Abstract
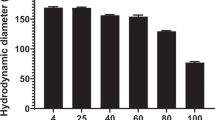
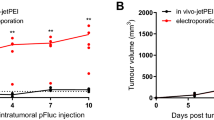
Successfully systemic gene therapy has been hindered by vector-related limitations, including toxicity and inefficient gene delivery to tumor cells after intravenous administration. In this study, we evaluated the potential of spherical polyethylenimine nanogels (M-PEIs) as a novel vector for intravenous delivery of plasmids to tumor cells. M-PEIs provided a sustained release of plasmids up to 14 days and were also effective in protecting plasmids from enzymatic degradation in serum-conditioned media. M-PEIs showed no obvious cytotoxicity to mammalian cells in vitro as well as to liver, heart and kidney in mice after intravenous injection. Importantly, following intravenous administration of M-PEIs/plasmid complexes into human hepatocellular carcinoma xenograft-bearing mice, green fluorescence protein reporter gene expression was predominantly found in the tumor. This study indicates that M-PEIs may be a candidate for systemic delivery of plasmids into tumors.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Smrekar B, Wightman L, Wolschek MF, Lichtenberger C, Ruzicka R, Ogris M . Tissue-dependent factors affect gene delivery to tumors in vivo. Gene Therapy 2003; 10: 1079–1088.
Cohen H, Levy RJ, Gao J, Fishbein I, Kousaev V, Sosnowski S et al. Sustained delivery and expression of DNA encapsulated in polymeric nanoparticles. Gene Therapy 2000; 7: 1896–1905.
Vasir JK, Labhasetwar V . Polymeric nanoparticles for gene delivery. Expert Opin Drug Deliv 2006; 3: 325–344.
Boussif O, Lezoualc'h F, Zanta MA, Mergny MD, Scherman D, Demeneix B et al. A versatile vector for gene and oligonucleotide transfer into cells in culture and in vivo: polyethylenimine. Proc Natl Acad Sci 1995; 92: 7297–7301.
Iwai M, Harada Y, Tanaka S, Muramatsu A, Mori T, Kashima K et al. Polyethylenimine-mediated suicide gene transfer induces a therapeutic effect for hepatocellular carcinoma in vivo by using an Epstein-Barr virus-based plasmid vector. Biochem Biophy Res Commun 2002; 33: 48–54.
Wolschek MF, Thallinger C, Kursa M, Rössler V, Allen M, Lichtenberger C . Specific systemic nonviral gene delivery to human hepatocellular carcinoma xenografts in SCID mice. Hepatology 2002; 36: 1106–1114.
Sweeney P, Karashima T, Ishikura H, Wiehle S, Yamashita M, Benedict WF . Efficient therapeutic gene delivery after systemic administration of a novel polyethylenimine/DNA vector in an orthotopic bladder cancer model. Cancer Res 2003; 63: 4017–4020.
Thomas M, Lu JJ, Ge Q, Zhang C, Chen J, Klibanov AM . Full deacylation of polyethylenimine dramatically boosts its gene delivery efficiency and specificity to mouse lung. Proc Natl Acad Sci 2005; 102: 5679–5684.
Kircheis R, Schüller S, Brunner S, Ogris M, Heider KH, Zauner W et al. Polycation-based DNA complexes for tumor-targeted gene delivery in vivo. J Gene Med 1999; 1: 111–120.
Chollet P, Favrot MC, Hurbin A, Coll JL . Side-effects of a systemic injection of linear polyethylenimine-DNA complexes. J Gene Med 2002; 4: 84–91.
Lungwitz U, Breunig M, Blunk T, Göpferich A . Polyethylenimine-based non-viral gene delivery systems. Eur J Pharm Biopharm 2005; 60: 247–266.
Merdan T, Kunath K, Petersen H, Bakowsky U, Voigt KH, Kopecek J et al. PEGylation of poly(ethylene imine) affects stability of complexes with plasmid DNA under in vivo conditions in a dose-dependent manner after intravenous injection into mice. Bioconjug Chem 2005; 16: 785–792.
Xu DM, Yao SD, Liu YB, Sheng KL, Hong J, Gong PJ . Size-dependent properties of M-PEIs nanogels for gene delivery in cancer cells. Int J Pharm 2007; 338: 291–296.
Xu DM, Hong J, Sheng KL, Dong L, Yao SD . Preparation of polyethyleneimine nanogels via photo-Fenton reaction. Radiat Phys Chem 2007; 76: 1606–1611.
Li Y, Tang Y, Ye L, Liu B, Liu K, Chen J et al. Establishment of a hepatocellular carcinoma cell line with unique metastatic characteristics through in vivo selection and screening for metastasis-related genes through cDNA microarray. J Cancer Res Clin Oncol 2003; 129: 43–51.
Fernández-Urrusuno R, Fattal E, Féger J, Couvreur P, Thérond P . Evaluation of hepatic antioxidant systems after intravenous administration of polymeric nanoparticles. Biomaterials 1997; 18: 511–517.
Wightman L, Kircheis R, Rössler V, Carotta S, Ruzicka R, Kursa M . Different behavior of branched and linear polyethylenimine for gene delivery in vitro and in vivo. J Gene Med 2001; 3: 362–372.
Fahrmeir J, Gunther M, Tietze N, Wagner E, Ogris M . Electrophoretic purification of tumor-targeted polyethylenimine-based polyplexes reduces toxic side effects in vivo. J Control Release 2007; 122: 236–245.
El-Aneed A . An overview of current delivery systems in cancer gene therapy. J Control Release 2004; 94: 1–14.
Kircheis R, Wightman L, Schreiber A, Robitza B, Rossler V, Kursa M et al. Polyethylenimine/DNA complexes shielded by transferrin target gene expression to tumors after systemic application. Gene Therapy 2001; 8: 28–40.
Mislick KA, Baldeschwieler JD . Evidence for the role of proteoglycans in cation-mediated gene transfer. Proc Natl Acad Sci USA 1996; 93: 12349–12354.
Remy JS, Kichler A, Mordvinov V, Schuber F, Behr JP . Targeted gene transfer into hepatoma cells with lipopolyamine-condensed DNA particles presenting galactose ligands: a stage toward artificial viruses. Proc Natl Acad Sci USA 1995; 92: 1744–1748.
Brunner S, Sauer T, Carotta S, Cotton M, Saltik M, Wagner E . Cell cycle dependence of gene transfer by lipoplex, polyplex, and recombinant adenovirus. Gene Therapy 2000; 7: 401–407.
Brunner S, Fürtbauer E, Sauer T, Kursa M, Wagner E . Overcoming the nuclear barrier: cell cycle independent nonviral gene transfer with linear polyethylenimine or electroporation. Mol Ther 2002; 5: 80–86.
Acknowledgements
This work is supported by grants from National Natural Science Foundation of China, Ministry of Science and Technology of China (973 and 863 project), Shanghai Commission of Science and Technology, a special grant from E-Institute of Shanghai University's Immunology Division and National Foundation for Excellence Doctoral Project and Shanghai Municipal Education Commission for Shuguang Project.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Dong, L., Xu, H., Liu, YB. et al. M-PEIs nanogels: potential nonviral vector for systemic plasmid delivery to tumor cells. Cancer Gene Ther 16, 561–566 (2009). https://doi.org/10.1038/cgt.2009.11
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2009.11
Keywords
This article is cited by
-
Calixarenes and related macrocycles as gene delivery vehicles
Journal of Inclusion Phenomena and Macrocyclic Chemistry (2014)