Abstract
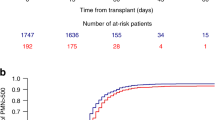
Acute GVHD has remained a significant cause of nonrelapse mortality after allogeneic hematopoietic cell transplantation (HCT) with nonmyeloablative conditioning. The role of TNF-α in the biology of acute GVHD after nonmyeloablative conditioning has not been studied thus far. Here, we measured TNF receptor 1 (TNFR1) as a surrogate marker for TNF-α in 106 patients before the start of the conditioning regimen (baseline) and 7 days after allogeneic HCT with nonmyeloablative conditioning. The nonmyeloablative regimen consisted of 2 Gy TBI alone (n=15), 2 Gy TBI plus fludarabine 90 mg/m2 (n=73), or 4 Gy TBI plus fludarabine 90 mg/m2 (n=18). TNFR1 levels increased significantly from baseline to day 7 after nonmyeloablative HCT (P<0.0001). Patients conditioned with 4 Gy TBI had higher TNFR1 day 7/baseline ratio than those conditioned with 2 Gy TBI (median 1.65 versus 1.25; P=0.01). In a multivariate Cox model, high TNFR1 day7/baseline ratio was associated with grades II–IV (HR=2.2, P=0.01) and grades III–IV (HR=2.9, P=0.007) acute GVHD, but had no impact on overall survival (P=0.8). In summary, our data suggest that nonmyeloablative conditioning induces the generation of TNF-α, and that the magnitude of TNF-α generation depends on the conditioning intensity (2 Gy versus 4 Gy TBI). Further, assessment of TNFR1 levels before and on day 7 after nonmyeloablative HCT provided useful information on subsequent risk of experiencing acute GVHD.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
McSweeney PA, Niederwieser D, Shizuru JA, Sandmaier BM, Molina AJ, Maloney DG et al. Hematopoietic cell transplantation in older patients with hematologic malignancies: replacing high-dose cytotoxic therapy with graft-versus-tumor effects. Blood 2001; 97: 3390–3400.
Sandmaier BM, Mackinnon S, Childs RW . Reduced intensity conditioning for allogeneic hematopoietic cell transplantation: current perspectives. Biol Blood Marrow Transplant 2007; 13: 87–97.
Baron F, Sandmaier BM, Storer BE, Maris MB, Langston AA, Lange T et al. Extended mycophenolate mofetil and shortened cyclosporine failed to reduce graft-versus-host disease after unrelated hematopoietic cell transplantation with nonmyeloablative conditioning. Biol Blood Marrow Transplant 2007; 13: 1041–1048.
Maris MB, Sandmaier BM, Storer BE, Maloney DG, Shizuru JA, Agura E et al. Unrelated donor granulocyte colony-stimulating factor-mobilized peripheral blood mononuclear cell transplantation after nonmyeloablative conditioning: the effect of postgrafting mycophenolate mofetil dosing. Biol Blood Marrow Transplant 2006; 12: 454–465.
Baron F, Storb R, Storer BE, Maris MB, Niederwieser D, Shizuru JA et al. Factors associated with outcomes in allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning after failed myeloablative hematopoietic cell transplantation. J Clin Oncol 2006; 24: 4150–4157.
Storb R, Yu C, Wagner JL, Deeg HJ, Nash RA, Kiem H-P et al. Stable mixed hematopoietic chimerism in DLA-identical littermate dogs given sublethal total body irradiation before and pharmacological immunosuppression after marrow transplantation. Blood 1997; 89: 3048–3054.
Baron F, Maris MB, Sandmaier BM, Storer BE, Sorror M, Diaconescu R et al. Graft-versus-tumor effects after allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning. J Clin Oncol 2005; 23: 1993–2003.
Baron F, Petersdorf EW, Gooley T, Sandmaier BM, Malkki M, Chauncey TR et al. What is the role for donor natural killer cells after nonmyeloablative conditioning? Biol Blood Marrow Transplant 2009; 15: 580–588.
Hill GR . Inflammation and bone marrow transplantation. Biol Blood Marrow Transplant 2008; 15: 139–141.
Ferrara JL, Levine JE, Reddy P, Holler E . Graft-versus-host disease. Lancet 2009; 373: 1550–1561.
Schmaltz C, Alpdogan O, Muriglan SJ, Kappel BJ, Rotolo JA, Ricchetti ET et al. Donor T cell-derived TNF is required for graft-versus-host disease and graft-versus-tumor activity after bone marrow transplantation. Blood 2003; 101: 2440–2445.
Holler E, Kolb HJ, Mittermuller J, Kaul M, Ledderose G, Duell T et al. Modulation of acute graft-versus-host-disease after allogeneic bone marrow transplantation by tumor necrosis factor alpha (TNF alpha) release in the course of pretransplant conditioning: role of conditioning regimens and prophylactic application of a monoclonal antibody neutralizing human TNF alpha (MAK 195F). Blood 1995; 86: 890–899.
Levine JE, Paczesny S, Mineishi S, Braun T, Choi SW, Hutchinson RJ et al. Etanercept plus methylprednisolone as initial therapy for acute graft-versus-host disease. Blood 2008; 111: 2470–2475.
Holler E, Kolb HJ, Hintermeier-Knabe R, Mittermüller J, Thierfelder S, Kaul M et al. Role of tumor necrosis factor alpha in acute graft-versus-host disease and complications following allogeneic bone marrow transplantation. Transplant Proc 1993; 25: 1234–1236.
Or R, Kalinkovich A, Nagler A, Weisman Z, Naparstek E, Weiss L et al. Soluble tumor necrosis factor (sTNF) receptors: a possible prognostic marker for bone marrow transplantation-related complications. Cytokines Mol Ther 1996; 2: 243–250.
Choi SW, Kitko CL, Braun T, Paczesny S, Yanik G, Mineishi S et al. Change in plasma tumor necrosis factor receptor 1 levels in the first week after myeloablative allogeneic transplantation correlates with severity and incidence of GVHD and survival. Blood 2008; 112: 1539–1542.
Kitko CL, Paczesny S, Yanik G, Braun T, Jones D, Whitfield J et al. Plasma elevations of tumor necrosis factor-receptor-1 at day 7 postallogeneic transplant correlate with graft-versus-host disease severity and overall survival in pediatric patients. Biol Blood Marrow Transplant 2008; 14: 759–765.
Mielcarek M, Martin PJ, Leisenring W, Flowers MED, Maloney DG, Sandmaier BM et al. Graft-versus-host disease after nonmyeloablative versus conventional hematopoietic stem cell transplantation. Blood 2003; 102: 756–762.
Sykes M . Mixed chimerism and transplant tolerance (review). Immunity 2001; 14: 417–424.
Baron F, Baker JE, Storb R, Gooley TA, Sandmaier BM, Maris MB et al. Kinetics of engraftment in patients with hematologic malignancies given allogeneic hematopoietic cell transplantation after nonmyeloablative conditioning. Blood 2004; 104: 2254–2262.
Devetten MP, Vose JM . Graft-versus-host disease: how to translate new insights into new therapeutic strategies (Review). Biol Blood Marrow Transplant 2004; 10: 815–825.
Mohty M, Blaise D, Faucher C, Vey N, Bouabdallah R, Stoppa A-M et al. Inflammatory cytokines and acute graft-versus-host disease after reduced-intensity conditioning allogeneic stem cell transplantation. Blood 2005; 106: 4407–4411.
Shlomchik WD, Couzens MS, Tang CB, McNiff J, Robert ME, Liu J et al. Prevention of graft versus host disease by inactivation of host antigen-presenting cells. Science 1999; 285: 412–415.
Kahl C, Storer BE, Sandmaier BM, Mielcarek M, Maris MB, Blume KG et al. Relapse risk in patients with malignant diseases given allogeneic hematopoietic cell transplantation after nonmyeloablative conditioning. Blood 2007; 110: 2744–2748.
Baron F, Schaaf-Lafontaine N, Humblet-Baron S, Meuris N, Castermans E, Baudoux E et al. T-cell reconstitution after unmanipulated, CD8-depleted or CD34-selected nonmyeloablative peripheral blood stem-cell transplantation. Transplantation 2003; 76: 1705–1713.
Castermans E, Baron F, Willems E, Schaaf-Lafontaine N, Meuris N, Gothot A et al. Evidence for neo-generation of T cells by the thymus after non-myeloablative conditioning. Haematologica 2008; 93: 240–247.
Filipovich AH, Weisdorf D, Pavletic S, Socie G, Wingard JR, Lee SJ et al. National Institutes of Health consensus development project on criteria for clinical trials in chronic graft-versus-host disease: I. Diagnosis and Staging Working Group report. Biol Blood Marrow Transplant 2005; 11: 945–956.
Sullivan KM . Graft-vs.-host disease. In: Blume KG, Forman SJ, Appelbaum FR (eds). Thomas’ Hematopoietic Cell Transplantation. Blackwell Publishing Ltd: Oxford, UK, 2004. pp 635–664.
Willems E, Baron F, Baudoux E, Wanten N, Seidel L, Vanbellinghen JF et al. Non-myeloablative transplantation with CD8-depleted or unmanipulated peripheral blood stem cells: a phase II randomized trial. Leukemia 2009; 23: 608–610.
Willems E, Baron F, Seidel L, Frere P, Fillet G, Beguin Y . Comparison of thrombotic microangiopathy after allogeneic hematopoietic cell transplantation with high-dose or nonmyeloablative conditioning. Bone Marrow Transplant 2009 (e-pub ahead of print 31 August 2009; doi:10.1038/bmt2009.230).
Hill GR, Crawford JM, Cooke KR, Brinson YS, Pan L, Ferrara JL . Total body irradiation and acute graft-versus-host disease: the role of gastrointestinal damage and inflammatory cytokines. Blood 1997; 90: 3204–3213.
Korngold R, Marini JC, de Baca ME, Murphy GF, Giles-Komar J . Role of tumor necrosis factor-alpha in graft-versus-host disease and graft-versus-leukemia responses. Biol Blood Marrow Transplant 2003; 9: 292–303.
Sorror ML, Maris MB, Storer B, Sandmaier BM, Diaconescu R, Flowers C et al. Comparing morbidity and mortality of HLA-matched unrelated donor hematopoietic cell transplantation after nonmyeloablative and myeloablative conditioning: influence of pretransplant comorbidities. Blood 2004; 104: 961–968.
Valcarcel D, Martino R, Caballero D, Martin J, Ferra C, Nieto JB et al. Sustained remissions of high-risk acute myeloid leukemia and myelodysplastic syndrome after reduced-intensity conditioning allogeneic hematopoietic transplantation: chronic graft-versus-host disease is the strongest factor improving survival. J Clin Oncol 2008; 26: 577–584.
Acknowledgements
We are grateful to physicians, physicians’ assistants, and clinical staff for their dedicated care of the patients. FB is a senior research associate and YB a research director of the National Fund for Scientific Research (F.R.S.-FNRS) Belgium. The study was in part supported by funds from the FNRS, the Anti-Cancer Center from the University of Liége, and by the Belgian Federation against Cancer (FBC).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Willems, E., Humblet-Baron, S., Dengis, O. et al. Elevations of tumor necrosis factor receptor 1 at day 7 and acute graft-versus-host disease after allogeneic hematopoietic cell transplantation with nonmyeloablative conditioning. Bone Marrow Transplant 45, 1442–1448 (2010). https://doi.org/10.1038/bmt.2009.360
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2009.360
Keywords
This article is cited by
-
Associations between febrile neutropenia-related parameters and the risk of acute GVHD or non-relapse mortality after allogeneic hematopoietic stem cell transplantation
Bone Marrow Transplantation (2019)
-
Thinking Out of the Box—New Approaches to Controlling GVHD
Current Hematologic Malignancy Reports (2014)
-
Frequency of CD4+FOXP3+ regulatory T-cells at early stages after HLA-mismatched allogeneic hematopoietic SCT predicts the incidence of acute GVHD
Bone Marrow Transplantation (2013)
-
Implications of TNF-α in the pathogenesis and management of GVHD
International Journal of Hematology (2011)