Abstract
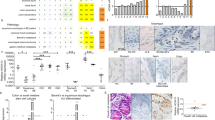
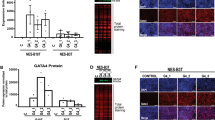
Cell surface presence of the coxsackie and adenovirus receptor (CAR) is considered a crucial prerequisite for the uptake of attenuated adenovirus. In cancers, however, a frequent loss of CAR has been noted potentially hampering the success of adenovirus-based therapy. In esophageal Barrett's carcinomas and its precursor lesions CAR presence has not been systematically determined yet. Immunohistochemical assessment in tissue specimens of 111 patients revealed CAR-positivity in all cases of Barrett's esophagus, including various degrees of intraepithelial neoplasia. In contrast, no considerable CAR presence was seen in squamous esophageal epithelium. Among Barrett's carcinomas, 93% displayed CAR presence, whereas CAR-negativity was observed preferentially in advanced cancers. Aiming to evaluate whether this loss of CAR impacts tumor-biologic properties of esophageal adenocarcinomas we studied cell lines OE19 and OE33 and observed an increased proliferation, migration and invasion upon siRNA-mediated functional CAR knock down. In conclusion, our results indicate that CAR may provide a valuable target for adenovirus-based therapy of Barrett's carcinomas and its precursor lesions. These data do also suggest that CAR does not contribute substantially to carcinogenesis in Barrett's esophagus, however, it may be speculated that loss of CAR promotes tumor progression in advanced stages of Barrett's carcinomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
Abbreviations
- BC:
-
Barrett's carcinoma
- BE:
-
Barrett's esophagus
- CAR:
-
coxsackie and adenovirus receptor
- IN:
-
intraepithelial neoplasia
- TJ:
-
tight junction
References
Bergelson JM, Cunningham JA, Droguett G, Kurt-Jones EA, Krithivas A, Hong JS et al. Isolation of a common receptor for Coxsackie B viruses and adenoviruses 2 and 5. Science 1997; 275: 1320–1323.
Cohen CJ, Shieh JT, Pickles RJ, Okegawa T, Hsieh JT, Bergelson JM . The coxsackie virus and adenovirus receptor is a transmembrane component of the tight junction. Proc Natl Acad Sci USA 2001; 98: 15191–15196.
Kasuya H, Takeda S, Shimoyama S, Shikano T, Nomura N, Kanazumi N et al. Oncolytic virus therapy—foreword. Curr Cancer Drug Targets 2007; 7: 123–125.
Heideman DA, Snijders PJ, Craanen ME, Bloemena E, Meijer CJ, Meuwissen SG et al. Selective gene delivery toward gastric and esophageal adenocarcinoma cells via EpCAM-targeted adenoviral vectors. Cancer Gene Ther 2001; 8: 342–351.
Korn WM, Macal M, Christian C, Lacher MD, McMillan A, Rauen KA et al. Expression of the coxsackievirus- and adenovirus receptor in gastrointestinal cancer correlates with tumor differentiation. Cancer Gene Ther 2006; 13: 792–797.
Matsumoto K, Shariat SF, Ayala GE, Rauen KA, Lerner SP . Loss of coxsackie and adenovirus receptor expression is associated with features of aggressive bladder cancer. Urology 2005; 66: 441–446.
Rauen KA, Sudilovsky D, Le JL, Chew KL, Hann B, Weinberg V et al. Expression of the coxsackie adenovirus receptor in normal prostate and in primary and metastatic prostate carcinoma: potential relevance to gene therapy. Cancer Res 2002; 62: 3812–3818.
Sachs MD, Rauen KA, Ramamurthy M, Dodson JL, De Marzo AM, Putzi M et al. Integrin alpha(v) and coxsackie adenovirus receptor expression in clinical bladder cancer. Urology 2002; 60: 531–536.
Bruning A, Runnebaum IB . The coxsackie adenovirus receptor inhibits cancer cell migration. Exp Cell Res 2004; 298: 624–631.
Huang KC, Altinoz M, Wosik K, Larochelle N, Koty Z, Zhu L et al. Impact of the coxsackie and adenovirus receptor (CAR) on glioma cell growth and invasion: requirement for the C-terminal domain. Int J Cancer 2005; 113: 738–745.
Okegawa T, Pong RC, Li Y, Bergelson JM, Sagalowsky AI, Hsieh JT . The mechanism of the growth-inhibitory effect of coxsackie and adenovirus receptor (CAR) on human bladder cancer: a functional analysis of car protein structure. Cancer Res 2001; 61: 6592–6600.
Okegawa T, Li Y, Pong RC, Bergelson JM, Zhou J, Hsieh JT . The dual impact of coxsackie and adenovirus receptor expression on human prostate cancer gene therapy. Cancer Res 2000; 60: 5031–5036.
Wang B, Chen G, Li F, Zhou J, Lu Y, Ma D . Inhibitory effect of coxsackie adenovirus receptor on invasion and metastasis phenotype of ovarian cancer cell line SKOV3. J Huazhong Univ Sci Technol Med Sci 2005; 25: 85–87, 93.
Anders M, Hansen R, Ding RX, Rauen KA, Bissell MJ, Korn WM . Disruption of 3D tissue integrity facilitates adenovirus infection by deregulating the coxsackie virus and adenovirus receptor. Proc Natl Acad Sci USA 2003; 100: 1943–1948.
Bruning A, Stickeler E, Diederich D, Walz L, Rohleder H, Friese K et al. Coxsackie and adenovirus receptor promotes adenocarcinoma cell survival and is expressionally activated after transition from preneoplastic precursor lesions to invasive adenocarcinomas. Clin Cancer Res 2005; 11: 4316–4320.
Flejou JF . Barrett's oesophagus: from metaplasia to dysplasia and cancer. Gut 2005; 54 (Suppl 1): i6–i12.
Montgomery E, Mamelak AJ, Gibson M, Maitra A, Sheikh S, Amr SS et al. Overexpression of claudin proteins in esophageal adenocarcinoma and its precursor lesions. Appl Immunohistochem Mol Morphol 2006; 14: 24–30.
Gonzalez-Correa CA, Brown BH, Smallwood RH, Stephenson TJ, Stoddard CJ, Bardhan KD . Low frequency electrical bioimpedance for the detection of inflammation and dysplasia in Barrett's oesophagus. Physiol Meas 2003; 24: 291–296.
Mullin JM, Valenzano MC, Trembeth S, Allegretti PD, Verrecchio JJ, Schmidt JD et al. Transepithelial leak in Barrett's esophagus. Dig Dis Sci 2006; 51: 2326–2336.
Rendon-Huerta E, Valenzano MC, Mullin JM, Trembeth SE, Kothari R, Hameed B et al. Comparison of three integral tight junction barrier proteins in Barrett's epithelium versus normal esophageal epithelium. Am J Gastroenterol 2003; 98: 1901–1903.
Gyorffy H, Holczbauer A, Nagy P, Szabó Z, Kupcsulik P, Páska C et al. Claudin expression in Barrett's esophagus and adenocarcinoma. Virchows Arch 2005; 447: 961–968.
Marsman WA, Buskens CJ, Wesseling JG, Offerhaus GJ, Bergman JJ, Tytgat GN et al. Gene therapy for esophageal carcinoma: the use of an explant model to test adenoviral vectors ex vivo. Cancer Gene Ther 2004; 11: 289–296.
Hamilton SR, Aaltonen LA, WHO Classification. Tumours of the Digestive System: Pathology & Genetics. IARC Press: Lyon, France, 2000.
Wittekind C, Meyer HJ, Bootz F . TNM Classification of Malignant Tumors (ed 6). John Wiley & Sons Inc, New York, NY, 2002.
Montgomery E, Goldblum JR, Greenson JK, Haber MM, Lamps LW, Lauwers GY et al. Dysplasia as a predictive marker for invasive carcinoma in Barrett esophagus: a follow-up study based on 138 cases from a diagnostic variability study. Hum Pathol 2001; 32: 379–388.
Sarbia M, Donner A, Franke C, Gabbert HE . Distinction between intestinal metaplasia in the cardia and in Barrett's esophagus: the role of histology and immunohistochemistry. Hum Pathol 2004; 35: 371–376.
Rockett JC, Larkin K, Darnton SJ, Morris AG, Matthews HR . Five newly established oesophageal carcinoma cell lines: phenotypic and immunological characterization. Br J Cancer 1997; 75: 258–263.
Heitmiller RF . Prophylactic esophagectomy in Barrett esophagus with high-grade dysplasia. Langenbecks Arch Surg 2003; 388: 83–87.
Prasad GA, Wang KK, Buttar NS, Wongkeesong LM, Krishnadath KK, Nichols III FC et al. Long-term survival following endoscopic and surgical treatment of high-grade dysplasia in Barrett's esophagus. Gastroenterology 2007; 132: 1226–1233.
Marsman WA, Buskens CJ, Wesseling JG, van Lanschot JJ, Bosma PJ . Gene therapy for Barrett's esophagus: adenoviral gene transfer in different intestinal models. Cancer Gene Ther 2005; 12: 778–786.
Raschperger E, Thyberg J, Pettersson S, Philipson L, Fuxe J, Pettersson RF . The coxsackie- and adenovirus receptor (CAR) is an in vivo marker for epithelial tight junctions, with a potential role in regulating permeability and tissue homeostasis. Exp Cell Res 2006; 312: 1566–1580.
Acknowledgements
We thank Dr J Bergelson (Division of Infectious Diseases, Children's Hospital of Philadelphia, PA) for providing the ‘CHO-CAR’ cell line.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Anders, M., Rösch, T., Küster, K. et al. Expression and function of the coxsackie and adenovirus receptor in Barrett's esophagus and associated neoplasia. Cancer Gene Ther 16, 508–515 (2009). https://doi.org/10.1038/cgt.2008.103
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2008.103
Keywords
This article is cited by
-
Coxsackie and adenovirus receptor is a critical regulator for the survival and growth of oral squamous carcinoma cells
Oncogene (2014)
-
A simple detection system for adenovirus receptor expression using a telomerase-specific replication-competent adenovirus
Gene Therapy (2013)
-
Impact of the coxsackievirus and adenovirus receptor on the adenoma–carcinoma sequence of colon cancer
British Journal of Cancer (2011)
-
Expression of coxsackie and adenovirus receptor distinguishes transitional cancer states in therapy-induced cellular senescence
Cell Death & Disease (2010)