Abstract
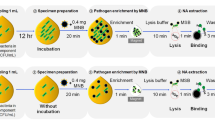
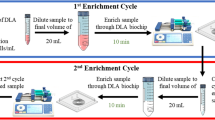
Transfusion of the ‘wrong’ stem cell product would almost inevitably be lethal, yet assays to confirm the contents of the product bag, except by checking labels and paperwork, are lacking. To increase the likelihood that a product mix-up would be detected in the transplant center, we developed a simple protocol for extended blood typing and hence, for confirmation of donor/product identity, on a tube segment. Apheresis samples were applied, directly or after erythrocyte enrichment, to commercially available blood typing assays, including lateral flow cards and gel agglutination cards. Without sample modification, low hematocrit and high leukocyte count obviated definitive blood typing. Using the most simple erythrocyte enrichment protocol, that is, centrifugation, reliable blood group analysis became possible with either assay. Other, more cumbersome pre-analytical protocols were also successful but provided no advantage. The preferred method was validated on 100 samples; ABD was correctly identified in 100% of cases. Of the other Rh Ags, all except two ‘small e’, in both cases in heterozygous individuals, were detected; there were no false positives. A simple, inexpensive point-of-care assay for extended blood typing of apheresis products is available, which can reduce the fatal risk of administering the wrong stem cell product.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Warkentin PI, Nick L, Shpall EJ . Hematopoietic cell procurement, processing and transplantation: regulation and accreditation. In: Appelbaum FR, Forman SJ, Negrin RS, Blume KG (eds). Thomas' Hematopoietic Cell Transplantation, 4th edn. Wiley-Blackwell: Hobokoen, NJ, USA, 2011.
Foeken LM, Green A, Hurley CK, Marry E, Wiegand T, Oudshoorn M . Monitoring the international use of unrelated donors for transplantation: the WMDA annual reports. Bone Marrow Transplant 2010; 45: 811–818.
Cohen H . SHOT 2009 Annual Report Summary. http://www.shotuk.org/wp-content/uploads/2010/07/SHOT-2009.pdf-Summary.pdf 2011 May 23 (cited 23 May 2011).
McCullough J, McKenna D, Kadidlo D, Maurer D, Noreen HJ, French K et al. Mislabeled units of umbilical cord blood detected by a quality assurance program at the transplantation center. Blood 2009; 114: 1684–1688.
Gassner C, Rainer E, Pircher E, Markut L, Kormoczi GF, Jungbauer C et al. Application of a multivariant, caucasian-specific, genotyped donor panel for performance validation of MDmulticard(R), ID-System(R), and Scangel(R) RhD/ABO Serotyping. Transfus Med Hemother 2009; 36: 219–225.
Lapierre Y, Rigal D, Adam J, Josef D, Meyer F, Greber S et al. The gel test: a new way to detect red cell antigen-antibody reactions. Transfusion 1990; 30: 109–113.
Race RR, Mourant AE . The Rh chromosome frequencies in England. Blood 1948; 3: 689–695.
Mathe G, Schwarzenberg L, Amiel JL, Schneider M, Cattan A, Schlumberger JR et al. Immunogenetic and immunological problems of allogeneic haemopoietic radio-chimaeras in man. Scand J Haematol 1967; 4: 193–216.
Thomas ED, Epstein RB . Bone marrow transplantation in acute leukemia. Cancer Res 1965; 25: 1521–1524.
Acknowledgements
The training provided to CC by technicians from the Immunohematology laboratory is gratefully acknowledged.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
PS and MS are employees of Medion Grifols Diagnostics, a product of which was used for these studies. None of the other authors have a potential conflict of interest to declare.
Additional information
The data were previously presented in abstract form at the 2010 ISBT annual meeting.
Rights and permissions
About this article
Cite this article
Cummerow, C., Schwind, P., Spicher, M. et al. Apheresis product identification in the transplant center: development of point-of-care protocols for extended blood typing of stem cell apheresis products. Bone Marrow Transplant 47, 860–865 (2012). https://doi.org/10.1038/bmt.2011.182
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2011.182
Keywords
This article is cited by
-
Minor RBC Ab and allo-SCT
Bone Marrow Transplantation (2014)