Abstract
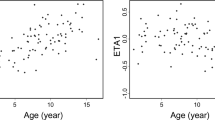
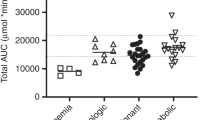
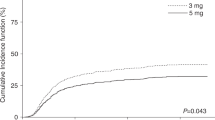
CsA is commonly used after haematological SCT (HSCT) as GVHD prophylaxis. In solid organ transplantation, area under the blood concentration vs time curve (AUC) correlates with clinical outcome. However, in HSCT, it has not been determined whether the AUC is superior to trough level monitoring to optimize clinical efficacy of CsA therapy. Therefore, the aim of this study was to investigate the relationships between CsA trough levels and/or AUC early after HSCT with clinical outcome. A total of 91 children (1.1–17.3 years) were treated consecutively with HSCT for a haematological malignancy. CsA trough levels were obtained and were used to estimate the AUC, retrospectively, with a NONMEM (Non-Linear Mixed Effects Modelling) method. Subsequently, these exposure parameters were correlated to the occurrence of acute GVHD, relapse risk (RR) and OS. Low CsA trough levels were found to correlate with the occurrence of acute GVHD. In addition, a CsA AUC over 3000 mcg h/l in AML patients was associated with a higher RR and a reduced OS. This was not the case for ALL patients. Thus, monitoring CsA exposure early after HSCT and adjusting the CsA dose to a predefined target trough level and AUC may provide a tool to influence GVHD/GVL balance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Storb R, Deeg HJ, Whitehead J, Appelbaum F, Beatty P, Bensinger W et al. Methotrexate and cyclosporine compared with cyclosporine alone for prophylaxis of acute graft versus host disease after marrow transplantation for leukemia. N Engl J Med 1986; 314: 729–735.
Ruutu T, Niederwieser D, Gratwohl A, Apperley JF . A survey of the prophylaxis and treatment of acute GVHD in Europe: a report of the European Group for Blood and Marrow, Transplantation (EBMT). Chronic Leukaemia Working Party of the EBMT. Bone Marrow Transplant 1997; 19: 759–764.
Cooney GF, Habucky K, Hoppu K . Cyclosporin pharmacokinetics in paediatric transplant recipients. Clin Pharmacokinet 1997; 32: 481–495.
Schrauder A, Saleh S, Sykora KW, Hoy H, Welte K, Boos J et al. Pharmacokinetic monitoring of intravenous cyclosporine A in pediatric stem-cell transplant recipients. The trough level is not enough. Pediatr Transplant 2009; 13: 444–450.
Knight SR, Morris PJ . The clinical benefits of cyclosporine C2-level monitoring: a systematic review. Transplantation 2007; 83: 1525–1535.
Lindholm A, Kahan BD . Influence of cyclosporine pharmacokinetics, trough concentrations, and AUC monitoring on outcome after kidney transplantation. Clin Pharmacol Ther 1993; 54: 205–218.
del Mar Fernandez De Gatta M, Santos-Buelga D, Dominguez-Gil A, Garcia MJ . Immunosuppressive therapy for paediatric transplant patients: pharmacokinetic considerations. Clin Pharmacokinet 2002; 41: 115–135.
Vossen JM, Heidt PJ, van den BH, Gerritsen EJ, Hermans J, Dooren LJ . Prevention of infection and graft-versus-host disease by suppression of intestinal microflora in children treated with allogeneic bone marrow transplantation. Eur J Clin Microbiol Infect Dis 1990; 9: 14–23.
Glucksberg H, Storb R, Fefer A, Buckner CD, Neiman PE, Clift RA et al. Clinical manifestations of graft-versus-host disease in human recipients of marrow from HLA-matched sibling donors. Transplantation 1974; 18: 295–304.
Shulman HM, Sullivan KM, Weiden PL, McDonald GB, Striker GE, Sale GE et al. Chronic graft-versus-host syndrome in man. A long-term clinicopathologic study of 20 Seattle patients. Am J Med 1980; 69: 204–217.
Willemze AJ, Cremers SC, Schoemaker RC, Lankester AC, den Hartigh J, Burggraaf J et al. Ciclosporin kinetics in children after stem cell transplantation. Br J Clin Pharmacol 2008; 66: 539–545.
Beal SL, Sheiner LB, Boeckmann AJ (eds). NONMEM Users Guides (1989–2006). Icon Development Solutions, Ellicott City, MD, USA, 2007.
Anderson BJ, Holford NH . Mechanism-based concepts of size and maturity in pharmacokinetics. Annu Rev Pharmacol Toxicol 2008; 48: 303–332.
Ghalie R, Fitzsimmons WE, Weinstein A, Manson S, Kaizer H . Cyclosporine monitoring improves graft-versus-host disease prophylaxis after bone marrow transplantation. Ann Pharmacother 1994; 28: 379–383.
Martin P, Bleyzac N, Souillet G, Galambrun C, Bertrand Y, Maire PH et al. Clinical and pharmacological risk factors for acute graft-versus-host disease after paediatric bone marrow transplantation from matched-sibling or unrelated donors. Bone Marrow Transplant 2003; 32: 881–887.
Przepiorka D, Shapiro S, Schwinghammer TL, Bloom EJ, Rosenfeld CS, Shadduck RK et al. Cyclosporine and methylprednisolone after allogeneic marrow transplantation: association between low cyclosporine concentration and risk of acute graft-versus-host disease. Bone Marrow Transplant 1991; 7: 461–465.
Punnett A, Sung L, Price V, Das P, Diezi M, Doyle J et al. Achievement of target cyclosporine concentrations as a predictor of severe acute graft versus host disease in children undergoing hematopoietic stem cell transplantation and receiving cyclosporine and methotrexate prophylaxis. Ther Drug Monit 2007; 29: 750–757.
Yee GC, Self SG, McGuire TR, Carlin J, Sanders JE, Deeg HJ . Serum cyclosporine concentration and risk of acute graft-versus-host disease after allogeneic marrow transplantation. N Engl J Med 1988; 319: 65–70.
Cole E, Keown P, Landsberg D, Halloran P, Shoker A, Rush D et al. Safety and tolerability of cyclosporine and cyclosporine microemulsion during 18 months of follow-up in stable renal transplant recipients: a report of the Canadian Neoral Renal Study Group. Transplantation 1998; 65: 505–510.
International Neoral Renal Transplantation Study Group. Cyclosporine microemulsion (Neoral) absorption profiling and sparse-sample predictors during the first 3 months after renal transplantation. Am J Transplant 2002; 2: 148–156.
Barkholt L, Remberger M, Bodegard H, Ringden O, Bottiger Y . Cyclosporine A (CsA) 2-h concentrations vary between patients without correlation to graft-versus-host disease after allogeneic haematopoietic stem cell transplantation. Bone Marrow Transplant 2007; 40: 683–689.
Locatelli F, Zecca M, Rondelli R, Bonetti F, Dini G, Prete A et al. Graft versus host disease prophylaxis with low-dose cyclosporine-A reduces the risk of relapse in children with acute leukemia given HLA-identical sibling bone marrow transplantation: results of a randomized trial. Blood 2000; 95: 1572–1579.
Porter DL, Collins RH, Hardy C, Kernan NA, Drobyski WR, Giralt S et al. Treatment of relapsed leukemia after unrelated donor marrow transplantation with unrelated donor leukocyte infusions. Blood 2000; 95: 1214–1221.
Kolb HJ, Schattenberg A, Goldman JM, Hertenstein B, Jacobsen N, Arcese W et al. Graft-versus-leukemia effect of donor lymphocyte transfusions in marrow grafted patients. Blood 1995; 86: 2041–2050.
Cardoso AA, Schultze JL, Boussiotis VA, Freeman GJ, Seamon MJ, Laszlo S et al. Pre-B acute lymphoblastic leukemia cells may induce T-cell anergy to alloantigen. Blood 1996; 88: 41–48.
Pons G, Treluyer JM, Dimet J, Merle Y . Potential benefit of Bayesian forecasting for therapeutic drug monitoring in neonates. Ther Drug Monit 2002; 24: 9–14.
Anderson BJ, Allegaert K, Holford NH . Population clinical pharmacology of children: general principles. Eur J Pediatr 2006; 165: 741–746.
Acknowledgements
We thank the medical, nursing and ancillary staff of the IHOBA unit, Department of Paediatrics, LUMC, for their care and management of patients undergoing bone marrow transplantation.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Willemze, A., Press, R., Lankester, A. et al. CsA exposure is associated with acute GVHD and relapse in children after SCT. Bone Marrow Transplant 45, 1056–1061 (2010). https://doi.org/10.1038/bmt.2009.299
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/bmt.2009.299
Keywords
This article is cited by
-
A decision support tool to find the best cyclosporine dose when switching from intravenous to oral route in pediatric stem cell transplant patients
European Journal of Clinical Pharmacology (2020)
-
Improved outcome of children transplanted for high-risk leukemia by using a new strategy of cyclosporine-based GVHD prophylaxis
Bone Marrow Transplantation (2016)
-
Pharmacokinetics, Pharmacodynamics and Pharmacogenomics of Immunosuppressants in Allogeneic Haematopoietic Cell Transplantation: Part I
Clinical Pharmacokinetics (2016)
-
Bayesian approach for the estimation of cyclosporine area under the curve using limited sampling strategies in pediatric hematopoietic stem cell transplantation
Theoretical Biology and Medical Modelling (2014)