla-Sarkar M, Lindner DJ, Liu YF, Williams BR, Sen GC, Silverman RH et al. Apoptosis and interferons: role of interferon-stimulated genes as mediators of apoptosis. Apoptosis 2003; 8: 237–249.
Lee SH, Kim JW, Lee HW, Cho YS, Oh SH, Kim YJ et al. Interferon regulatory factor-1 (IRF-1) is a mediator for interferon-gamma induced attenuation of telomerase activity and human telomerase reverse transcriptase (hTERT) expression. Oncogene 2003; 22: 381–391.
Saha B, Jyothi Prasanna S, Chandrasekar B, Nandi D . Gene modulation and immunoregulatory roles of interferon gamma. Cytokine 2010; 50: 1–14.
Kobayashi N, Nishikawa M, Takakura Y . The hydrodynamics-based procedure for controlling the pharmacokinetics of gene medicines at whole body, organ and cellular levels. Adv Drug Deliv Rev 2005; 57: 713–731.
Liu F, Song Y, Liu D . Hydrodynamics-based transfection in animals by systemic administration of plasmid DNA. Gene Ther 1999; 6: 1258–1266.
Zhang G, Budker V, Wolff JA . High levels of foreign gene expression in hepatocytes after tail vein injections of naked plasmid DNA. Hum Gene Ther 1999; 10: 1735–1737.
Kobayashi N, Kuramoto T, Chen S, Watanabe Y, Takakura Y . Therapeutic effect of intravenous interferon gene delivery with naked plasmid DNA in murine metastasis models. Mol Ther 2002; 6: 737–744.
Lui VW, He Y, Falo L, Huang L . Systemic administration of naked DNA encoding interleukin 12 for the treatment of human papillomavirus DNA-positive tumor. Hum Gene Ther 2002; 13: 177–185.
Budker V, Budker T, Zhang G, Subbotin V, Loomis A, Wolff JA . Hypothesis: naked plasmid DNA is taken up by cells in vivo by a receptor-mediated process. J Gene Med 2000; 2: 76–88.
Kobayashi N, Kuramoto T, Yamaoka K, Hashida M, Takakura Y . Hepatic uptake and gene expression mechanisms following intravenous administration of plasmid DNA by conventional and hydrodynamics-based procedures. J Pharmacol Exp Ther 2001; 297: 853–860.
Nomura T, Yasuda K, Yamada T, Okamoto S, Mahato RI, Watanabe Y et al. Gene expression and antitumor effects following direct interferon-gamma gene transfer with naked plasmid DNA and DC-chol liposome complexes in mice. Gene Ther 1999; 6: 121–129.
Zhaorigetu S, Yanaka N, Sasaki M, Watanabe H, Kato N . Silk protein, sericin, suppresses DMBA-TPA-induced mouse skin tumorigenesis by reducing oxidative stress, inflammatory responses and endogenous tumor promoter TNF-alpha. Oncol Rep 2003; 10: 537–543.
Kim J, Shin DM . Biomarkers of squamous cell carcinoma of the head and neck. Histol Histopathol 1997; 12: 205–218.
Burke F, East N, Upton C, Patel K, Balkwill FR . Interferon gamma induces cell cycle arrest and apoptosis in a model of ovarian cancer: enhancement of effect by batimastat. Eur J Cancer 1997; 33: 1114–1121.
Detjen KM, Farwig K, Welzel M, Wiedenmann B, Rosewicz S . Interferon gamma inhibits growth of human pancreatic carcinoma cells via caspase-1 dependent induction of apoptosis. Gut 2001; 49: 251–262.
Das A, Banik NL, Ray SK . Molecular mechanisms of the combination of retinoid and interferon-gamma for inducing differentiation and increasing apoptosis in human glioblastoma T98G and U87MG cells. Neurochem Res 2009; 34: 87–101.
Kane A, Yang I . Interferon-gamma in brain tumor immunotherapy. Neurosurg Clin N Am 2010; 21: 77–86.
Salvesen GS, Riedl SJ . Caspase mechanisms. Adv Exp Med Biol 2008; 615: 13–23.
Dai C, Krantz SB . Interferon gamma induces upregulation and activation of caspases 1, 3, and 8 to produce apoptosis in human erythroid progenitor cells. Blood 1999; 93: 3309–3316.
Rathbun RK, Christianson TA, Faulkner GR, Jones G, Keeble W, O’Dwyer M et al. Interferon-gamma-induced apoptotic responses of Fanconi anemia group C hematopoietic progenitor cells involve caspase 8-dependent activation of caspase 3 family members. Blood 2000; 96: 4204–4211.
Kurzrock R, Rosenblum MG, Sherwin SA, Rios A, Talpaz M, Quesada JR et al. Pharmacokinetics, single-dose tolerance, and biological activity of recombinant gamma-interferon in cancer patients. Cancer Res 1985; 45: 2866–2872.
Acknowledgements
This work was supported by the National Research Foundation of Korea (NRF) grant funded by the Korean government (MEST) Grant No-2009-0071504. The authors acknowledge a graduate fellowship provided by the Korean Ministry of Education and Human Resources Development through the Brain Korea 21 project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Ko, JH., Jung, BG., Park, YS. et al. Inhibitory effects of interferon-gamma plasmid DNA on DMBA-TPA induced mouse skin carcinogenesis. Cancer Gene Ther 18, 646–654 (2011). https://doi.org/10.1038/cgt.2011.36
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2011.36
Keywords
This article is cited by
-
Current status of skin cancers with a focus on immunology and immunotherapy
Cancer Cell International (2023)
-
Novel Fish Oil-based Bigel System for Controlled Drug Delivery and its Influence on Immunomodulatory Activity of Imiquimod Against Skin Cancer
Pharmaceutical Research (2017)
-
PAD2 overexpression in transgenic mice augments malignancy and tumor-associated inflammation in chemically initiated skin tumors
Cell and Tissue Research (2017)