Abstract
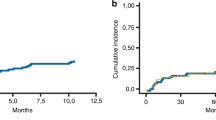
The assessment of a hematopoietic stem cell transplant (HSCT)-specific comorbidity index (HCT-CI) has been developed to predict the risk of TRM in patients undergoing allogeneic HSCT. As the myeloablative fludarabine/i.v. busulfan (FluBu4) regimen has been associated with limited extra-hematologic toxicity, we analyzed whether the HCT-CI represents a useful tool in transplant patients conditioned with this regimen. Of the 52 consecutive patients who received an allogeneic HSCT with FluBu4 at our institution, 50 were evaluable for assessing pre-transplant HCT-CI. Patients were divided into three groups: score 0 (n=7); score 1–2 (n=17) and score >3 (n=26). The three groups did not differ significantly in age, diagnosis, previous lines of chemotherapy and type of donor. High-risk disease was present in 57% of low, 82% of intermediate and 85% of high HCT-CI score groups (P=ns). Two-year TRM and OS was 14.3 and 85.7% in the low score group, 23.5 and 58.8% in the intermediate score group and 15.4 and 50% in the high HCT-CI score group (P=ns). In this study, the HCT-CI lacked sensitivity to reliably predict TRM although patients with no comorbidities showed a trend for improved survival.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Bearman SI, Appelbaum FR, Buckner CD, Petersen FB, Fisher LD, Clift RA et al. Regimen-related toxicity in patients undergoing bone marrow transplantation. J Clin Oncol 1988; 6: 1562–1568.
Sorror ML, Maris MB, Storer B, Sandmaier BM, Diaconescu R, Flowers C et al. Comparing morbidity and mortality of HLA-matched unrelated donor hematopoietic cell transplantation after nonmyeloablative and myeloablative conditioning: influence of pretransplantation comorbidities. Blood 2004; 104: 961–968.
Sorror ML, Maris MB, Storb R, Baron F, Sandmaier BM, Maloney DG et al. Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood 2005; 106: 2912–2919.
Chunduri S, Dobogai LC, Peace D, Saunthararajah Y, Chen HY, Mahmud N et al. Comparable kinetics of myeloablation between fludarabine/full-dose busulfan and fludarabine/melphalan conditioning regimens in allogeneic peripheral blood stem cell transplantation. Bone Marrow Transplant 2006; 38: 477–482.
Beri R, Chunduri S, Sweiss K, Peace DJ, Mactal-Haaf C, Dobogai LC et al. Reliability of a pretransplant i.v. BU test dose performed 2 weeks before myeloablative FluBu conditioning regimen. Bone Marrow Transplant 2010; 45: 249–253.
Chunduri S, Dobogai LC, Peace D, Saunthararajah Y, Quigley J, Chen YH et al. Fludarabine/i.v. BU conditioning regimen: myeloablative, reduced intensity or both? Bone Marrow Transplant 2008; 41: 935–940.
Sorror ML, Sandmaier BM, Storer BE, Maris MB, Baron F, Maloney DG et al. Comorbidity and disease status-based risk stratification of outcomes among patients with acute myeloid leukemia or myelodysplasia receiving allogeneic hematopoietic cell transplantation. J Clin Oncol 2007; 25: 4246–4254.
Sorror ML, Storer BE, Maloney DG, Sandmaier BM, Martin PJ, Storb R . Outcomes after allogeneic hematopoietic cell transplantation with nonmyeloablative or myeloablative regimens for treatment of lymphoma and chronic lymphocytic leukemia. Blood 2008; 111: 446–452.
Sorror ML, Giralt S, Sandmaier BM, De Lima M, Shahjahan M, Maloney DG et al. Hematopoietic cell transplantation-specific comorbidity index as an outcome predictor for patients with acute myeloid leukemia in first remission: combined FHCRC and MDACC experiences. Blood 2007; 110: 4608–4613.
Guilfoyle R, Demers A, Bredeson C, Richardson E, Rubinger M, Szwajcer D et al. Performance status, but not the hematopoietic cell transplantation comorbidity index (HCT-CI), predicts mortality at a Canadian transplant center. Bone Marrow Transplant 2009; 43: 133–139.
Majhail NS, Brunstein CG, McAvoy S, DeFor TE, Al-Hazzouri A, Setubal D et al. Does the hematopoietic cell transplantation specific comorbidity index predict transplant outcomes? A validation study in a large cohort of umbilical cord blood and matched related donor transplants. Biol Blood Marrow Transplant 2008; 14: 985–992.
Pollack SM, Steinberg SM, Odom J, Dean RM, Fowler DH, Bishop MR . Assessment of the hematopoietic cell transplantation comorbidity index in non-Hodgkin lymphoma patients receiving reduced-intensity allogeneic hematopoietic stem cell transplantation. Biol Blood Marrow Transplant 2009; 15: 223–230.
Farina L, Bruno B, Patriarca F, Spina F, Sorasio R, Morelli M et al. The hematopoietic cell transplantation comorbidity index (HCT-CI) predicts clinical outcomes in lymphoma and myeloma patients after reduced-intensity or non-myeloablative allogeneic stem cell transplantation. Leukemia 2009; 23: 1131–1138.
Lim ZY, Ingram W, Brand R, Ho A, Kenyon M, Devereux S et al. Impact of pretransplant comorbidities on alemtuzumab-based reduced intensity conditioning allogeneic hematopoietic SCT for patients with high-risk myelodysplastic syndrome and AML. Bone Marrow Transplant 2010; 45: 633–639.
Kataoka K, Nannya Y, Ueda K, Kumano K, Takahashi T, Kurokawa M . Differential prognostic impact of pretransplant comorbidity on transplant outcomes by disease status and time from transplant: a single Japanese transplant centre study. Bone Marrow Transplant 2010; 45: 513–520.
Sorror M, Storer B, Sandmaier BM, Maloney DG, Chauncey TR, Langston A et al. Hematopoietic cell transplantation comorbidity index and Karnofsky performance status are independent predictors of morbidity and mortality after allogeneic nonmyeloablative hematopoietic cell transplantation. Cancer 2008; 112: 1992–2001.
Barba P, Piñana JL, Martino R, Valcárcel D, Amorós A, Sureda A et al. Comparison of two pretransplant predictive models and a flexible HCT-CI using different cut points to determine low, intermediate and high risk groups: the flexible HCT-CI is the best predictor of NRM and OS in a population of patients undergoing allo-RIC. Biol Blood Marrow Transplant 2010; 16: 413–420.
Sorror ML, Storer B, Storb RF . Validation of the hematopoietic cell transplantation-specific comorbidity index (HCT-CI) in single and multiple institutions: limitations and inferences. Biol Blood Marrow Transplant 2009; 15: 757–758.
Acknowledgements
This project was supported by the University of Illinois at Chicago (UIC) Center for Clinical and Translational Science (CCTS) award number UL1RR029879 from the National Center for Research Resources. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Center for Research Resources or the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Patel, P., Sweiss, K., Nimmagadda, S. et al. Comorbidity index does not predict outcome in allogeneic myeloablative transplants conditioned with fludarabine/i.v. busulfan (FluBu4). Bone Marrow Transplant 46, 1326–1330 (2011). https://doi.org/10.1038/bmt.2010.293
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2010.293
Keywords
This article is cited by
-
Toxicity and efficacy of busulfan and fludarabine myeloablative conditioning for HLA-identical sibling allogeneic hematopoietic cell transplantation in AML and MDS
Bone Marrow Transplantation (2016)
-
I.v. BU/fludarabine plus melphalan or TBI in unrelated cord blood transplantation for high-risk hematological diseases
Bone Marrow Transplantation (2015)