Abstract
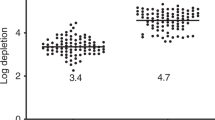
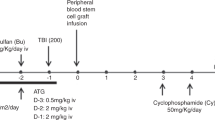
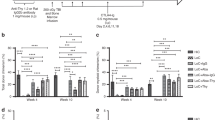
We evaluated the depletion of TCR-alpha/beta cells from the graft of children with high-risk AML, who received transplantation from unrelated (n=20) and haploidentical donors (n=13). The preparative regimen included treosulfan, melphalan, fludarabine and anti-thymocyte globulin. Grafts were PBSC engineered by TCR-alpha/beta and CD19 depletion. The graft contained a median of 9 × 106/kg of CD34+ and 20 × 103/kg of αβ-T cells. Post-transplant immune suppression included tacrolimus till day +30 and Mtx in 21 patients, tacrolimus in 5, Mtx in 2 and no prophylaxis in 5 patients. Sixteen patients received native or TCR-alpha/beta-depleted donor lymphocytes at a median of 47 (40–204) days. Median follow-up is 1.76 years. Primary engraftment was achieved in 33 patients (100%). Cumulative incidence of acute GvHD (aGvHD) grade 2–3 was 39 (26–60)%, half of them had skin-only aGvHD. Cumulative incidence of chronic GvHD was 30(18–50)%. Transplant-related mortality is 10(4–26)%. Event-free survival (EFS) is 60(43–76)% and overall survival (OS) is 67(50–84)% at 2 years. In a subgroup of patients, who received transplantation in CR, EFS is 66(48–84)% and OS−72(53–90)% at 2 years. Our data suggest that TCR-alpha/beta and CD19 depletion is a robust method of graft manipulation, which can be used to engineer grafts for children with AML
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Niewerth D, Creutzig U, Bierings MB, Kaspers GJ . A review on allogeneic stem cell transplantation for newly diagnosed pediatric acute myeloid leukemia. Blood 2010; 116: 2205–2214.
Hasle H . A critical review of which children with acute myeloid leukaemia need stem cell procedures. Br J Haematol 2014; 166: 23–33.
Locatelli F, Masetti R, Rondelli R, Zecca M, Fagioli F, Rovelli A et al. Outcome of children with high-risk acute myeloid leukemia given autologous or allogeneic hematopoietic cell transplantation in the AIEOP AML-2002/01 study. Bone Marrow Transplant 2015; 50: 181–188.
Burke MJ, Wagner JE, Cao Q, Ustun C, Verneris MR . Allogeneic hematopoietic cell transplantation in first remission abrogates poor outcomes associated with high-risk pediatric acute myeloid leukemia. Biol Blood Marrow Transplant 2013; 19: 1021–1025.
Shaw PJ, Kan F, Woo Ahn K, Spellman SR, Aljurf M, Ayas M et al. Outcomes of pediatric bone marrow transplantation for leukemia and myelodysplasia using matched sibling, mismatched related, or matched unrelated donors. Blood 2010; 116: 4007–4015.
Leung W, Campana D, Yang J, Pei D, Coustan-Smith E, Gan K et al. High success rate of hematopoietic cell transplantation regardless of donor source in children with very high-risk leukemia. Blood 2011; 118: 223–230.
Di Stasi A, Milton DR, Poon LM, Hamdi A, Rondon G, Chen J et al. Similar transplantation outcomes for acute myeloid leukemia and myelodysplastic syndrome patients with haploidentical versus 10/10 human leukocyte antigen-matched unrelated and related donors. Biol Blood Marrow Transplant 2014; 20: 1975–1981.
Booth C, Lawson S, Veys P . The current role of T cell depletion in paediatric stem cell transplantation. Br J Haematol 2013; 162: 177–190.
Ho VT, Soiffer RJ . The history and future of T-cell depletion as graft-versus-host disease prophylaxis for allogeneic hematopoietic stem cell transplantation. Blood 2001; 98: 3192–3204.
Aversa F, Tabilio A, Velardi A, Cunningham I, Terenzi A, Falzetti F et al. Treatment of high-risk acute leukemia with T-cell-depleted stem cells from related donors with one fully mismatched HLA haplotype. N Engl J Med 1998; 339: 1186–1193.
Schumm M, Lang P, Bethge W, Faul C, Feuchtinger T, Pfeiffer M et al. Depletion of T-cell receptor alpha/beta and CD19 positive cells from apheresis products with the CliniMACS device. Cytotherapy 2013; 15: 1253–1258.
Chaleff S, Otto M, Barfield RC, Leimig T, Iyengar R, Martin J et al. A large-scale method for the selective depletion of alphabeta T lymphocytes from PBSC for allogeneic transplantation. Cytotherapy 2007; 9: 746–754.
Handgretinger R . New approaches to graft engineering for haploidentical bone marrow transplantation. Semin Oncol 2012; 39: 664–673.
Bertaina A, Merli P, Rutella S, Pagliara D, Bernardo ME, Masetti R et al. HLA-haploidentical stem cell transplantation after removal of alphabeta+ T and B cells in children with nonmalignant disorders. Blood 2014; 124: 822–826.
Lang P, Feuchtinger T, Teltschik HM, Schwinger W, Schlegel P, Pfeiffer M et al. Improved immune recovery after transplantation of TCRalphabeta/CD19-depleted allografts from haploidentical donors in pediatric patients. Bone Marrow Transplant 2015; 50: S6–10.
Przepiorka D, Weisdorf D, Martin P, Klingemann HG, Beatty P, Hows J et al. 1994 Consensus Conference on Acute GVHD Grading. Bone Marrow Transplant 1995; 15 (6): 825–828.
Laberko A, MM, Shelikhova L, Balashov D, Skvortsova J, Boyakova E et al. Analysis of risk factors of viral reactivation after haploidentical and matched unrelated hematopoietic stem cell transplantation with TCR alpha/beta and CD19 depletion. Bone Marrow Transplant 2015; 50: S187–S188.
Handgretinger R, Schumm M, Lang P, Greil J, Reiter A, Bader P et al. Transplantation of megadoses of purified haploidentical stem cells. Ann N Y Acad Sci 1999; 872: 351–361.
Atta EH, de Sousa AM, Schirmer MR, Bouzas LF, Nucci M, Abdelhay E . Different outcomes between cyclophosphamide plus horse or rabbit antithymocyte globulin for HLA-identical sibling bone marrow transplant in severe aplastic anemia. Biol Blood Marrow Transplant 2012; 18: 1876–1882.
Oevermann L, Lang P, Feuchtinger T, Schumm M, Teltschik HM, Schlegel P et al. Immune reconstitution and strategies for rebuilding the immune system after haploidentical stem cell transplantation. Ann N Y Acad Sci 2012; 1266: 161–170.
Eyrich M, Leiler C, Lang P, Schilbach K, Schumm M, Bader P et al. A prospective comparison of immune reconstitution in pediatric recipients of positively selected CD34+ peripheral blood stem cells from unrelated donors vs recipients of unmanipulated bone marrow from related donors. Bone Marrow Transplant 2003; 32: 379–390.
Wachowiak J, Sykora KW, Cornish J, Chybicka A, Kowalczyk JR, Gorczynska E et al. Treosulfan-based preparative regimens for allo-HSCT in childhood hematological malignancies: a retrospective study on behalf of the EBMT pediatric diseases working party. Bone Marrow Transplant 2011; 46: 1510–1518.
Passweg JR, Baldomero H, Bader P, Bonini C, Cesaro S, Dreger P et al. Hematopoietic SCT in Europe 2013: recent trends in the use of alternative donors showing more haploidentical donors but fewer cord blood transplants. Bone Marrow Transplant 2015; 50: 476–482.
Lankester AC, Locatelli F, Bader P, Rettinger E, Egeler M, Katewa S et al. Will post-transplantation cell therapies for pediatric patients become standard ofcare? Biol Blood Marrow Transplant 2015; 21: 402–411.
Acknowledgements
We would wish to thank the BMT Unit staff, especially doctors Olga Tatarinova, Elena Gutovskaya, Daria Shasheleva, Zhanna Shekhovtsova, Irina Shipitsina, Rimma Khismatullina, Vera Tzetlina. ‘Podari Zhizn’ charitable fund for continued support of the transplantation program. Stefan Morsch stiftung for years of collaboration in unrelated transplantation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Maschan, M., Shelikhova, L., Ilushina, M. et al. TCR-alpha/beta and CD19 depletion and treosulfan-based conditioning regimen in unrelated and haploidentical transplantation in children with acute myeloid leukemia. Bone Marrow Transplant 51, 668–674 (2016). https://doi.org/10.1038/bmt.2015.343
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2015.343