Abstract
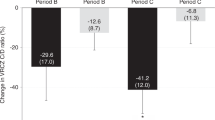
Although voriconazole has been shown to interact with calcineurin inhibitors, this interaction has not been thoroughly examined. The purpose of this study was to evaluate the drug interaction between voriconazole and calcineurin inhibitors among recipients of allogeneic hematopoietic stem cell transplantation (HSCT). Twenty-one recipients of allogeneic HSCT were evaluated. Those recipients had been on CsA (n=10) or tacrolimus (n=11) when voriconazole (400 mg per day orally, or 8 mg/kg per day, i.v.) was initiated. Trough concentrations of calcineurin inhibitors were measured before and periodically after initiating voriconazole to determine the concentration/dose (C/D) ratio of calcineurin inhibitors. Median C/D ratio significantly increased by initiating voriconazole: from 86.0 (range, 43.5–178.8) to 120.2 (range, 86.1–379.4) in CsA (P<0.05), and from 595.9 (range, 51.3–1643.3) to 890.7 (range, 94.1–4658.3) (ng/ml)/(mg/kg) in tacrolimus (P<0.01). Median increases in the C/D ratio did not differ significantly between CsA and tacrolimus (82.1%, ranging from −9.4 to 266.9% vs 115.6%, ranging from 25.4 to 307.6%). These results indicate that voriconazole alters the blood concentration of calcineurin inhibitors with a wide range of interindividual variability after allogeneic HSCT. Dose adjustment of calcineurin inhibitors on initiating voriconazole should not be decided uniformly, but determined on an individual basis by close monitoring of their blood concentrations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Dykewicz CA, Jaffe HW . Guidelines for preventing opportunistic infections among hematopoietic stem cell transplant recipients. Biol Blood Marrow Transplant 2006; 6: 670–672.
Marr KA, Carter RA, Crippa F, Wald A, Corey L . Epidemiology and outcome of mould infections in hematopoietic stem cell transplant recipients. Clin Infect Dis 2002; 34: 909–917.
Kami M, Machida U, Okuzumi K, Matsumura T, Mori S, Hori A et al. Effect of fluconazole prophylaxis on fungal blood cultures: an autopsy-based study involving 720 patients with haematological malignancy. Br J Haematol 2002; 117: 40–46.
Herbrecht R, Denning DW, Patterson TF, Bennett JE, Greene RE, Oestmann JW et al. Voriconazole versus amphotericin B for primary therapy of invasive aspergillosis. N Engl J Med 2002; 347: 408–415.
Jeu L, Piacenti FJ, Lyakhovetskiy AG, Fung HB . Voriconazole. Clin Ther 2003; 25: 1321–1381.
Saad AH, DePestel DD, Carver PL . Factors influencing the magnitude and clinical significance of drug interactions between azole antifungals and select immunosuppressants. Pharmacotherapy 2006; 26: 1730–1744.
Romero AJ, Le Pogamp P, Nilsson LG, Wood N . Effect of voriconazole on the pharmacokinetics of cyclosporine in renal transplant patients. Clin Pharmacol Ther 2002; 71: 226–234.
Wood N, Tank K, Allan R, Fielding A . Effect of voriconazole on the pharmacokinetics of tacrolimus. In: Program and Abstracts of the 41st Interscience Conference on Antimicrobial Agents and Chemotherapy, Chicago, Illinois, 22–25 September 2001 (abstract A-20). American Society of Microbiology: Washington, DC, 2001.
Tintillier M, Kirch L, Goffin E, Cuvelier C, Pochet JM . Interaction between voriconazole and tacrolimus in a kidney-transplanted patient. Nephrol Dial Transplant 2005; 20: 664–665.
Venkataramanan R, Zang S, Gayowski T, Singh N . Voriconazole inhibition of the metabolism of tacrolimus in a liver transplant recipient and in human liver microsomes. Antimicrob Agents Chemother 2002; 46: 3091–3093.
Pai MP, Allen S . Voriconazole inhibition of tacrolimus metabolism. Clin Infect Dis 2003; 36: 1089–1091.
Groll AH, Kolve H, Ehlert K, Paulussen M, Vormoor J . Pharmacokinetic interaction between voriconazole and ciclosporin A following allogeneic bone marrow transplantation. J Antimicrob Chemother 2004; 53: 113–114.
Pfizer Inc. Vfend (voriconazole) package insert. New York, NY, 2008.
Trifilio S, Ortiz R, Pennick G, Verma A, Pi J, Stosor V et al. Voriconazole therapeutic drug monitoring in allogeneic hematopoietic stem cell transplant recipients. Bone Marrow Transplant 2005; 35: 509–513.
Acknowledgements
This study was supported in part by grants from Nagao Memorial Fund.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Mori, T., Aisa, Y., Kato, J. et al. Drug interaction between voriconazole and calcineurin inhibitors in allogeneic hematopoietic stem cell transplant recipients. Bone Marrow Transplant 44, 371–374 (2009). https://doi.org/10.1038/bmt.2009.38
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2009.38
Keywords
This article is cited by
-
Intrapatient variability in concentration/dose ratio of tacrolimus predicts transplant-associated thrombotic microangiopathy
International Journal of Hematology (2021)
-
Pharmacokinetic comparison of cyclosporin A and tacrolimus in graft-versus-host disease prophylaxis
Annals of Hematology (2017)
-
Influence of CYP2C19*2/*17 genotype on adverse drug reactions of voriconazole in patients after allo-HSCT: a four-case report
Journal of Cancer Research and Clinical Oncology (2017)
-
Different effects of lansoprazole and rabeprazole on the plasma voriconazole trough levels in allogeneic hematopoietic cell transplant recipients
Annals of Hematology (2016)
-
A prospective feasibility study of primary prophylaxis against invasive fungal disease with voriconazole following umbilical cord blood transplantation with fludarabine-based conditioning
International Journal of Hematology (2014)